Abstract
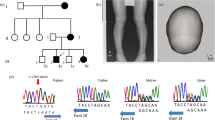
A nonsense mutation at amino acid residue 184 in the human peptidase D (PEPD) gene caused the production of a truncated polypeptide. Characterizing molecular defects in patients provides clues to elucidate the relationship between the phenotype and the genotype.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Additional information
Received: November 8, 1999 / Accepted: November 24, 1999
Rights and permissions
About this article
Cite this article
Kikuchi, S., Tanoue, A., Endo, F. et al. A novel nonsense mutation of the PEPD gene in a Japanese patient with prolidase deficiency. J Hum Genet 45, 102–104 (2000). https://doi.org/10.1007/s100380050023
Published:
Issue Date:
DOI: https://doi.org/10.1007/s100380050023
This article is cited by
-
Human prolidase and prolidase deficiency: an overview on the characterization of the enzyme involved in proline recycling and on the effects of its mutations
Amino Acids (2008)
-
Purification and characterization of recombinant human liver prolidase expressed in Saccharomyces cerevisiae
Archives of Toxicology (2005)
-
Characterization of a new PEPD allele causing prolidase deficiency in two unrelated patients: natural-occurrent mutations as a tool to investigate structure–function relationship
Journal of Human Genetics (2004)