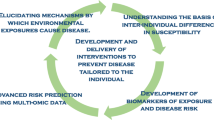
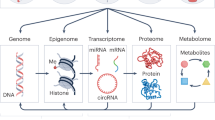
Abstract
During the 1920s, the forerunners of exposure science collaborated with health professionals to investigate the causes of occupational diseases. With the birth of U.S. regulatory agencies in the 1970s, interest in the environmental origins of human diseases waned, and exposure scientists focused instead upon levels of selected contaminants in air and water. In fact, toxic chemicals enter the body not only from exogenous sources (air, water, diet, drugs, and radiation) but also from endogenous processes, including inflammation, lipid peroxidation, oxidative stress, existing diseases, infections, and gut flora. Thus, even though current evidence suggests that non-genetic factors contribute about 90% of the risks of chronic diseases, we have not explored the vast majority of human exposures that might initiate disease processes. The concept of the exposome, representing the totality of exposures received by a person during life, encompasses all sources of toxicants and, therefore, offers scientists an agnostic approach for investigating the environmental causes of chronic diseases. In this context, it is appropriate to regard the “environment” as the body's internal chemical environment and to define “exposures” as levels of biologically active chemicals in this internal environment. To explore the exposome, it makes sense to employ a top-down approach based upon biomonitoring (e.g. blood sampling) rather than a bottom-up approach that samples air, water, food, and so on. Because sources and levels of exposure change over time, exposomes can be constructed by analyzing toxicants in blood specimens obtained during critical stages of life. Initial investigations could use archived blood from prospective cohort studies to measure important classes of toxic chemicals, notably, reactive electrophiles, metals, metabolic products, hormone-like substances, and persistent organic compounds. The exposome offers health scientists an avenue for integrating research that is currently fractured along lines related to particular diseases and risk factors, and can thereby promote discovery of the key exposures responsible for chronic diseases. By embracing the exposome as its operational paradigm, exposure science can play a major role in discovering and mitigating these exposures.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Ames B.N. Dietary carcinogens and anticarcinogens. Oxygen radicals and degenerative diseases. Science 1983: 221 (4617): 1256–1264.
CDC. Fourth National Report on Human Exposure to Environmental Chemicals. National Center for Environmental Health, Centers for Disease Control and Prevention, Atlanta, GA, 2009.
Cherrie J.W. The beginning of the science underpinning occupational hygiene. Ann Occup Hyg 2003: 47 (3): 179–185.
Collins F. Has the revolution arrived? Nature 2010: 464 (7289): 674–675.
Collins F.S., Morgan M., and Patrinos A. The Human Genome Project: lessons from large-scale biology. Science 2003: 300 (5617): 286–290.
Collins F.S. The case for a US prospective cohort study of genes and environment. Nature 2004: 429 (6990): 475–477.
Dalle-Donne I., Rossi R., Colombo R., Giustarini D., and Milzani A. Biomarkers of oxidative damage in human disease. Clin Chem 2006: 52 (4): 601–623.
Dunn W.B., Broadhurst D., Ellis D.I., Brown M., Halsall A., and O’Hagan S., et al. A GC-TOF-MS study of the stability of serum and urine metabolomes during the UK Biobank sample collection and preparation protocols. Int J Epidemiol 2008: 37 (Suppl 1): i23–i30.
Ehrenberg L., Moustacchi E., and Osterman Golkar S. Dosimetry of genotoxic agents and dose-response relationships of their effects. Mut Res 1983: 123: 121–182.
Epel E.S., Blackburn E.H., Lin J., Dhabhar F.S., Adler N.E., and Morrow J.D., et al. Accelerated telomere shortening in response to life stress. Proc Natl Acad Sci USA 2004: 101 (49): 17312–17315.
Gibaldi M., and Perrier D. Pharmacokinetics, Second Edition, Revised and Expanded. Marcel Dekker, New York, 1982.
Greenburg L. Benzol poisoning as an industrial hazard. US Pub Health Repts 1926: 41: 1516–1539.
Greenburg L.W., and Smith G.W. A new instrument for sampling aerial dust. US Bur Mines Rept Invest 1922, 2392.
Hindorff L.A., Junkins H.A., Hall P.N., Mehta J.P., and Manolio T.A. A Catalog of Published Genome-Wide Association Studies. www.genome.gov/gwastudiesAccessed Sep. 10, 2010.
Hood L., Heath J.R., Phelps M.E., and Lin B. Systems biology and new technologies enable predictive and preventative medicine. Science 2004: 306 (5696): 640–643.
Li H., Grigoryan H., Funk W.E., Lu S.S., Rose S., and Williams E.R., et al. Adductomics of Human Serum Albumin by Fixed-Step Selected Reaction Monitoring. Mol Cell Proteomics, in press.
Lichtenstein P., Holm N.V., Verkasalo P.K., Iliadou A., Kaprio J., and Koskenvuo M., et al. Environmental and heritable factors in the causation of cancer--analyses of cohorts of twins from Sweden, Denmark, and Finland. N Engl J Med 2000: 343 (2): 78–85.
Liebler D.C. Protein damage by reactive electrophiles: targets and consequences. Chem Res Toxicol 2008: 21 (1): 117–128.
Manolio T.A., Collins F.S., Cox N.J., Goldstein D.B., Hindorff L.A., and Hunter D.J., et al. Finding the missing heritability of complex diseases. Nature 2009: 461 (7265): 747–753.
Mounicou S., Szpunar J., and Lobinski R. Metallomics: the concept and methodology. Chem Soc Rev 2009: 38 (4): 1119–1138.
Ollier W., Sprosen T., and Peakman T. UK Biobank: from concept to reality. Pharmacogenomics 2005: 6 (6): 639–646.
Patel C.J., Bhattacharya J., and Butte A.J. An Environment-Wide Association study (EWAS) on type 2 diabetes mellitus. PLoS One 2010: 5 (5): e10746.
Rappaport S.M. The rules of the game: an analysis of OSHA's enforcement strategy. Am J Ind Med 1984: 6 (4): 291–303.
Rappaport S.M. Assessing workplace exposures: turning to the past for guidance. Occup Environ Med 2009: 66 (7): 429–430.
Rappaport S.M., and Smith M.T. Environment and disease risks. Science 2010: 330: 460–461.
Roach S.A. A method of relating the incidence of pneumoconiosis to airborne dust exposure. Brit J Ind Med 1953: 10: 220–226.
Roach S.A. A more rational basis for air-sampling programs. Am Ind Hyg Assoc J 1966: 27: 1–12.
Rodgers A., Ezzati M., Vander Hoorn S., Lopez A.D., Lin R.B., and Murray C.J. Distribution of major health risks: findings from the Global Burden of Disease study. PLoS Med 2004: 1 (1): e27.
Rubino F.M., Pitton M., Di Fabio D., and Colombi A. Toward an “omic” physiopathology of reactive chemicals: Thirty years of mass spectrometric study of the protein adducts with endogenous and xenobiotic compounds. Mass Spectrom Rev 2009: 28 (5): 725–784.
Saracci R., and Vineis P. Disease proportions attributable to environment. Environ Health 2007: 6: 38.
Sherwood R.J., and Greenhalgh D.M.S. A personal air sampler. Ann Occup Hyg 1960: 2: 127–132.
Smyth H.F., and Smyth Jr H.F. Spray painting hazards as determined by the Pennsylvania and the National Safety Council surveys. J Ind Hyg 1928: 10 (6): 163–214.
Stratton M.R., Campbell P.J., and Futreal P.A. The cancer genome. Nature 2009: 458 (7239): 719–724.
Vineis P., Khan A.E., Vlaanderen J., and Vermeulen R. The impact of new research technologies on our understanding of environmental causes of disease: the concept of clinical vulnerability. Environ Health 2009: 8: 54.
Wild C.P. Complementing the genome with an “exposome”: the outstanding challenge of environmental exposure measurement in molecular epidemiology. Cancer Epidemiol Biomarkers Prev 2005: 14 (8): 1847–1850.
Willett W.C. Balancing life-style and genomics research for disease prevention. Science 2002: 296 (5568): 695–698.
Acknowledgements
I am grateful for the assistance of Dana Boyd Barr in compiling the JESEE statistics mentioned in this commentary. I also appreciate helpful discussions with Martyn Smith, Christopher Wild, Thomas McKone, and Tina Bahadori. The author acknowledges support through Grant U54ES016115 from the National Institute for Environmental Health Sciences of the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no conflict of interest.
Rights and permissions
About this article
Cite this article
Rappaport, S. Implications of the exposome for exposure science. J Expo Sci Environ Epidemiol 21, 5–9 (2011). https://doi.org/10.1038/jes.2010.50
Published:
Issue Date:
DOI: https://doi.org/10.1038/jes.2010.50
Keywords
This article is cited by
-
Application of clusterization algorithms for analysis of semivolatile pollutants in Arkhangelsk snow
Analytical and Bioanalytical Chemistry (2023)
-
Advances in the Use of Residual Newborn Dried Blood Spots Within Environmental Epidemiology
Current Epidemiology Reports (2023)
-
Restricted spirometry and cardiometabolic comorbidities: results from the international population based BOLD study
Respiratory Research (2022)
-
A review on the application of the exposome paradigm to unveil the environmental determinants of age-related diseases
Human Genomics (2022)
-
EpiVisR: exploratory data analysis and visualization in epigenome-wide association analyses
BMC Bioinformatics (2022)