Abstract
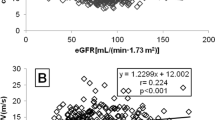
Recent studies suggest a close relationship between renal dysfunction and new onset diabetes (NOD). The aim of the study was to investigate the association between subclinical functional and structural renal abnormalities and NOD in primary hypertension (PH). This observational prospective study (9.1±2.2 years follow-up) includes 231 consecutive untreated non-diabetic patients with PH and without overt nephropathy. The primary end point was NOD. Albuminuria (albumin to creatinine ratio, ACR), glomerular filtration rate (eGFR), and renal structure and hemodynamics (ultrasound scan and Doppler) were evaluated at baseline. During 2106 person-years of follow-up, 10 patients developed diabetes (incidence rate 4.7/1000 person-years). Patients with NOD showed a higher body mass index, serum uric acid, serum creatinine and ACR, and lower eGFR and renal volume (RV) to resistive index (RI) ratio (RV/RI) at baseline, as compared with the 221 controls that did not develop diabetes. When all renal variables were taken into consideration, RV/RI was the only variable significantly related to diabetes (hazard ratio 1.04, P=0.0342). Patients in the lowest tertile of RV/RI were more likely to develop diabetes (10.4 vs 2.6 vs 0%, P=0.0044). For each s.d. decrease of RV/RI, the risk of NOD increased by 68% (P=0.0012). Subclinical functional and structural renal abnormalities are independent predictors of diabetes in PH.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Rahman M, Pressel S, Davis BR, Nwachuku C, Wright Jr JT, Whelton PK et al. Cardiovascular outcomes in high-risk hypertensive patients stratified by baseline glomerular filtration rate. Ann Intern Med 2006; 144: 172–180.
Mykkänen L, Haffner SM, Kuusisto J, Pyorälä K, Laakso M . Microalbuminuria precedes the development of NIDDM. Diabetes 1994; 43: 552–557.
Brantsma AH, Bakker SJ, Hillege HL, de Zeeuw D, de Jong PE, Gansevoort RT . Urinary albumin excretion and its relation with C-reactive protein and the metabolic syndrome in the prediction of type 2 diabetes. Diabetes Care 2005; 28: 2525–2530.
Halimi JM, Bonnet F, Lange C, Balkau B, Tichet J, Marre M . Urinary albumin excretion is a risk factor for diabetes mellitus in men, independently of initial metabolic profile and development of insulin resistance. The DESIR Study. J Hypertens 2008; 26: 2198–2206.
Wang Z, Hoy WE . Albuminuria as a marker of the risk of developing type 2 diabetes in non-diabetic Aboriginal Australians. Int J Epidemiol 2006; 35: 1331–1335.
Chen J, Muntner P, Hamm LL, Jones DW, Batuman V, Fonseca V et al. The metabolic syndrome and chronic kidney disease in US adults. Ann Intern Med 2004; 140: 167–174.
Lorenzo C, Nath SD, Hanley AJ, Abboud HE, Gelfond JA, Haffner SM . Risk of type 2 diabetes among individuals with high and low glomerular filtration rates. Diabetologia 2009; 52: 1290–1297.
Lorenzo C, Nath SD, Hanley AJ, Abboud HE, Haffner SM . Relation of low glomerular filtration rate to metabolic disorders in individuals without diabetes and with normoalbuminuria. Clin J Am Soc Nephrol 2008; 3: 783–779.
Kodama S, Saito K, Yachi Y, Asumi M, Sugawara A, Totsuka K et al. Association between serum uric acid and development of type 2 diabetes. Diabetes Care 2009; 32: 1737–1742.
Viazzi F, Leoncini G, Vercelli M, Deferrari G, Pontremoli R . Serum uric acid levels predict new onset type 2 diabetes in hospitalized patients with primary hypertension: the MAGIC study. Diabetes Care 2011; 34: 126–128.
National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 2002; 106: 3143–3421.
Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro 3rd AF, Feldman HI et al. A new equation to estimate glomerular filtration rate. Ann Intern Med 2009; 150: 604–612.
Mittelstaedt CA . Gen Ultrasound. Churchill: London, 1992, pp 883–1042.
Ikee R, Kobayashi S, Hemmi N, Imakiire T, Kikuchi Y, Moriya H et al. Correlation between the resistive index by Doppler ultrasound and kidney function and histology. Am J Kidney Dis 2005; 46: 603–609.
Pontremoli R, Viazzi F, Martinoli C, Ravera M, Nicolella C, Berruti V et al. Increased renal resistive index in patients with essential hypertension: a marker of target organ damage. Nephrol Dial Transplant 1999; 14: 360–365.
Ratto E, Leoncini G, Viazzi F, Vaccaro V, Falqui V, Parodi A et al. Ambulatory arterial stiffness index and renal abnormalities in primary hypertension. J Hypertens 2006; 24: 2033–2038.
Berni A, Boddi M, Fattori EB, Cecioni I, Berardino S, Montuschi F et al. Serum uric acid levels and renal damage in hyperuricemic hypertensive patients treated with renin-angiotensin system blockers. Am J Hypertens 2010; 23: 675–680.
Viazzi F, Leoncini G, Ratto E, Falqui V, Parodi A, Conti N et al. Mild hyperuricemia and subclinical renal damage in untreated primary hypertension. Am J Hypertens 2007; 20: 1276–1282.
Yasuno S, Ueshima K, Oba K, Fujimoto A, Hirata M, Ogihara T et al. Is pulse pressure a predictor of new-onset diabetes in high-risk hypertensive patients?: a subanalysis of the Candesartan Antihypertensive Survival Evaluation in Japan (CASE-J) trial. Diabetes Care 2010; 33: 1122–1127.
Chen JY, Chou CH, Lee YL, Tsai WC, Lin CC, Huang YY et al. Association of central aortic pressures indexes with development of diabetes mellitus in essential hypertension. Am J Hypertens 2010; 23: 1069–1073.
O'Rourke MF, Hashimoto J . Mechanical factors in arterial aging: a clinical perspective. J Am Coll Cardiol 2007; 50: 1–13 Review.
Afsar B, Elsurer R, Sezer S, Ozdemir FN . Insulin resistance is associated with increased renal resistive index independent of other factors in newly diagnosed type 2 diabetes mellitus and hypertensive patients. Metabolism 2010; 59: 279–284.
Carstensen B, Kristensen JK, Ottosen P, Borch-Johnsen K . The Danish National Diabetes Register: trends in incidence, prevalence and mortality. Diabetologia 2008; 51: 2187–2196.
Jansson SP, Andersson DK, Svärdsudd K . Prevalence and incidence rate of diabetes mellitus in a Swedish community during 30 years of follow-up. Diabetologia 2007; 50: 703–710.
Acknowledgements
This work was supported by grants from the Italian Ministero della Salute (Bando Giovane Ricercatore 2008, CUP G35J11000130001) and from the University of Genoa (Progetti di ricerca di ateneo 2010).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Viazzi, F., Leoncini, G., Derchi, L. et al. Subclinical functional and structural renal abnormalities predict new onset type 2 diabetes in patients with primary hypertension. J Hum Hypertens 27, 95–99 (2013). https://doi.org/10.1038/jhh.2012.5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jhh.2012.5
Keywords
This article is cited by
-
The internist and the renal resistive index: truths and doubts
Internal and Emergency Medicine (2015)