Abstract
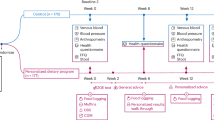
We performed a randomised, placebo-controlled, crossover study to examine the effects of sodium and potassium supplementation on blood pressure (BP) and arterial stiffness in untreated (pre)hypertensive individuals. During the study, subjects were on a fully controlled diet that was relatively low in sodium and potassium. After a 1-week run-in period, subjects received capsules with supplemental sodium (3 g d−1, equals 7.6 g d−1 of salt), supplemental potassium (3 g d−1) or placebo, for 4 weeks each, in random order. Fasting office BP, 24-h ambulatory BP and measures of arterial stiffness were assessed at baseline and every 4 weeks. Of 37 randomized subjects, 36 completed the study. They had a mean pre-treatment BP of 145/81 mm Hg and 69% had systolic BP ⩾140 mm Hg. Sodium excretion was increased by 98 mmol per 24 h and potassium excretion by 63 mmol per 24 h during active interventions, compared with placebo. During sodium supplementation, office BP was significantly increased by 7.5/3.3 mm Hg, 24-h BP by 7.5/2.7 mm Hg and central BP by 8.5/3.6 mm Hg. During potassium supplementation, 24-h BP was significantly reduced by 3.9/1.6 mm Hg and central pulse pressure by 2.9 mm Hg. Pulse wave velocity and augmentation index were not significantly affected by sodium or potassium supplementation. In conclusion, increasing the intake of sodium caused a substantial increase in BP in subjects with untreated elevated BP. Increased potassium intake, on top of a relatively low-sodium diet, had a beneficial effect on BP. Arterial stiffness did not materially change during 4-week interventions with sodium or potassium.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
He J, Whelton PK . Elevated systolic blood pressure as a risk factor for cardiovascular and renal disease. J Hypertens Suppl 1999; 17: S7–13.
Lewington S, Clarke R, Qizilbash N, Peto R, Collins R . Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet 2002; 360: 1903–1913.
Kearney PM, Whelton M, Reynolds K, Muntner P, Whelton PK, He J . Global burden of hypertension: Analysis of worldwide data. Lancet 2005; 365: 217–223.
Aburto NJ, Ziolkovska A, Hooper L, Elliott P, Cappuccio FP, Meerpohl JJ . Effect of lower sodium intake on health: systematic review and meta-analyses. BMJ 2013; 346: f1326.
Cutler JA, Follmann D, Allender PS . Randomised trials of sodium reduction: an overview. Am J Clin Nutr 1997; 65: 643S–651S.
Geleijnse JM, Kok FJ, Grobbee DE . Blood pressure response to changes in sodium and potassium intake: a metaregression analysis of randomised trials. J Hum Hypertens 2003; 17: 471–480.
He FJ, Li J, Macgregor GA . Effect of longer term modest salt reduction on blood pressure: Cochrane systematic review and meta-analysis of randomised trials. BMJ 2013; 346: f1325.
Midgley JP, Matthew AG, Greenwood CM, Logan AG . Effect of reduced dietary sodium on blood pressure: a meta-analysis of randomised controlled trials. JAMA 1996; 275: 1590–1597.
Brown IJ, Tzoulaki I, Candeias V, Elliott P . Salt intakes around the world: implications for public health. Int J Epidemiol 2009; 38: 791–813.
WHO. Guideline: Sodium intake for adults and children. Geneva, World Health Organization (WHO), 2012.
US Department of Agriculture and US Department of Health and Human Services Dietary Guidelines for Americans, 2010. 7th Edition, Washington, DC: US Government Printing Office, 2010.
He FJ, MacGregor GA . How far should salt intake be reduced? Hypertension 2003; 42: 1093–1099.
van Mierlo LAJ, Greyling A, Zock PL, Kok FJ, Geleijnse JM . Suboptimal potassium intake and potential impact on population blood pressure. Arch Intern Med 2010; 170: 1501–1502.
National Research Council Dietary Reference Intakes for Water, Potassium, Sodium, Chloride, and Sulfate. Washington, DC: The National Academies Press, 2005.
Aburto NJ, Hanson S, Gutierrez H, Hooper L, Elliott P, Cappuccio FP . Effect of increased potassium intake on cardiovascular risk factors and disease: systematic review and meta-analyses. BMJ 2013; 346: f1378.
Vlachopoulos C, Aznaouridis K, Stefanadis C . Prediction of cardiovascular events and all-cause mortality with arterial stiffness: a systematic review and meta-analysis. J Am Coll Cardiol 2010; 55: 1318–1327.
Laurent S, Cockcroft J, Van Bortel L, Boutouyrie P, Giannattasio C, Hayoz D et al. Expert consensus document on arterial stiffness: methodological issues and clinical applications. Eur Heart J 2006; 27: 2588–2605.
Dickinson KM, Keogh JB, Clifton PM . Effects of a low-salt diet on flow-mediated dilatation in humans. Am J Clin Nutr 2009; 89: 485–490.
Dickinson KM, Clifton PM, Keogh JB . A reduction of 3 g/day from a usual 9 g/day salt diet improves endothelial function and decreases endothelin-1 in a randomised crossover study in normotensive overweight and obese subjects. Atherosclerosis 2014; 233: 32–38.
Gates PE, Tanaka H, Hiatt WR, Seals DR . Dietary sodium restriction rapidly improves large elastic artery compliance in older adults with systolic hypertension. Hypertension 2004; 44: 35–41.
He FJ, Marciniak M, Visagie E, Markandu ND, Anand V, Dalton RN et al. Effect of modest salt reduction on blood pressure, urinary albumin, and pulse wave velocity in white, black, and asian mild hypertensives. Hypertension 2009; 54: 482–488.
Jablonski KL, Fedorova OV, Racine ML, Geolfos CJ, Gates PE, Chonchol M et al. Dietary sodium restriction and association with urinary marinobufagenin, blood pressure, and aortic stiffness. Clin J Am Soc Nephrol 2013; 8: 1952–1959.
Seals DR, Tanaka H, Clevenger CM, Monahan KD, Reiling MJ, Hiatt WR et al. Blood pressure reductions with exercise and sodium restriction in postmenopausal women with elevated systolic pressure: role of arterial stiffness. J Am Coll Cardiol 2001; 38: 506–513.
Todd AS, Macginley RJ, Schollum JB, Johnson RJ, Williams SM, Sutherland WH et al. Dietary salt loading impairs arterial vascular reactivity. Am J Clin Nutr 2010; 91: 557–564.
Todd AS, Macginley RJ, Schollum JBW, Williams SM, Sutherland WHF, Mann JI et al. Dietary sodium loading in normotensive healthy volunteers does not increase arterial vascular reactivity or blood pressure. Nephrology (Carlton) 2012; 17: 249–256.
Berry SE, Mulla UZ, Chowienczyk PJ, Sanders TAB . Increased potassium intake from fruit and vegetables or supplements does not lower blood pressure or improve vascular function in uk men and women with early hypertension: a randomised controlled trial. Br J Nutr 2010; 104: 1839–1847.
Graham UM, McCance DR, Young IS, Mullan KR . A randomised controlled trial evaluating the effect of potassium supplementation on vascular function and the renin-angiotensin-aldosterone system. J Hum Hypertens 2014; 28: 333–339.
He FJ, Marciniak M, Carney C, Markandu ND, Anand V, Fraser WD et al. Effects of potassium chloride and potassium bicarbonate on endothelial function, cardiovascular risk factors, and bone turnover in mild hypertensives. Hypertension 2010; 55: 681–688.
Matthesen SK, Larsen T, Vase H, Lauridsen TG, Pedersen EB . Effect of potassium supplementation on renal tubular function, ambulatory blood pressure and pulse wave velocity in healthy humans. Scand J Clin Lab Invest 2012; 72: 78–86.
Siebelink E, Geelen A, de Vries JHM . Self-reported energy intake by ffq compared with actual energy intake to maintain body weight in 516 adults. Br J Nutr 2011; 106: 274–281.
Schofield WN . Predicting basal metabolic rate, new standards and review of previous work. Hum Nutr Clin Nutr 1985; 39: 5–41.
NEVO-tabel 2011, RIVM/Voedingscentrum, Den Haag 2011.
Friedewald WT, Levy RI, Fredrickson DS . Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 1972; 18: 499–502.
Dolan E, Stanton A, Thijs L, Hinedi K, Atkins N, McClory S et al. Superiority of ambulatory over clinic blood pressure measurement in predicting mortality: the Dublin outcome study. Hypertension 2005; 46: 156–161.
Pase MP, Grima NA, Sarris J . The effects of dietary and nutrient interventions on arterial stiffness: a systematic review. Am J Clin Nutr 2011; 93: 446–454.
He FJ, Campbell NRC, Macgregor GA . Reducing salt intake to prevent hypertension and cardiovascular disease. Rev Panam Salud Publica 2012; 32: 293–300.
Graudal NA, Hubeck-Graudal T, Jurgens G . Effects of low sodium diet versus high sodium diet on blood pressure, renin, aldosterone, catecholamines, cholesterol, and triglyceride. Cochrane Database Syst Rev 2011, CD004022.
He FJ, MacGregor GA . A comprehensive review on salt and health and current experience of worldwide salt reduction programmes. J Hum Hypertens 2009; 23: 363–384.
Acknowledgements
The research is funded by TI Food and Nutrition, a public–private partnership on pre-competitive research in food and nutrition. The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. We gratefully acknowledge the assistance of Harrie Robins, Sarah Mount and Danielle Schoenaker in the conduct of the study. We thank the dieticians for dietary calculations and catering, Karin Borgonjen-van den Berg for randomisation and blinding procedures, Anita Bruggink-Hoopman and Diana Emmen-Benink for blood sampling, Marlies Diepeveen-de Bruin for analysing duplicate diets and Hendriek Boshuizen for statistical support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Journal of Human Hypertension website
Supplementary information
Rights and permissions
About this article
Cite this article
Gijsbers, L., Dower, J., Mensink, M. et al. Effects of sodium and potassium supplementation on blood pressure and arterial stiffness: a fully controlled dietary intervention study. J Hum Hypertens 29, 592–598 (2015). https://doi.org/10.1038/jhh.2015.3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jhh.2015.3
This article is cited by
-
Association of urinary sodium and potassium excretion with systolic blood pressure in the Dietary Approaches to Stop Hypertension Sodium Trial
Journal of Human Hypertension (2021)
-
High sodium intake and sodium to potassium ratio may be linked to subsequent increase in vascular damage in adults aged 40 years and older: the Korean multi-rural communities cohort (MRCohort)
European Journal of Nutrition (2019)
-
Relationships between urinary electrolytes excretion and central hemodynamics, and arterial stiffness in hypertensive patients
Hypertension Research (2017)