Abstract
Objective:
To assess pain and compare its severity in preterm infants during application of nasal-continuous positive airway pressure (nCPAP) and heated, humidified high-flow nasal cannulae (HHHFNC).
Study Design:
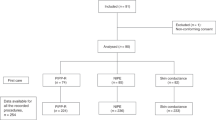
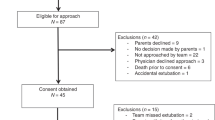
An observational cross-sectional study. Sixty preterm infants, categorized into nCPAP (n=37) and HHHFNC groups (n=23). Pain response was assessed using Premature Infant Pain Profile (PIPP), duration of first cry and salivary-cortisol concentrations.
Result:
The PIPP scores were significantly higher in the nCPAP compared with HHHFNC group (10 (7–12) vs 4 (2–6), P<0.01). None of the infants in the HHHFNC group had severe pain defined as a PIPP score >12, compared with 5 (13.5%) infants in the nCPAP group. Salivary-cortisol concentrations were significantly higher in nCPAP group compared with the HHHFNC group (5.0 (3.6–5.9) vs 1.6 (1.0–2.3) nmol l−1, P<0.01). A lower incidence of cry was observed for infants in the HHHFNC group compared with the nCPAP group (11 (47.8%) vs 30 (81.1%), P<0.001), however, the duration of first cry was not significantly different between groups. The respiratory rate was significantly lower after application of HHHFNC compared with nCPAP (P<0.001). There were no significant differences between groups with regard to fraction of inspired oxygen (FiO2), oxygen saturation by pulse oximeter (SpO2) and heart rate.
Conclusion:
The application of HHHFNC in preterm infants is associated with less pain compared with nCPAP, as it is associated with less PIPP scores and lower salivary-cortisol concentrations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Vitaliti SM, Costantino G, Li Puma L, Re MP, Vergara B, Pinello G . Painful procedures in the NICU. J Matern Fetal Neonatal Med 2012; 25 (Suppl 4): 146–147.
American Academy of Pediatrics Committee on Fetus and Newborn; American Academy of Pediatrics Section on Surgery; Canadian Paediatric Society Fetus and Newborn Committee American Academy of Pediatrics Committee on Fetus and Newborn; American Academy of Pediatrics Section on Surgery; Canadian Paediatric Society Fetus and Newborn Committee, Batton DG, Barrington KJ, Wallman C . Prevention and management of pain in the neonate: an update. Pediatrics 2006; 118: 2231–2241.
Simons SH, van Dijk M, Anand KS, Roofthooft D, van Lingen RA, Tibboel D . Do we still hurt newborn babies? A prospective study of procedural pain and analgesia in neonates. Arch Pediatr Adolesc Med 2003; 157: 1058–1064.
Grunau RE, Holsti L, Peters JW . Long-term consequences of pain in human neonates. Semin Fetal Neonatal Med 2006; 11: 268–275.
Fischer C, Bertelle V, Hohlfeld J, Forcada-Guex M, Stadelmann-Diaw C, Tolsa J-Fo . Nasal trauma due to continuous positive airway pressure in neonates. Arch Dis Child-Fetal Neonatal Ed 2010; 95: F447–F451.
Yong SC, Chen SJ, Boo NY . Incidence of nasal trauma associated with nasal prong versus nasal mask during continuous positive airway pressure treatment in very low birthweight infants: a randomized control study. Arch Dis Child Fetal Neonatal Ed 2005; 90: F480–F483.
Collins CL, Barfield C, Horne RS, Davis PG . A comparison of nasal trauma in preterm infants extubated to either heated humidified high-flow nasal cannulae or nasal continuous positive airway pressure. Eur J Pediatr 2014; 173: 181–186.
Haq I, Gopalakaje S, Fenton AC, McKean MC, JO'Brien C, Brodlie M . The evidence for high flow nasal cannula devices in infants. Pediatr Respir Rev 2014; 15: 124–134.
Yoder BA, Stoddard RA, Li M, King J, Dirnberger DR, Abbasi S . Heated humidified high-flow nasal cannula versus nasal CPAP for respiratory support in neonates. Pediatrics 2013; 131: e1482–e1490.
Collins CL, Holberton JR, Barfield C, Davis PG . A randomized controlled trial to compare heated humidified high-flow nasal cannulae with nasal continuous positive airway pressure postextubation in premature infants. J Pediatr 2013; 162: 949–954.
Manley BJ, Owen LS, Doyle LW, Andersen CC, Cartwright DW, Pritchard MA et al. High-flow nasal cannulae in very preterm infants after extubation. N Eng J Med 2013; 369: 1425–1433.
Fernandez-Alvarez JR, Gandhi RS, Amess P, Mahoney L, Watkins R, Rabe H . Heated humidified high-flow nasal cannula versus low-flow nasal cannula as weaning mode from nasal CPAP in infants ≤28 weeks of gestation. Eur J Pediatr 2014; 173: 93–98.
Stevens B, Johnston C, Petryshen P, Taddio A . Premature infant pain profile: development and initial validation. Clin J Pain 1996; 12: 13–22.
Maxwell LG, Malavolta CP, Fraga MV . Assessment of pain in the Neonate. Clin Perinatol 2013; 40: 457–459.
Ballantyne M, Stevens B, McAllister M, Dionne K, Jack A . Validation of the premature infant pain profile in the clinical setting. Clin J Pain 1999; 15: 297–303.
Klingenberg C, Pettersen M, Hansen EA, Gustavsen LJ, Dahl IA, Leknessund A et al. Patient comfort during treatment with heated humidified high flow nasal cannulae versus nasal continuous positive airway pressure: a randomized cross-over trial. Arch Dis Child Fetal Neonatal Ed 2014; 99: F134–F137.
Debillon T, Zupan V, Ravault N, Magny J, Dehan M . Development and initial validation of the EDIN scale, a new tool for assessing prolonged pain in preterm infants. Arch Dis Child-Fetal Neonatal Ed 2001; 85: F36–F41.
Arias MC, Guinsburg R . Differences between uni-and multidimensional scales for assessing pain in term newborn infants at the bedside. Clinics (Sao Paulo 2012; 67: 1165–1170.
South MM, Strauss RA, South AP, Boggess JF, Thorp JM . The use of non-nutritive sucking to decrease the physiologic pain response during neonatal circumcision: a randomized controlled trial. Am J Obstet Gynecol 2005; 193: 537–542.
Slater R, Cornelissen L, Fabrizi L, Patten D, Yoxen J, Worley A et al. Oral sucrose as an analgesic drug for procedural pain in newborn infants: a randomised controlled trial. Lancet 2010; 376 (9748): 1225–1232.
Cohen AM, Cook N, Harris MC, Ying GS, Binenbaum G . The pain response to mydriatic eyedrops in preterm infants. J Perinatol 2013; 33: 462–465.
Gallo AM . The fifth vital sign: implementation of the Neonatal Infant Pain Scale. J Obstet Gynecol Neonatal Nurs 2003; 32: 199–206.
Cabral DM, Antonini SR, Custódio RJ, Martinelli CE Jr, da Silva CA . Measurement of salivary cortisol as a marker of stress in newborns in a neonatal intensive care unit. Horm Res Pediatr 2013; 79: 373–378.
Herrington CJ, Olomu IN, Geller SM . Salivary cortisol as indicators of pain in preterm infants: a pilot study. Clin Nurs Res 2004; 13: 53–68.
Aardal E, Holm AC . Cortisol in saliva-reference ranges and relation to cortisol in serum. Eur J Clin Chem Clin Biochem 1995; 33: 927–932.
Calixto C, Martinez FE, Jorge SM, Moreira AC, Martinelli CE Jr . Correlation between plasma and salivary cortisol levels in preterm infants. J Pediatr 2002; 140: 116–118.
Ben-Aryeh H, Lapid S, Szargel R, Benderly A, Gutman D . Composition of whole unstimulated saliva of human infants. Arch Oral Biol 1984; 29: 357–362.
Joyce BA, Keck JF, Gerkensmeyer J . Evaluation of pain management interventions for neonatal circumcision pain. J Pediatr Health Care 2001; 15: 105–114.
Talge NM, Donzella B, Kryzer EM, Gierens A, Gunnar MR . It's not that bad: error introduced by oral stimulants in salivary cortisol research. Dev Psychobiol 2005; 47: 369–376.
Bonner KM, Mainous RO . The nursing care of the infant receiving bubble CPAP therapy. Adv Neonatal Care 2008; 8: 78–95.
Stevens B, Gibbins S, Franck LS . Treatment of pain in the neonatal intensive care unit. Pediatr Clin North Am 2000; 47: 633–650.
Enders J, Gebauer C, Pulzer F, Robel-Tillig E, Knupfer M . Analgosedation with low-dose morphine for preterm infants with CPAP: risks and benefits. Acta Pediatrica 2008; 97: 880–883.
Schuurmans J, Benders M, Lemmers P, van Bel F . Neonatal morphine in extremely and very preterm neonates: its effect on the developing brain—a review. J Matern Fetal Neonatal Med 2014 (e-pub ahead of print 29 April 2014.
Pillai Riddell RR, Racine NM, Turcotte K, Uman LS, Horton RE, Din Osmun L et al. Non-pharmacological management of infant and young child procedural pain. Cochrane Database Syst Rev 2011; (10): CD006275.
Holsti L, Grunau RE . Considerations for using sucrose to reduce procedural pain in preterm infants. Pediatrics 2010; 125: 1042–1047.
Shetty S, Greenough A . Review finds insufficient evidence to support the routine use of heated, humidified high-flow nasal cannula use in neonates. Acta Pediatr 2014; 103 (9): 898–903.
Acknowledgements
We thank Sabah Abdelnaser and Saber Elsheikh for their help in laboratory assessment of salivary cortisol, nursing staff of NICU, Mansoura University Children's Hospital, for their help in assessment of PIPP scores and patient recruitment, and all the parents and infants who took part in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Osman, M., Elsharkawy, A. & Abdel-Hady, H. Assessment of pain during application of nasal-continuous positive airway pressure and heated, humidified high-flow nasal cannulae in preterm infants. J Perinatol 35, 263–267 (2015). https://doi.org/10.1038/jp.2014.206
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2014.206
This article is cited by
-
Delivery Room Respiratory Stabilization of Preterm Neonates: A Randomized, Controlled Trial
Indian Journal of Pediatrics (2022)
-
Clinical validation of the Neonatal Infant Stressor Scale with preterm infant salivary cortisol
Pediatric Research (2020)
-
Enteral Nutrition Tolerance And REspiratory Support (ENTARES) Study in preterm infants: study protocol for a randomized controlled trial
Trials (2019)
-
Nasal high flow treatment in preterm infants
Maternal Health, Neonatology and Perinatology (2017)
-
Nasal high flow therapy in very low birth weight infants with mild respiratory distress syndrome: a single center experience
Italian Journal of Pediatrics (2017)