Abstract
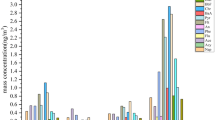
Emissions of complex mixtures of polycyclic aromatic hydrocarbons (PAHs) and other compounds into the environment represent a potential threat to the health of humans. Information regarding the dose and duration of exposure is essential to determine the degree of risk and to identify sensitive receptors within a population. Although measurements of chemical concentrations in air may be used to estimate exposures, internal biomarkers provide more accurate information regarding the dose of exposure and retention of toxic chemicals. This study was conducted in a population in rural China exposed to PAHs from a variety of sources. The study population was located in an area known to have an elevated incidence of birth defects. Parents of children born with a neural tube defect (NTD) were recruited as case participants and parents of children born with no visible birth defect were recruited as controls. The study was designed to test the hypothesis that parents of children born with a NTD would exhibit a biomarker of exposure at higher levels than the parents of a child with no visible birth defect. A total of 35 mothers and 32 fathers were recruited as case participants, and 18 mothers and 19 fathers were recruited as control participants. Venous blood was collected from the study participants by hospital staff as soon as possible following the birth of the child. PAHs were isolated from the whole blood by solvent extraction and DNA was isolated from a separate aliquot of blood for 32P-postlabeling to measure bulky adducts. Single Nucleotide Polymorphisms (SNPs) in phase II enzymes were also monitored in an attempt to identify sensitive receptors. Both total and carcinogenic PAH (cPAH) concentrations were elevated in the parents of case children. Both values were elevated significantly in mothers, whereas only cPAH concentrations were elevated significantly in fathers. Levels of DNA adducts were highly variable and displayed a reverse pattern to that of PAH levels in blood. None of the polymorphisms evaluated were correlated with PAH levels or DNA adducts. For mothers, whose total PAH concentration was above the median concentration, the age-adjusted odds ratio (OR) for having a child with a NTD was 8.7. Although this suggests that PAHs may be a contributing factor to the risk of NTDs, the lack of a correlation with DNA adducts would suggest a possible non-genotoxic mechanism. Alternatively, the PAHs may be a surrogate for a different exposure that is more directly related to the birth defects. The results have shown that blood levels of PAHs may be used to identify populations exposed to elevated concentrations of combustion by-products.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Abdelrahim M., Ariazi E., Kim K., Khan S., Barhoumi R., Burghardt R., Liu S., Hill D., Finnell R., Wlodarczyk B., Jordan V.C., and Safe S. 3-Methylcholanthrene and other aryl hydrocarbon receptor agonists directly activate estrogen receptor alpha. Cancer Res 2006: 66 (4): 2459–2467.
ATSDR. Toxicological Profile for Polycyclic Aromatic Hydrocarbons. US Department of Health and Human Services, Public Health Service, Atlanta, GA, 1995.
ATSDR. Toxicological Profile for Arsenic. US Department of Health and Human Services, Public Health Service, Atlanta, GA, 2007.
Bailey L.R., Roodi N., Verrier C.S., Yee C.J., Dupont W.D., and Parl F.F. Breast cancer and CYPIA1, GSTM1, and GSTT1 polymorphisms: evidence of a lack of association in Caucasians and African Americans. Cancer Res 1998: 58 (1): 65–70.
Bennett W.P., Alavanja M.C., Blomeke B., Vahakangas K.H., Castren K., Welsh J.A., Bowman E.D., Khan M.A., Flieder D.B., and Harris C.C. Environmental tobacco smoke, genetic susceptibility, and risk of lung cancer in never-smoking women. J Natl Cancer Inst 1999: 91 (23): 2009–2014.
Bertrand J.P., Chau N., Patris A., Mur J.M., Pham Q.T., Moulin J.J., Morviller P., Auburtin G., Figueredo A., and Martin J. Mortality due to respiratory cancers in the coke oven plants of the Lorraine coalmining industry (Houilleres du Bassin de Lorraine). Br J Ind Med 1987: 44 (8): 559–565.
Binkova B., Giguere Y., Rossner Jr P., Dostal M., and Sram R.J. The effect of dibenzo[a1]pyrene and benzo[a]pyrene on human diploid lung fibroblasts: the induction of DNA adducts, expression of p53 and p21(WAF1) proteins and cell cycle distribution. Mutat Res 2000: 471 (1–2): 57–70.
Binkova B., Lewtas J., Miskova I., Lenicek J., and Sram R. DNA adducts and personal air monitoring of carcinogenic polycyclic aromatic hydrocarbons in an environmentally exposed population. Carcinogenesis 1995: 16 (5): 1037–1046.
Binkova B., Topinka J., Mrackova G., Gajdosova D., Vidova P., Stavkova Z., Peterka V., Pilcik T., Rimar V., Dobias L., Farmer P.B., and Sram R.J. Coke oven workers study: the effect of exposure and GSTM1 and NAT2 genotypes on DNA adduct levels in white blood cells and lymphocytes as determined by 32P-postlabelling. Mutat Res 1998: 416 (1–2): 67–84.
Carpenter D.O., Arcaro K., and Spink D.C. Understanding the human health effects of chemical mixtures. Environ Health Perspect 2002: 110 (Suppl 1): S25–S42.
Chen B., Hu Y., Jin T., Zheng L., Wang Q., Shen Y., and Zhou Y. Higher urinary 1-hydroxypyrene concentration is associated with cooking practice in a Chinese population. Toxicol Lett 2007: 171 (3): 119–125.
Cizmas L., Barhoumi R., Burghardt R.C., Reeves W.R., He L., McDonald T.J., and Donnelly K.C. A comparison of two methods for fractionating complex mixtures in preparation for toxicity analysis. J Toxicol Environ Health A 2003: 66 (14): 1351–1370.
Dai L., Zhu J., Zhou G., Wang Y., Wu Y., Miao L., and Liang J. Dynamic monitoring of neural tube defects in China during 1996–2000. Zhonghua Yu Fang Yi Xue Za Zhi 2002: 36 (6): 402–405.
Deakin M., Elder J., Hendrickse C., Peckham D., Baldwin D., Pantin C., Wild N., Leopard P., Bell D.A., Jones P., Duncan H., Brannigan K., Alldersea J., Fryer A.A., and Strange R.C. Glutathione S-transferase GSTT1 genotypes and susceptibility to cancer: studies of interactions with GSTM1 in lung, oral, gastric and colorectal cancers. Carcinogenesis 1996: 17 (4): 881–884.
Dejmek J., Solanskỳ I., Benes I., Lenícek J., and Srám R.J. The impact of polycyclic aromatic hydrocarbons and fine particles on pregnancy outcome. Environ Health Perspect 2000: 108 (12): 1159–1164.
Dodds L., and Seviour R. Congenital anomalies and other birth outcomes among infants born to women living near a hazardous waste site in Sydney, Nova Scotia. Can J Public Health 2001: 92 (5): 331–334.
Georgiadis P., Demopoulos N.A., Topinka J., Stephanou G., Stoikidou M., Bekyrou M., Katsouyianni K., Sram R., Autrup H., and Kyrtopoulos S.A. Impact of phase I or phase II enzyme polymorphisms on lymphocyte DNA adducts in subjects exposed to urban air pollution and environmental tobacco smoke. Toxicol Lett 2004: 149 (1–3): 269–280.
Gu X., Lin L., Zheng X., Zhang T., Song X., Wang J., Li X., Li P., Chen G., Wu J., Wu L., and Liu J. High prevalence of NTDs in Shanxi province: a combined epidemiological approach. Birth Defects Res A Clin Mol Teratol 2007: 79 (10): 702–707.
Gupta R.C. Nonrandom binding of the carcinogen N-hydroxy-2-acetylaminofluorene to repetitive sequences of rat liver DNA in vivo. Proc Natl Acad Sci USA 1984: 81 (22): 6943–6947.
Hand P.A., Inskip A., Gilford J., Alldersea J., Elexpuru-Camiruaga J., Hayes J.D., Jones P.W., Strange R.C., and Fryer A.A. Allelism at the glutathione S-transferase GSTM3 locus: interactions with GSTM1 and GSTT1 as risk factors for astrocytoma. Carcinogenesis 1996: 17 (9): 1919–1922.
Hemminki K., Grzybowska E., Chorazy M., Twardowska-Saucha K., Sroczynski J.W., Putman K.L., Randerath K., Phillips D.H., and Hewer A. Aromatic DNA adducts in white blood cells of coke workers. Int Arch Occup Environ Health 1990a: 62 (6): 467–470.
Hemminki K., Grzybowska E., Chorazy M., Twardowska-Saucha K., Sroczynski J.W., Putman K.L., Randerath K., Phillips D.H., Hewer A., Santella R.M., Young T.L., and Perera F.P. DNA adducts in human environmentally exposed to aromatic compounds in an industrial area of Poland. Carcinogenesis 1990b: 11 (7): 1229–1231.
Hou S.M., Lambert B., and Hemminki K. Relationship between hprt mutant frequency, aromatic DNA adducts and genotypes for GSTM1 and NAT2 in bus maintenance workers. Carcinogenesis 1995: 16 (8): 1913–1917.
IARC. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. IARC, Lyon, France, 2004.
Kelsey K.T., Wrensch M., Zuo Z.F., Miike R., and Wiencke J.K. A population-based case-control study of the CYP2D6 and GSTT1 polymorphisms and malignant brain tumors. Pharmacogenetics 1997: 7 (6): 463–468.
Krewski D., and Thomas R.D. Carcinogenic mixtures. Risk Anal 1992: 12 (1): 105–113.
Lan Q., He X., Costa D.J., Tian L., Rothman N., Hu G., and Mumford J.L. Indoor coal combustion emissions GSTM1 and GSTT1 genotypes, and lung cancer risk: a case-control study in Xuan Wei, China. Cancer Epidemiol Biomarkers Prev 2000: 9 (6): 605–608.
Lee D.J., Fleming L.E., Leblanc W.G., Arheart K.L., Chung-Bridges K., Christ S.L., Caban A.J., and Pitman T. Occupation and lung cancer mortality in a nationally representative U.S. Cohort: The National Health Interview Survey (NHIS). J Occup Environ Med 2006: 48 (8): 823–832.
Li S., Moore C.A., Li Z., Berry R.J., Gindler J., Hong S.X., Liu Y., Mulinare J., Wong L.Y., Gu H.Q., and Erickson J.D. A population-based birth defects surveillance system in the People′s Republic of China. Paediatr Perinat Epidemiol 2003: 17 (3): 287–293.
Li Z., Ren A., Zhang L., Ye R., Li S., Zheng J., Hong S., Wang T., and Li Z. Extremely high prevalence of neural tube defects in a 4-county area in Shanxi Province, China. Birth Defects Res A Clin Mol Teratol 2006: 76 (4): 237–240.
Lissowska J., Bardin-Mikolajczak A., Fletcher T., Zaridze D., Szeszenia-Dabrowska N., Rudnai P., Fabianova E., Cassidy A., Mates D., Holcatova I., Vitova V., Janout V., Mannetje A., Brennan P., and Boffetta P. Lung cancer and indoor pollution from heating and cooking with solid fuels: the IARC international multicentre case-control study in Eastern/Central Europe and the United Kingdom. Am J Epidemiol 2005: 162 (4): 326–333.
Liu S., Abdelrahim M., Khan S., Ariazi E., Jordan V.C., and Safe S. Aryl hydrocarbon receptor agonists directly activate estrogen receptor alpha in MCF-7 breast cancer cells. Biol Chem 2006: 387 (9): 1209–1213.
Mathews T.J., Honein M.A., and Erickson J.D. Spina bifida and anencephaly prevalence—United States 1991–2001. MMWR Recomm Rep 2002: 51 (RR-13): 9–11.
Miller K.P., Borgeest C., Greenfeld C., Tomic D., and Flaws J.A. In utero effects of chemicals on reproductive tissues in females. Toxicol Appl Pharmacol 2004: 198 (2): 111–113.
Misaki K., Suzuki M., Nakamura M., Handa H., Iida M., Kato T., Matsui S., and Matsuda T. Aryl hydrocarbon receptor and estrogen receptor ligand activity of organic extracts from road dust and diesel exhaust particulates. Arch Environ Contam Toxicol 2008: 55 (2): 199–209.
Mooney L.A., Santella R.M., Covey L., Jeffrey A.M., Bigbee W., Randall M.C., Cooper T.B., Ottman R., Tsai W.Y., and Wazneh L., et al. Decline of DNA damage and other biomarkers in peripheral blood following smoking cessation. Cancer Epidemiol Biomarkers Prev 1995: 4 (6): 627–634.
Naufal Z., Zhou G.D., McDonald T., Li Z., Li Z., and Donnelly K.C. Genotoxicity of organic extracts of house dust from Shanxi, China. J Toxicol Environ Health A 2007: 70 (24): 2080–2088.
Nielsen T., Jorgensen H.E., Larsen J.C., and Poulsen M. City air pollution of polycyclic aromatic hydrocarbons and other mutagens: occurrence, sources and health effects. Sci Total Environ 1996: 189–190: 41–49.
Neal M.S., Zhu J., and Foster W.G. Quantification of benzo[a]pyrene and other PAHs in the serum and follicular fluid of smokers versus non-smokers. Reprod Toxicol 2008: 25 (1): 100–106.
Peluso M., Srivatanakul P., Munnia A., Jedpiyawongse A., Meunier A., Sangrajrang S., Piro S., Ceppi M., and Boffetta P. DNA adduct formation among workers in a Thai industrial estate and nearby residents. Sci Total Environ 2008: 389 (2–3): 283–288.
Perera F., Hemminki K., Jedrychowski W., Whyatt R., Campbell U., Hsu Y., Santella R., Albertini R., and O′Neill J.P. In utero DNA damage from environmental pollution is associated with somatic gene mutation in newborns. Cancer Epidemiol Biomarkers Prev 2002: 11 (10 Part 1): 1134–1137.
Perera F.P., Tang D., Rauh V., Lester K., Tsai W.Y., Tu Y.H., Weiss L., Hoepner L., King J., Del Priore G., and Lederman S.A. Relationships among polycyclic aromatic hydrocarbon-DNA adducts, proximity to the World Trade Center, and effects on fetal growth. Environ Health Perspect 2005: 113 (8): 1062–1067.
Phillips D.H. Detection of DNA modifications by the 32P-postlabelling assay. Mutat Res 1997: 378 (1–2): 1–12.
Puisieux A., Lim S., Groopman J., and Ozturk M. Selective targeting of p53 gene mutational hotspots in human cancers by etiologically defined carcinogens. Cancer Res 1991: 51 (22): 6185–6189.
Reddy M.V., and Randerath K. Nuclease P1-mediated enhancement of sensitivity of 32P-postlabeling test for structurally diverse DNA adducts. Carcinogenesis 1986: 7 (9): 1543–1551.
Ryberg D., Hewer A., Phillips D.H., and Haugen A. Different susceptibility to smoking-induced DNA damage among male and female lung cancer patients. Cancer Res 1994: 54 (22): 5801–5803.
Santodonato J Review of the estrogenic and antiestrogenic activity of polycyclic aromatic hydrocarbons: relationship to carcinogenicity. Chemosphere 1997: 34 (4): 835–848.
Shaw G.M., Carmichael S.L., Vollset S.E., Yang W., Finnell R.H., Blom H., Midttun O., and Ueland P.M. Mid-pregnancy cotinine and risks of orofacial clefts and neural tube defects. J Pediatr 2009: 154 (1): 17–19.
Singh V.K., Patel D.K., Ram S., Mathur N., Siddiqui M.K., and Behari J.R. Blood levels of polycyclic aromatic hydrocarbons in children of Lucknow, India. Arch Environ Contam Toxicol 2008: 54 (2): 348–354.
Srám R.J., Binková B., Rössner P., Rubes J., Topinka J., and Dejmek J. Adverse reproductive outcomes from exposure to environmental mutagens. Mutat Res 1999: 428 (1–2): 203–215.
Taioli E., Sram R.J., Binkova B., Kalina I., Popov T.A., Garte S., and Farmer P.B. Biomarkers of exposure to carcinogenic PAHs and their relationship with environmental factors. Mutat Res 2007: 620 (1–2): 16–21.
USEPA. Method 8270C, Semivolatile Organic Compounds by Gas Chromatography/mass spectrometry (GC/MS), Revision 3 Test Methods for Evaluating Solid Waste, Physical/Chemical Methods. National Technical Information Service, Washington, DC, 1996 pp. 1–54.
USEPA. Integrated Risk Information System (IRIS) 2006. Available at EPA electronic database www.epa.gov/ncea/iris
van Delft J.H., Steenwinkel M.S., van Asten J.G., de Vogel N., Bruijntjes-Rozier T.C., Schouten T., Cramers P., Maas L., van Herwijnen M.H., van Schooten F., and Hopmans P.M. Biological monitoring the exposure to polycyclic aromatic hydrocarbons of coke oven workers in relation to smoking and genetic polymorphisms for GSTM1 and GSTT1. Ann Occup Hyg 2001: 45 (5): 395–408.
Vyskocil A., Viau C., and Camus M. Risk assessment of lung cancer related to environmental PAH pollution sources. Hum Exp Toxicol 2004: 23 (3): 115–127.
Watts R.R. Manual of Analytical Methods for the Analysis of Pesticides in Humans and Environmental Samples. In: United States Environmental Protection Agency H.E.R.L., Environmental Toxicology Division (Edn.). United States Environmental Protection Agency, Washington, DC, 1980.
Yach D., and Wipfli H. A century of smoke. Ann Trop Med Parasitol 2006: 100 (5–6): 465–479.
Acknowledgements
We thank the staff at all the participating county hospitals in Shanxi, China. We also wish to acknowledge the US Environmental Protection Agency (USEPA) and the National Institute of Environmental Health Sciences Superfund Basic Research Program (NIEHS SBRP) for sponsoring this work. Kristin Marano is recognized for her excellent editorial support.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Naufal, Z., Zhiwen, L., Zhu, L. et al. Biomarkers of exposure to combustion by-products in a human population in Shanxi, China. J Expo Sci Environ Epidemiol 20, 310–319 (2010). https://doi.org/10.1038/jes.2009.19
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jes.2009.19
Keywords
This article is cited by
-
Effect of ambient polycyclic aromatic hydrocarbons and nicotine on the structure of Aβ42 protein
Frontiers of Environmental Science & Engineering (2023)
-
Metabolism of benzo(a)pyrene by aortic subcellular fractions in the setting of abdominal aortic aneurysms
Molecular and Cellular Biochemistry (2016)