Abstract
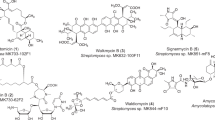
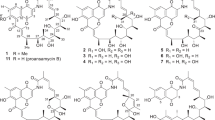
Resistance to currently available antibiotics has become a widely recognized crisis in the medical community. To address this, many companies and researchers are refocusing their attention towards natural products, which have an excellent track record of producing effective antibacterial drugs. The AMRI natural product library was screened for activity against multi-drug resistant Staphylococcus aureus (MDRSA). The active samples were counter screened for cytotoxicity against the human hepatocellular carcinoma HepG2 cell line to determine an in vitro therapeutic index (in vitro TI). Those samples with a high in vitro TI were selected for fractionation and dereplication. This led to the discovery of a new anthracycline structure. This metabolite, named mutactimycin E (1), exhibited moderate activity against several gram positive organisms. Here we report the isolation, structure elucidation and biological activities of this new compound.
Similar content being viewed by others
Article PDF
References
Dictionary of Natural Products, Chapman & Hall/CRC, Version 16:2, December 2007
Jin WZ, Cheng J, Zhang YB, Li HL, Tao PZ . Isolation and structure determination of mutactimycin A, a new anthracycline antibiotic. Kangshengsu 15: 399–406 ( 1990)
Mikami Y, Yazawa K, Ohashi S, Maeda A, Akao M . SO-75R1, a new mutactimycin derivative produced by Nocardia brasiliensis. J Antibiot 45: 995–997 ( 1992)
Speitling M, Nattewan P, Yazawa K, Mikami Y, Gruen-Wollny I, Ritzau M, Laatsch H, Graefe U . Demethyl mutactimycin, new anthracycline antibiotics from Nocardia and Streptomyces strains. J Antibiot 51: 693–698 ( 1998)
Zitouni A, Mathieu F, Coppel Y, Pont F, Sabau N, Lebrihi A . Mutactimycin PR, a New Anthracycline Antibiotic from Saccharothrix sp. SA 103. J Antibiot 57: 373–378 ( 2004)
Zhang Y, Jin W . Isolation and structure determination of minor components of mutactimycins, a group of new anthracycline antibiotics. Kangshengsu 16: 157–164 ( 1991)
Alder JD . Daptomycin, a new drug class for the treatment of Gram-positive infections. Drugs Today 41: 81–90 ( 2005)
Ford CW, Zurenko GE, Barbachyn MR . The discovery of linezolid, the first oxazolidinone antibacterial agent. Cur Drug Targets 1: 19–36 ( 2001)
Sunagawa M, Matsumura H, Inoue T, Fukusawa M, Kato M . A novel carbapenem antibiotic, SM-7338 structure-activity relationships. J Antibiot 43: 519–532 ( 1990)
Data were generated in house and by Micromyx, LLC ( Kalamazoo, MI)
Clinical Laboratory Standards (CLSI/NCCLS) Approved Standard M7–A7 ( 2006)
Mueller H, Kassack MU, Wiese M . Comparison of the usefulness of the MTT, ATP, and calcein assays to predict the potency of cytotoxic agents in various human cancer cell lines. J Biomol Screen 9( 6): 506–515 ( 2004)
Moravec R, Beck M, Hannah R . Promega Cell Notes 2: 13–16 ( 2001)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hopp, D., Rabenstein, J., Rhea, J. et al. Mutactimycin E, a New Anthracycline Antibiotic with Gram-positive Activity. J Antibiot 61, 675–679 (2008). https://doi.org/10.1038/ja.2008.95
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ja.2008.95
Keywords
This article is cited by
-
Dereplication strategies in natural product research: How many tools and methodologies behind the same concept?
Phytochemistry Reviews (2017)