Abstract
Drug-resistant bacteria are still emerging, and screening of new skeletal antibiotics is important. During our continuous screening for antimicrobial agents, we discovered a new antimicrobial, named quadoctomycin, from solid culture of Streptomyces sp. MM168-141F8. The substance was purified by solvent extraction, silica gel chromatography and HPLC. Structural elucidation of quadoctomycin was performed by MS and NMR analyses and chemical degradation. Quadoctomycin possesses a 48-membered polyol macrolide skeleton in which an α-D-mannoside is connected to C-22 by an O-glycosidic linkage. The structure of quadoctomycin was found to be related to that of monazomycin A based on the analyses of NMR spectra in the same solvent (pyridine-d5). Quadoctomycin showed potent antibacterial activity against Staphylococcus aureus, including methicillin-resistant S. aureus, and other Gram-positive pathogenic bacteria such as Enterococcus faecalis and E. faecium (including drug-resistant strains), but did not show activity toward Gram-negative bacteria or Candida albicans.
Similar content being viewed by others
Introduction
Antimicrobial resistance has become a global problem that crosses national borders. In 2011, the Director General of the World Health Organization issued an important warning about antimicrobial resistance and a coming global crisis: "no action today means no cure tomorrow”.1 In 2013, the US Centers for Disease Control and Prevention enumerated several pathogens that are considered a serious threat and for which effective antibiotic development is urgently needed. These bacteria include Clostridium difficile, carbapenem-resistant Enterobacteriaceae, extended spectrum β-lactamase-producing Enterobacteriaceae, drug-resistant Neisseria gonorrhoeae, multidrug-resistant Acinetobacter, drug-resistant Campylobacter, vancomycin-resistant Enterococcus, multidrug-resistant (MDR) Pseudomonas aeruginosa, drug-resistant non-typhoidal Salmonella, drug-resistant Salmonella typhi, drug-resistant Shigella, methicillin-resistant Staphylococcus aureus, drug-resistant Streptococcus pneumoniae and multidrug-resistant Mycobacterium tuberculosis.2
Conventionally, to overcome drug-resistant bacteria, development of next-generation semi-synthetic antibiotics has been carried by limited modification of existing antibiotic skeletons. However, as these superbugs spread more quickly than anticipated, it is becoming difficult to overcome resistance by this method. Compounds with new skeletons and mechanisms of action are becoming the scaffolds for the drug discovery process and they bring about changes to current chemotherapy. Natural products have the potential to present novel chemical scaffolds and provide new leads to organic chemists. The search for antibiotics with new structures from natural products is expected to be one approach to developing useful and innovative new drugs for overcoming antimicrobial resistance.3, 4 Indeed, the development and launch of several such antimicrobials effective against multidrug-resistant M. tuberculosis will be a change in the current treatment regimen.5, 6
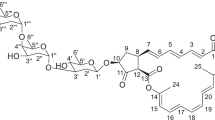
During our screening for antibiotics with a new skeleton and/or new mechanism(s) of action against antimicrobial resistance, we found a novel 48-membered macrolide, named quadoctomycin (Figure 1), in the solid culture material of a Streptomyces strain. In this paper, we describe the taxonomy and fermentation of the producing strain, and the isolation, structure elucidation and biological activities of quadoctomycin.
Results and Discussion
Screening
In continuous screening for new antimicrobial substances against various test organisms, we found antibacterial activity against Gram-positive bacteria including methicillin-resistant S. aureus in the solid culture material of strain MM168-141F8, isolated from a soil sample collected in Shinagawa-ku, Tokyo, Japan. Small-scale culture of the strain was extracted with n-butanol and the dried extract was purified by semi-preparative HPLC. It was suggested that the active fractions gave a protonated molecule with m/z 1436.9961 by HPLC-high resolution ESI/MS (HRESI/MS). This molecule is not described in the Dictionary of Natural Products database. Therefore, we decided to isolate this active substance.
Taxonomy of the antibiotics-producing strain
On oatmeal agar (ISP medium No. 3), strain MM168-141F8 formed well-branched substrate mycelia and straight to flexuous aerial mycelia. The substrate mycelia were light beige, the aerial mycelia were bayberry gray. Mature spore chains consisted of 5–⩾20 spores. The spores were cylindrical to oval with a spiny surface and 0.7 × 1.0–1.0 × 1.2 μm in size (Figure 2). The diaminopimelic acid isomers in whole-cell hydrolysates of strain MM168-141F8 were determined to be the LL-form. A partial 16S ribosomal RNA gene sequence (1425 bp) was determined. The GenBank/EMBL/DDBJ accession number for the 16S rRNA gene sequence of strain MM168-141F8 is LC270914. The strain showed high similarity with those of the genus Streptomyces, such as S. djakartensis NBRC 15409T (1417/1419 bp, 99.86%; T: Type strain) and Streptomyces tuirus NBRC 15617T (1413/1420 bp, 99.51%). These phenotypic and genotypic data suggested that strain MM168-141F8 belongs to the genus Streptomyces. Therefore, the strain was tentatively designated Streptomyces sp. MM168-141F8.
Fermentation and isolation
Quadoctomycin was monitored using antimicrobial activity against S. aureus Smith. The fermentation of quadoctomycin was carried out in solid culture. A total of 7 ml of seed culture was transferred into 14 K-1 flasks each containing 40 g of production medium. The fermentation was carried out at 30 °C for 14 days in the dark. The producing culture (560 g) was extracted with acetone and the extract was concentrated. Water was added and the mixture was extracted with n-butanol. The organic layer was concentrated and the residue was partitioned with n-hexane/methanol. As the antimicrobial activity was in the methanol layer, the methanol solution was concentrated to dryness to obtain a brown solid (1 g). The residue was applied to a silica gel column and eluted with chloroform/methanol/H2O to yield 339 mg of light-brown solid. The solid, containing quadoctomycin, was further purified by HPLC, which was developed with 35% acetonitrile aqueous solution containing 0.01% trifluoroacetic acid to give 55 mg of quadoctomycin as a white powder.
Structure determination
The physicochemical properties of quadoctomycin are summarized in the Materials and Methods section. The optical rotation of the compound was +15° (c 0.5, methanol). It did not show a characteristic UV spectrum. The molecular formula of quadoctomycin was determined to be C76H141NO23 by HRESI/MS. Although the 1H and 13C NMR spectra of quadoctomycin in methanol-d4 initially showed a simple signal, minor signals appeared gradually, giving complicated spectra as shown in Supplementary Figure S2. The reason is not clear, but this finding was also supported by LC-HRESI/MS, which gave peaks of the same m/z with different retention times in an extracted ion chromatogram (see, Supplementary Figure S3). Pyridine-d5 at 35 °C was suitable for NMR analysis. Table 1 summarizes 1H and 13C NMR data for quadoctomycin. The 13C NMR and DEPT-135 spectra revealed that quadoctomycin contains 76 carbon atoms, including 14 methyl-, 19 methylene-, 34 sp3 methine-, six sp2 methine-, two fully substituted sp2- and one carbonyl-carbon(s).
The structure of quadoctomycin was determined by 1H-1H COSY, HSQC-TOCSY and HMBC. 1H-1H COSY showed correlations from isolated olefin, hydroxy methine or methyl signals to adjacent protons from H-7 to H-10-Me, H-11 to H-15, H-16-Me to H-19, H-20-Me to H-24-Me, H-25 to H-32-Me, H-37 to H-38-Me, H-39 to H-49 and from H-50 to H-50-Me. Other correlations from characteristic hydroxy methine signals from H-1’ to H-6’ suggested the presence of a sugar moiety. The HSQC-TOCSY spectra confirmed these connectivities and other correlations from H-2 to H-8-Me, H-33 to H-37 and H-51 to H-56, establishing the partial structures shown in bold in Figure 3. The connectivities of these partial structures were analyzed by HMBC. The cross-peaks observed from H-10-Me to C-9-11, H-16-Me to C-15-17, H-20-Me to C-19-C-21, H-21 to C-19, H-24-Me to C-23-25, H-32-Me to C31-33, H-38-Me to C-37-39, H-50-Me to C-49-51 and the methylene proton of H-2 and the oxymethine of H-47 to C-1, established a 48-membered macrolide skeleton. The sugar moiety was established by the correlation from H-1’ to C-5’ and the moiety was connected to C-22 by the cross-peaks between H-22/C-22 and C-1’/H-1’ in the HMBC spectrum. The planar structure of quadoctomycin determined from these results as shown in Figure 3.
Quadoctomycin has four double bonds and their geometries were determined to be 12E, 16E, 20E and 28E from the large proton–proton coupling constants (3JH12H13=15.4 Hz, 3JH28H29=15.4 Hz), 13C chemical shifts (δC-16Me 13.1 p.p.m. and δC-20Me 13.4 p.p.m.) and NOESY cross-peaks observed between H-15 and H-17, H-17 and H-19 and H-19 and H-21.
The sugar moiety was determined to be α-mannoside by proton–proton (3JH1’H2’=1.5 Hz, 3JH2’H3’=3.3 Hz, 3JH3’H4’=9.2 Hz and 3JH4’H5’=9.2 Hz) and direct carbon–proton coupling constants (1JC1’H1’= 170.0 Hz) and NOESY observed between H-1’ and H-2’, H-2’ and H-3’ and H-3’ and H-5’. The stereochemistry of the sugar moiety was determined by the optical rotations of methanolysis product for quadoctomycin. Quadoctomycin (40 mg) was dissolved in hydrogen chloride-methanol at 65 °C for 3 h, which gave 1.6 mg of methyl-α-D-mannoside, for which NMR and optical rotation data were in good agreement with those of an authentic sample (see Supplementary figure S13 to S16). Figure 1 shows the structure of quadoctomycin elucidated from all the results. The absolute stereochemistry of the aglycon of quadoctomycin is currently being studied.
Quadoctomycin is structurally related to monazomycin A,7 discovered by Professor Hamao Umezawa et al. in 1963.8 NMR data for quadoctomycin were closely related to that for monazomycin A in methanol-d4, but the NMR assignment for monazomycin A has not been published. We compared quadoctomycin and monazomycin A in pyridine-d5 at 35 °C. The assignment of monazomycin A was established by analyzing 1D and 2D NMR spectra (not described in detail, see Supplementary Figure S17-22, Supplementary Table S1). The differences in chemical shifts were around C-10, and from C-50 to C-54 of the side chain, however the remaining 1H and 13C chemical shifts of monazomycin A were almost the same as those for quadoctomycin. Thus, quadoctomycin was determined to be a novel 48-membered ring macrolide having a methyl group at C-10 and a different side chain length from C-47 of monazomycin A.
Actinomycetes are known to produce many macrolides. Among them, the one with the largest ring structure is a 60-membered ring, quinolidomicin, reported by Hayakawa et al.9, 10 As far as the authors know, quadoctomycin has the second largest ring structure (48-membered) equal to that of monazomycin A,7, 8 B.11
Biological activities
Quadoctomycin showed potent antibacterial activity against S. aureus (including methicillin-resistant S. aureus) with minimum inhibitory concentrations (MICs) of 1–2 μg ml−1 (Table 2). Quadoctomycin also had antibacterial activity against other Gram-positive pathogenic bacteria such as Enterococcus faecalis and E. faecium (including drug-resistant strains), Micrococcus luteus, Bacillus subtilis, B. cereus, Mycobacterium smegmatis and Corynebacterium bovis, with minimum inhibitory concentrations of 0.5–2 μg ml−1. Quadoctomycin did not show antimicrobial activity toward Gram-negative bacteria or Candida albicans (Table 2). Antimicrobial activities of quadoctomycin showed equal or two to fourfold more potent than those of monazomycin A as shown in Table 2.
Materials and methods
General
The optical rotations of compounds were obtained using a P-1030 polarimeter (JASCO, Tokyo, Japan). UV spectra were recorded with a U-2800 UV-Vis spectrophotometer (Hitachi High-Technologies, Tokyo, Japan). IR spectra were recorded with a FT/IR-4100 Fourier-transform infrared spectrometer (JASCO). 1H and 13C NMR spectra, including 2D NMR, were measured with an ECZ600R spectrometer (JEOL Resonance, Tokyo, Japan) and an AVANCE III 500 spectrometer (Bruker, Billerica, MA, USA) using solvent signals as an internal reference. Mass spectra were recorded using a LTQ Orbitrap XL mass spectrometer (Thermo Fisher Scientific, San Jose, CA, USA). The novelty of quadoctomycin was searched using the Dictionary of Natural Products on DVD databases (Chapman & Hall/CRC Press, Boca Raton, FL, USA) and SciFinder (Chemical Abstracts Service, Columbus, OH, USA). Standard methyl-α-D-mannoside was purchased from Sigma Aldrich, St Louis, MO, USA. Monazomycin A was obtained from our inhouse library.
Taxonomic studies of the producing strain MM168-141F8
Morphological properties were observed following incubation at 30°C for 21 days on yeast extract-malt extract agar (ISP medium No. 2), oatmeal agar (ISP medium No. 3) and inorganic salt starch agar (ISP medium No. 4). Detailed observation of mycelial morphology was performed using a scanning electron microscope (SU-1510, Hitachi High-Technologies) after strain MM168-141F8 was incubated on ISP medium No. 3 at 30 °C for 8 days. The type of diaminopimelic acid isomers in whole-cell hydrolysates was determined by the method of Staneck and Roberts.12 The total DNA of MM168-141F8 was prepared using a Genomic DNA Extraction Kit Mini (RBC Bioscience Co., New Taipei, Taiwan) according to the manufacturer’s instructions. 16S rRNA (positions 56–1495, Escherichia coli numbering system13) was amplified by PCR and sequenced. A search for the most closely related sequences was performed using the EZ Taxon database (http://www.ezbiocloud.net/).14
Fermentation and purification of quadoctomycin
A slant culture of Streptomyces sp. MM168-141F8 was inoculated into a 500-ml baffled Erlenmeyer flask containing 110 ml of a seed medium consisting of 2.0% galactose, 2.0% dextrin, 1.0% Bacto Soytone (Becton Dickinson and Company, Franklin Lakes, NJ, USA), 0.5% corn steep liquor (Kogo Starch, Chiba, Japan), 0.2% (NH4)2SO4 and 0.2% CaCO3, pH 7.4 before sterilization. The culture was incubated at 27 °C for 3 days on a rotary shaker at 180 rpm. Production was performed using a solid medium (15 g of pressed barley and 25 ml of deionized water). Portions of 7 ml of this seed culture were inoculated into 14 K-1 flasks containing solid medium. The culture was incubated statically at 30 °C for 14 days. The whole fermentation culture (560 g) was extracted with acetone (560 ml, twice) and the acetone extract obtained by filtration. The solvent was concentrated in vacuo; water (300 ml) was added and extracted with an equivalent volume of n-BuOH. The organic layer was evaporated and partitioned by n-hexane/MeOH (10 ml each). The MeOH layer was concentrated to dryness to obtain a brown solid (1 g). The residue was suspended in a small amount of chloroform/MeOH (1:1) and applied to a silica gel column (50 g, Silica gel 60 Art 1.07734; Merck KGaA, Darmstadt, Germany) and washed with chloroform/MeOH/H2O (v/v 3:1:0.15) and eluted with chloroform/MeOH/H2O (v/v 10:5:1) to give 339 mg of light-brown solid. The solid containing the quadoctomycin was further purified by HPLC (600E System, Waters, Milford, MA, USA) using an ODS column (Shiseido, Capcell Pak UG120, 30 mm id × 250 mm; Tokyo, Japan) developed with 35% acetonitrile aqueous solution containing 0.01% trifluoroacetic acid to give 55 mg of quadoctomycin: white powder; [α]D28 +15° (c 0.5, MeOH); IR νmax (KBr) cm−1 3406, 2965, 2934, 1780, 1677, 1458, 1434, 1385, 1092, 1070, 1049, 974; UV λmax (MeOH) end absorption; 1H NMR (pyridine-d5, 600 MHz), 13C NMR (pyridine-d5, 150 MHz) data are given in Table 1; HRESI/MS [M+H]+ m/z 1436.9961 (calcd. for C76H142NO23, 1436.9967).
Methanolysis of quadoctomycin
Quadoctomycin (40 mg) was dissolved in 3 m hydrogen chloride-MeOH and stirred at 65 °C for 3 h. The solution was concentrated in vacuo. The residue was dissolved in a small amount of MeOH and applied to Sephadex LH-20 (GE Healthcare Bio-Sciences, Uppsala, Sweden). The fractions containing methyl glycoside were collected and dried. The residue was subjected to hydrophilic interaction liquid chromatography (XBridge Amide, 3.5 μm, 10 mm id × 150 mm, Waters, 90–50% acetonitrile, three times). The pure fraction containing methyl glycoside was collected and concentrated to dryness to give 1.6 mg of methyl-α-mannoside: colorless solid; [α]D28 +71° (c 0.1, H2O); 1H NMR (D2O, 500 MHz) δ 4.74 (1H, d, 1.7, H-1), 3.91 (1H, dd, 3.4, 1.7, H-2), 3.88 (1H, dd 12.3, 2.0, H-6b), 3.73 (1H, dd, 12.3, 5.7, H-6a), 3.73 (1H, m, H-3), 3.62 (1H, t 9.7, H-4), 3.59 (1H, m, H-5), 3.39 (3H, s, H-1OMe); 13C NMR (D2O, 125 MHz) δ 103.5 (d, C-1), 75.2 (d, C-5), 73.2 (d, C-3), 72.6 (d, C-2), 69.4 (d, C-4), 63.6 (t, C-6), 57.4 (q, C-1OMe); HRESI/MS [M+Na]+ m/z 217.0685 (calcd for C7H14O6Na, 217.0683).
Antimicrobial activity
Minimum inhibitory concentrations were determined by the standard agar dilution method per Clinical Laboratory Standards Institute guidelines.15 Bacteria were incubated on Mueller-Hinton agar (Becton Dickinson) at 37 °C for 18 h, and yeast were incubated for 42 h.
Dedication
This article is dedicated to Professor Dr. Hamao Umezawa in honor of his profound contributions to basic science and the improvement of human health.
References
World Health Organization World Health Day – 7 April 2011 http://www.who.int/world-health-day/2011/en/ (2011).
United States Centers for Disease Control and Prevention. Antibiotic resistance threats in the United States 2013 (2013).
Quan, D., Nagalingam, G., Payne, R. & Triccas, J.A. New tuberculosis drug leads from naturally occurring compounds. Int. J. Infect. Dis. 56, 212–220 (2017).
Sieniawska, E. Targeting mycobacterial enzymes with natural products. Chem. Biol. 22, 1288–1300 (2015).
Kwon, Y.S., Jeong, B.H. & Koh, W.J. Tuberculosis: clinical trials and new drug regimens. Curr. Opin. Pulm. Med. 20, 280–286 (2014).
First-line chemotherapy in the retreatment of bacteriological relapses of pulmonary tuberculosis following a shortcourse regimen. Lancet 307, 162–163 (1976).
Nakayama, H., Furihata, K., Seto, H. & Otake, N. Structure of monazomycin, A new ionophohous antibiotic. Tetrahedron Lett. 22, 5217–5220 (1981).
Akasaki, K., Karasawa, K., Watanabe, M., Yonehara, H. & Umezawa, H. Monazomycin, a new antibiotic produced by a Streptomyces. J. Antibiot. Ser. A 16, 127–131 (1963).
Hayakawa, Y., Shinya, K., Furihata, K. & Seto, H. Structure of a novel 60-membered macrolide, quinolidomicin A1. J. Am. Chem. Soc. 115, 3014–3015 (1993).
Hayakawa, Y., Shinya, K., Furihata, K. & Seto, H. Quinolidomicins A1, A2 and B1, novel 60-membered macrolide antibiotics. II. Structure elucidation. J. Antibiot. 46, 1563–1569 (1993).
Kuo, M.S. et al. Monazomycin B, a new macrolide antibiotic of the monazomycin family. J. Antibiot. 43, 438–440 (1990).
Staneck, J.L. & Roberts, G.D. Simplified approach to identification of aerobic actinomycetes by thin-layer chromatography. Appl. Microbiol. 28, 226–231 (1974).
Brosius, J., Palmer, M.L., Kennedy, P.J. & Noller, H.F. Complete nucleotide sequence of a 16S ribosomal RNA gene from Escherichia coli. Proc. Natl Acad. Sci. USA 75, 4801–4805 (1978).
Kim, O.S. Introducing EzTaxon-e: a prokaryotic 16S rRNA gene sequence database with phylotypes that represent uncultured species. Int. J. Syst. Evol. Microbiol. 62, 716–721 (2012).
Clinical and Laboratory Standards Institute Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically; Approved Standard-Eighth Edition M07-A8 CLSI (2009).
Acknowledgements
We thank Dr K Furihata, University of Tokyo, for providing NMR spectra of monazomycin A. We thank Ms N Kinoshita, Ms R Arisaka and Ms Y Shibuya, Institute of Microbial Chemistry (BIKAKEN), Tokyo for providing technical assistance.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on The Journal of Antibiotics website
Supplementary information
Rights and permissions
About this article
Cite this article
Sawa, R., Kubota, Y., Umekita, M. et al. Quadoctomycin, a 48-membered macrolide antibiotic from Streptomyces sp. MM168-141F8. J Antibiot 71, 91–96 (2018). https://doi.org/10.1038/ja.2017.140
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ja.2017.140
This article is cited by
-
Naphthoquinone-derived polyol macrolides from natural sources
Russian Chemical Bulletin (2019)