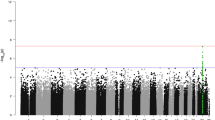
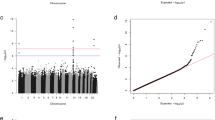
Abstract
Familial hyperaldosteronism type II (FH-II) is an inherited form of hyperaldosteronism associated with hypertension in most patients. The mutations that cause FH-II are unknown, but linkage analysis has mapped them to chromosome 7p22. As FH-II is clinically indistinguishable from sporadic primary aldosteronism, a common and treatable condition, unravelling the cause of FH-II has important implications for these sporadic cases. To investigate whether FH-II is caused by large deletions or insertions, we examined the virtual karyotype of four pairs of affected and unaffected individuals using high-density bead chips. We also sequenced the coding regions of five 7p22 candidate genes that were prioritized because of their putative role in cell growth. We found no evidence of single-nucleotide polymorphism (SNP) copy number variation between pairs, and from the widest gap on the chip, chromosome 7p22 deletions or insertions exceeding ∼50 kb in these pedigrees can be excluded. We found 15 SNPs (two of which were novel), but none of them were non-synonymous and segregated with the disease in the FH-II pedigrees. We have been able to exclude large genomic deletions or insertions at 7p22 and refine the candidate gene list for this locus, but the mutations causing FH-II remain elusive.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Accession codes
References
Fogari R, Preti P, Zoppi A, Rinaldi A, Fogari E, Mugellini A . Prevalence of primary aldosteronism among unselected hypertensive patients: a prospective study based on the use of an aldosterone/renin ratio above 25 as a screening test. Hypertens Res 2007; 30 (2): 111–117.
Torpy DJ, Gordon RD, Lin JP, Huggard PR, Taymans SE, Stowasser M et al. Familial hyperaldosteronism type II: description of a large kindred and exclusion of the aldosterone synthase (CYP11B2) gene. J Clin Endocrinol Metab 1998; 83 (9): 3214–3218.
Gordon RD, Stowasser M, Tunny TJ, Klemm SA, Finn WL, Krek AL . Clinical and pathological diversity of primary aldosteronism, including a new familial variety. Clin Exp Pharmacol Physiol 1991; 18 (5): 283–286.
Lafferty AR, Torpy DJ, Stowasser M, Taymans SE, Lin JP, Huggard P et al. A novel genetic locus for low renin hypertension: familial hyperaldosteronism type II maps to chromosome 7 (7p22). J Med Genet 2000; 37 (11): 831–835.
So A, Duffy DL, Gordon RD, Jeske YW, Lin-Su K, New MI et al. Familial hyperaldosteronism type II is linked to the chromosome 7p22 region but also shows predicted heterogeneity. J Hypertens 2005; 23 (8): 1477–1484.
Elphinstone MS, Gordon RD, So A, Jeske YW, Stratakis CA, Stowasser M . Genomic structure of the human gene for protein kinase A regulatory subunit R1-beta (PRKAR1B) on 7p22: no evidence for mutations in familial hyperaldosteronism type II in a large affected kindred. Clin Endocrinol (Oxf) 2004; 61 (6): 716–723.
Jeske YW, So A, Kelemen L, Sukor N, Willys C, Bulmer B et al. Examination of chromosome 7p22 candidate genes RBaK, PMS2 and GNA12 in familial hyperaldosteronism type II. Clin Exp Pharmacol Physiol 2008; 35 (4): 380–385.
Newton-Cheh C, Guo CY, Gona P, Larson MG, Benjamin EJ, Wang TJ et al. Clinical and genetic correlates of aldosterone-to-renin ratio and relations to blood pressure in a community sample. Hypertension 2007; 49 (4): 846–856.
Huang JT, Lee V . Identification and characterization of a novel human FOXK1 gene in silico. Int J Oncol 2004; 25 (3): 751–757.
Minn AJ, Gupta GP, Siegel PM, Bos PD, Shu W, Giri DD et al. Genes that mediate breast cancer metastasis to lung. Nature 2005; 436 (7050): 518–524.
Fearns C, Pan Q, Mathison JC, Chuang TH . Triad3A regulates ubiquitination and proteasomal degradation of RIP1 following disruption of Hsp90 binding. J Biol Chem 2006; 281 (45): 34592–34600.
Lu L, Han AP, Chen JJ . Translation initiation control by heme-regulated eukaryotic initiation factor 2alpha kinase in erythroid cells under cytoplasmic stresses. Mol Cell Biol 2001; 21 (23): 7971–7980.
Michaelson D, Abidi W, Guardavaccaro D, Zhou M, Ahearn I, Pagano M et al. Rac1 accumulates in the nucleus during the G2 phase of the cell cycle and promotes cell division. J Cell Biol 2008; 181 (3): 485–496.
Rozen S, Skaletsky H . Primer3 on the WWW for general users and for biologist programmers. Methods Mol Biol 2000; 132: 365–386.
Lupski JR, Reid JG, Gonzaga-Jauregui C, Rio Deiros D, Chen DC, Nazareth L et al. Whole-genome sequencing in a patient with Charcot-Marie-Tooth neuropathy. N Engl J Med 2010; 362 (13): 1181–1191.
Ng SB, Buckingham KJ, Lee C, Bigham AW, Tabor HK, Dent KM et al. Exome sequencing identifies the cause of a mendelian disorder. Nat Genet 2010; 42 (1): 30–35.
Dreyer SD, Zhou G, Baldini A, Winterpacht A, Zabel B, Cole W et al. Mutations in LMX1B cause abnormal skeletal patterning and renal dysplasia in nail patella syndrome. Nat Genet 1998; 19 (1): 47–50.
Hall TA . BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 1999; 41: 95–98.
International HapMap Consortium. The International HapMap Project. Nature 2003; 426 (6968): 789–796.
Wilson FH, Disse-Nicodeme S, Choate KA, Ishikawa K, Nelson-Williams C, Desitter I et al. Human hypertension caused by mutations in WNK kinases. Science 2001; 293 (5532): 1107–1112.
Stenson PD, Mort M, Ball EV, Howells K, Phillips AD, Thomas NS et al. The human gene mutation database: 2008 update. Genome Med 2009; 1 (1): 13.1–13.6.
Hoischen A, van Bon BW, Gilissen C, Arts P, van Lier B, Steehouwer M et al. De novo mutations of SETBP1 cause Schinzel-Giedion syndrome. Nat Genet 2010; 42 (6): 483–485.
Nikopoulos K, Gilissen C, Hoischen A, van Nouhuys CE, Boonstra FN, Blokland EA et al. Next-generation sequencing of a 40 Mb linkage interval reveals TSPAN12 mutations in patients with familial exudative vitreoretinopathy. Am J Hum Genet 2010; 86 (2): 240–247.
Acknowledgements
This work was funded by the British Heart Foundation (PG 08/081/25727). We are grateful to the Cambridge Centre of Microarray Resources for performing the microarray work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Journal of Human Hypertension website
Supplementary information
Rights and permissions
About this article
Cite this article
Carss, K., Stowasser, M., Gordon, R. et al. Further study of chromosome 7p22 to identify the molecular basis of familial hyperaldosteronism type II. J Hum Hypertens 25, 560–564 (2011). https://doi.org/10.1038/jhh.2010.93
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jhh.2010.93
Keywords
This article is cited by
-
Pathogenesis of Familial Hyperaldosteronism Type II: New Concepts Involving Anion Channels
Current Hypertension Reports (2019)
-
An update on adrenal endocrinology: significant discoveries in the last 10 years and where the field is heading in the next decade
Hormones (2018)
-
Genetic screening in arterial hypertension
Nature Reviews Endocrinology (2017)
-
ARMC5 is not implicated in familial hyperaldosteronism type II (FH-II)
Journal of Human Hypertension (2017)
-
A case of monozygotic twins with primary aldosteronism: a case report
Journal of Human Hypertension (2017)