Abstract
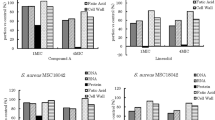
In vitro antimicrobial activity of telavancin, a rapidly bactericidal lipoglycopeptide, was evaluated against 1500 strains of MRSA recently isolated in Japan. Telavancin had potent activity, with MIC values that ranged from 0.12 μg/ml to 0.5 μg/ml and a MIC90 value of 0.5 μg/ml. The MIC90s of vancomycin and linezolid were 1.0μg/ml and 2 μg/ml, respectively. No vancomycin intermediate resistant or vancomycin-resistant MRSAs were detected in this surveillance study.
Similar content being viewed by others
Article PDF
References
Segreti J . Efficacy of current agents used in the treatment of Gram-positive infections and the consequences of resistance. Clin Microbiol Infect 11 ( Suppl. 3): 29–35 ( 2005)
Tenover FC, Biddle JW, Lancaster MV . Increasing resistance to vancomycin and other glycopeptides in Staphylococcus aureus. Emerg Infect Dis 7: 327–332 ( 2001)
Appelbaum PC . The emergence of vancomycin-intermediate and vancomycin-resistant Staphylococcus aureus. Clin Microbiol Infect 12: ( Suppl. 1) 16–23 ( 2006)
Potoski BA, Adams J, Clarke L, Shutt K, Linden PK, Baxter C, Pasculle AW, Capitano B, Peleg AY, Szabo D, Paterson DL . Epidemiological profile of linezolid-resistant coagulase-negative staphylococci. Clin Infect Dis 43: 165–171 ( 2006)
Moise-Broder PA, Sakoulas G, Eliopoulos GM, Schentag JJ, Forrest A, Moellering Jr RC . Accessory gene regulator group II polymorphism in methicillin-resistant Staphylococcus aureus is predictive of failure of vancomycin therapy. Clin Infect Dis 38: 1700–1705 ( 2004)
Sakoulas G, Moise-Broder PA, Schentag J, Forrest A, Moellering Jr RC, Eliopoulos GM . Relationship of MIC and bactericidal activity to efficacy of vancomycin for treatment of methicillin-resistant Staphylococcus aureus bacteremia. J Clin Microbiol 42: 2398–2402 ( 2004)
Stryjewski ME, Chu VH, O'Riordan WD, Warren BL, Dunbar LM, Young DM, Vallée M, Fowler Jr VG, Morganroth J, Barriere SL, Kitt MM, Corcy GR . Telavancin versus standard therapy for treatment of complicated skin and skin structure infections caused by Gram-positive bacteria: FAST 2 study. Antimicrob Agents Chemother 50: 862–867 ( 2006)
Higgins DL, Chang R, Debabov DV, Leung J, Wu T, Krause KM, Sandvik E, Hubbard JM, Kaniga K, Schmidt Jr DE, Gao Q, Cass RT, Karr DE, Benton BM, Humphrey PP . Telavancin, a multifunctional lipoglycopeptide, disrupts both cell wall synthesis and cell membrane integrity in methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother 49: 1127–1134 ( 2006)
Pace JL, Judice JK . Telavancin (Theravance). Curr Opin Investig Drugs 6: 216–225 ( 2005)
Sahm DF, Benton BM, Jones ME, Krause KM, Thornsberry C, Draghi DC . Telavancin demonstrates a low potential for in vitro selection of resistance among key target Gram-positive species. Poster C1-0681, Program and Abstracts of the 46th Interscience Conference on Antimicrobial Agents and Chemotherapy, San Francisco, September 27–30 ( 2006)
Clinical and Laboratory Standards Institute . Performance standards for antimicrobial susceptibility testing; sixteenth informational supplement. Volume 26, number 3. M100-S16, Clinical and Laboratory Standards Institute (formerly NCCLS), Wayne, PA ( 2006)
Clinical and Laboratory Standards Institute . Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; seventh edition. M7-A7, Clinical and Laboratory Standards Institute (formerly NCCLS), Wayne, PA ( 2006)
Draghi DC, Benton BM, Jones ME, Krause KM, Thornsberry C, Sahm DF . Baseline antistaphylococcal profile of telavancin: Results of the 2004∼2005 US surveillance initiative. Poster E-0715, Program and Abstracts of the 46th Interscience Conference on Antimicrobial Agents and Chemotherapy, San Francisco, September 27–30 ( 2006)
Sahm DF, Benton BM, Krause KM, Jones ME, Thornsberry C, Draghi DC . Baseline surveillance of telavancin in vitro activity against staphylococci in Europe and Israel. Poster 193. Abstracts of Papers of 44th Annual Meeting of Infectious Diseases Society of America, Toronto, October 12–15 ( 2006)
Leuthner KD, Cheung CM, Rybak MJ . Comparative activity of the new lipoglycopeptide telavancin in the presence and absence of serum against 50 glycopeptide non-susceptible staphylococci and three vancomycin-resistant Staphylococcus aureus. J Antimicrob Chemother 58: 338–343 ( 2006)
Tenover FC, Weigel LM, Appelbaum PC, McDougal LK, Chaitram J, McAllister S, Clark N, Killgore G, O'Hara CM, Jevitt L, Patel JB, Bozdogan B . Vancomycin-resistant Staphylococcus aureus isolate from a patient in Pennsylvania. Antimicrob Agents Chemother 48: 275–280 ( 2004)
Draghi DC, Blosser RS, Flamm RK, Sahm DF . A baseline assessment of the activity of telavancin against a collection of Gram-positive isolates, including resistant phenotypes. [Poster 1553]. Clin Microbiol Infect 11: ( Suppl. 2) 505 ( 2005)
Farrell DJ, Shackcloth J, Williams L . Activity of telavancin and comparator antibiotics against clinically relevant gram-positive isolates including strains with diverse mechanisms of resistance. Poster E-1746, Program and Abstracts of the 45th Interscience Conference on Antimicrobial Agents and Chemotherapy, Washington, DC, December 16–19 ( 2005)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hatano, K., Matsuzaki, K., Sato, Y. et al. In Vitro Antimicrobial Activity of Telavancin against Methicillin-resistant Staphylococcus aureus Clinical Isolates from Japan (2006). J Antibiot 60, 709–712 (2007). https://doi.org/10.1038/ja.2007.91
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ja.2007.91