Abstract
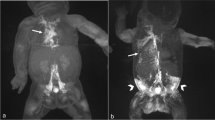
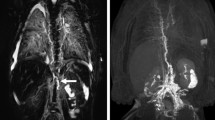
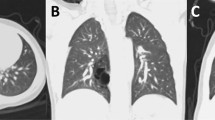
Historically, congenital pulmonary lymphangiectasia (CPL) has had poor treatment outcomes despite aggressive therapy. There are recent reports of ethiodized oil (Lipiodol) lymphangiography successfully treating lymphatic leakage in adults. In this report, we describe two infants with CPL complicated by chylothoraces successfully treated by instillation of ethiodized oil into the lymphatic system. Congenital atresia of the thoracic duct was demonstrated on the lymphangiogram in both patients before treatment. Both patients have shown good short-term outcomes without supplemental oxygen or fat restricted diets at 9 months of age. Ethiodized oil lymphangiography represents a new treatment modality for some patients with CPL.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Esther CR Jr, Barker PM . Pulmonary lymphangiectasia: diagnosis and clinical course. Pediatr Pulmonol 2004; 38 (4): 308–313.
Bouchard S, Di Lorenzo M, Youssef S, Simard P, Lapierre JG . Pulmonary lymphangiectasia revisited. J Pediatr Surg 2000; 35 (5): 796–800.
Bellini C, Boccardo F, Campisi C, Toma P, Taddei G, Villa G et al. Congenital pulmonary lymphangiectasia. Orphanet J Rare Dis 2006; 1: 43.
Alejandre-Lafont E, Krompiec C, Rau WS, Krombach GA . Effectiveness of therapeutic lymphography on lymphatic leakage. Acta Radiol 2011; 52 (3): 305–311.
Matsumoto T, Yamagami T, Kato T, Hirota T, Yoshimatsu R, Masunami T et al. The effectiveness of lymphangiography as a treatment method for various chyle leakages. Br J Radiol 2009; 82 (976) 286–290.
Nadolski GJ, Itkin M . Feasibility of ultrasound-guided intranodal lymphangiogram for thoracic duct embolization. J Vasc Interv Radiol 2012; 23 (5): 613–616.
Clemens R, Kreindel T, Lillis A, Alomari A . Characterization of central conducting lymphatic anomalies. Pediatr Radiol 2013; 43 (Suppl 2) S319.
Rajebi MR, Chaudry G, Padua HM, Dillon B, Yilmaz S, Arnold RW et al. Intranodal lymphangiography: feasibility and preliminary experience in children. J Vasc Interv Radiol 2011; 22 (9): 1300–1305.
Lo CM, Ngan H, Tso WK, Liu CL, Lam CM, Poon RT et al. Randomized controlled trial of transarterial lipiodol chemoembolization for unresectable hepatocellular carcinoma. Hepatology 2002; 35: 1164–1171.
Yamagami T, Masunami T, Kato T, Tanaka O, Hirota T, Nomoto T et al. Spontaneous healing of chyle leakage after lymphangiography. Br J Radiol 2005; 78 (933): 854–857.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Gray, M., Kovatis, K., Stuart, T. et al. Treatment of congenital pulmonary lymphangiectasia using ethiodized oil lymphangiography. J Perinatol 34, 720–722 (2014). https://doi.org/10.1038/jp.2014.71
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2014.71
This article is cited by
-
Lymphatic anomalies in congenital heart disease
Pediatric Radiology (2022)
-
Neonatal lymphatic flow disorders: impact of lymphatic imaging and interventions on outcomes
Journal of Perinatology (2021)
-
Imaging of fetal lymphangiectasias: prenatal and postnatal imaging findings
Pediatric Radiology (2020)
-
Chyloptysis with chylopericardium, a rare case and mini-review
BMC Pulmonary Medicine (2018)