Abstract
Objective:
To assess whether weight gain above or below Institute of Medicine (IOM) recommended amounts in an ethnically diverse obstetric population with normal glucose tolerance is associated with differences in neonatal adiposity.
Study Design:
In this prospective cohort study, healthy women with normal glucose tolerance based on the International Association of Diabetes and Pregnancy Study Groups guidelines were enrolled. Gestational weight at multiple time points were collected. Neonatal adiposity was measured by air displacement plethysmography at 24 to 72 h of life. Analyses included Fisher’s exact test, analysis of variance and a trajectory analysis using a group-based weight gain trajectory model with a censored normal distribution.
Results:
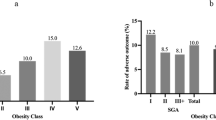
Overweight and obese women were more likely to exceed IOM weight gain guidelines. Regardless, there was no significant difference in %body fat of neonates born to mothers who either met or exceeded gestational weight gain (GWG) guidelines. GWG timing influenced neonatal anthropometrics: women who gained excessively by the first prenatal visit had neonates with significantly higher birth weight (3.91 vs 3.45 kg, P<0.001) and %body fat (13.7 vs 10.9%, P=0.0001) compared with women who had steady and moderate GWG.
Conclusion:
Avoidance of excessive GWG in the first trimester may prevent high amounts of neonatal adiposity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Catalano PM, Farrell K, Thomas A, Huston-Presley L, Mencin P, de Mouzon SH et al. Perinatal risk factors for childhood obesity and metabolic dysregulation. Am J Clin Nutr 2009; 90 (5): 1303–1313.
Vohr BR, McGarvey ST, Tucker R . Effects of maternal gestational diabetes on offspring adiposity at 4-7 years of age. Diabetes Care 1999; 22 (8): 1284–1291.
Freinkel N . Banting Lecture. Of pregnancy and progeny. Diabetes 1980; 29 (12): 1023–1035.
Metzger BE, Phelps RL, Freinkel N, Navickas IA . Effects of gestational diabetes on diurnal profiles of plasma glucose, lipids, and individual amino acids. Diabetes Care 1980; 3 (3): 402–409.
Sewell MF, Huston-Presley L, Super DM, Catalano P . Increased neonatal fat mass, not lean body mass, is associated with maternal obesity. Am J Obstet Gynecol 2006; 195 (4): 1100–1103.
HAPO Study Cooperative Research Group. Hyperglycaemia and Adverse Pregnancy Outcome (HAPO) Study: associations with maternal body mass index. BJOG 2010; 117 (5): 575–584.
Badon SE, Dyer AR, Josefson JL HAPO Study Cooperative Research Group. Gestational weight gain and neonatal adiposity in the Hyperglycemia and Adverse Pregnancy Outcome study-North American region. Obesity (Silver Spring) 2014; 22 (7): 1731–1738.
Hull HR, Thornton JC, Ji Y, Paley C, Rosenn B, Mathews P et al. Higher infant body fat with excessive gestational weight gain in overweight women. Am J Obstet Gynecol 2011; 205 (3): 211–217.
Whitelaw A . Subcutaneous fat in newborn infants of diabetic mothers: an indication of quality of diabetic control. Lancet 1977; 1 (8001): 15–18.
Oken E, Gillman MW . Fetal origins of obesity. Obes Res 2003; 11 (4): 496–506.
Pettitt DJ, Knowler WC, Bennett PH, Aleck KA, Baird HR . Obesity in offspring of diabetic Pima Indian women despite normal birth weight. Diabetes Care 1987; 10 (1): 76–80.
Catalano PM, Thomas A, Huston-Presley L, Amini SB . Increased fetal adiposity: a very sensitive marker of abnormal in utero development. Am J Obstet Gynecol 2003; 189 (6): 1698–1704.
Silverman BL, Landsberg L, Metzger BE . Fetal hyperinsulinism in offspring of diabetic mothers. Association with the subsequent development of childhood obesity. Ann N Y Acad Sci 1993; 699: 36–45.
Waters TP, Huston-Presley L, Catalano PM . Neonatal body composition according to the revised institute of medicine recommendations for maternal weight gain. J Clin Endocrinol Metab 2012; 97 (10): 3648–3654.
Starling AP, Brinton JT, Glueck DH, Shapiro AL, Harrod CS, Lynch AM et al. Associations of maternal BMI and gestational weight gain with neonatal adiposity in the Healthy Start study. Am J Clin Nutr 2015; 101 (2): 302–309.
International Association of Diabetes and Pregnancy Study Groups Consensus Panel, Metzger BE International Association of Diabetes and Pregnancy Study Groups Consensus Panel, Gabbe SG International Association of Diabetes and Pregnancy Study Groups Consensus Panel, Persson B International Association of Diabetes and Pregnancy Study Groups Consensus Panel, Buchanan TA International Association of Diabetes and Pregnancy Study Groups Consensus Panel, Catalano PA et alInternational Association of Diabetes and Pregnancy Study Groups Consensus Panel. International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care 2010; 33 (3): 676–682.
Urlando A, Dempster P, Aitkens S . A new air displacement plethysmograph for the measurement of body composition in infants. Pediatr Res 2003; 53 (3): 486–492.
Jones BL, Nagin DS, Roeder KA . SAS procedure based on mixture models for estimating developmental trajectories. Sociol Methods Res 2001; 29 (3): 374–393.
Institute of Medicine Weight Gain During Pregnancy: Reexamining the Guidelines. The National Academies Press: Washington, DC, USA, 2009.
Davenport MH, Ruchat SM, Giroux I, Sopper MM, Mottola MF . Timing of excessive pregnancy-related weight gain and offspring adiposity at birth. Obstet Gynecol 2013; 122 (2): 255–261.
Catalano PM, Thomas AJ, Avallone DA, Amini SB . Anthropometric estimation of neonatal body composition. Am J Obstet Gynecol 1995; 173 (4): 1176–1181.
Crozier SR, Inskip HM, Godfrey KM, Cooper C, Harvey NC, Cole ZA et al. Weight gain in pregnancy and childhood body composition: findings from the Southampton Women's Survey. Am J Clin Nutr 2010; 91 (6): 1745–1751.
Metzger BE, Lowe LP, Dyer AR, Trimble ER, Chaovarindr U, Coustan DR et al. Hyperglycemia and adverse pregnancy outcomes. N Engl J Med 2008; 358 (19): 1991–2002.
Walsh JM, McGowan CA, Mahony RM, Foley ME, McAuliffe FM . Obstetric and metabolic implications of excessive gestational weight gain in pregnancy. Obesity (Silver Spring) 2014; 22 (7): 1594–1600.
Herring SJ, Oken E, Rifas-Shiman SL, Rich-Edwards JW, Stuebe AM, Kleinman KP et al. Weight gain in pregnancy and risk of maternal hyperglycemia. Am J Obstet Gynecol 2009; 201 (1): 61–67.
Crume TL, Shapiro AL, Brinton JT, Glueck DH, Martinez M, Kohn M et al. Maternal fuels and metabolic measures during pregnancy and neonatal body composition: the healthy start study. J Clin Endocrinol Metab 2015; 100 (4): 1672–1680.
Catalano PM, Presley L, Minium J, Hauguel-de Mouzon S . Fetuses of obese mothers develop insulin resistance in utero. Diabetes Care 2009; 32 (6): 1076–1080.
Josefson JL, Zeiss DM, Rademaker AW, Metzger BE . Maternal leptin predicts adiposity of the neonate. Horm Res Paediatr 2014; 81 (1): 13–19.
Lawlor DA, Relton C, Sattar N, Nelson SM . Maternal adiposity—a determinant of perinatal and offspring outcomes? Nat Rev Endocrinol 2012; 8 (11): 679–688.
Chawla R, Badon SE, Rangarajan J, Reisetter AC, Armstrong LL, Lowe LP et al. Genetic risk score for prediction of newborn adiposity and large-for-gestational-age birth. J Clin Endocrinol Metab 2014; 99 (11): E2377–E2386.
Gale C, Logan KM, Jeffries S, Parkinson JR, Santhakumaran S, Uthaya S et al. Sexual dimorphism in relation to adipose tissue and intrahepatocellular lipid deposition in early infancy. Int J Obes (Lond) 2015; 39 (4): 629–632.
Committee opinion no. 504: screening and diagnosis of gestational diabetes mellitus. Obstet Gynecol 2011; 118 (3): 751–753.
Acknowledgements
We thank the participants who volunteered for this study. Funding for this study was provided by grant number K12 HD055884 from the Eunice Kennedy Shriver National Institute of Child Health and Human Development. We would like to acknowledge the critical review and comments provided by Alan Peaceman, MD and Linda Van Horn, RD, PhD, both at Northwestern University Feinberg School of Medicine. The funding source had no involvement in the study design, data analysis and interpretation, or writing of this manuscript.
Disclaimer
The findings and conclusions in this article are those of the authors and do not necessarily represent the views of Planned Parenthood Federation of America.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Josefson, J., Simons, H., Zeiss, D. et al. Excessive gestational weight gain in the first trimester among women with normal glucose tolerance and resulting neonatal adiposity. J Perinatol 36, 1034–1038 (2016). https://doi.org/10.1038/jp.2016.145
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2016.145
This article is cited by
-
Neonatal body composition by air displacement plethysmography in healthy term singletons: a systematic review
BMC Pediatrics (2019)
-
A reduction in sedentary behaviour in obese women during pregnancy reduces neonatal adiposity: the DALI randomised controlled trial
Diabetologia (2019)
-
Air displacement plethysmography (pea pod) in full-term and pre-term infants: a comprehensive review of accuracy, reproducibility, and practical challenges
Maternal Health, Neonatology and Perinatology (2018)
-
Maternal and neonatal demographics of macrosomic infants admitted to the neonatal intensive care unit
Journal of Perinatology (2017)