Abstract
Molecular fossils of 2-methylhopanoids are prominent biomarkers in modern and ancient sediments that have been used as proxies for cyanobacteria and their main metabolism, oxygenic photosynthesis. However, substantial culture and genomic-based evidence now indicates that organisms other than cyanobacteria can make 2-methylhopanoids. Because few data directly address which organisms produce 2-methylhopanoids in the environment, we used metagenomic and clone library methods to determine the environmental diversity of hpnP, the gene encoding the C-2 hopanoid methylase. Here we show that hpnP copies from alphaproteobacteria and as yet uncultured organisms are found in diverse modern environments, including some modern habitats representative of those preserved in the rock record. In contrast, cyanobacterial hpnP genes are rarer and tend to be localized to specific habitats. To move beyond understanding the taxonomic distribution of environmental 2-methylhopanoid producers, we asked whether hpnP presence might track with particular variables. We found hpnP to be significantly correlated with organisms, metabolisms and environments known to support plant–microbe interactions (P-value<10−6); in addition, we observed diverse hpnP types in closely packed microbial communities from other environments, including stromatolites, hot springs and hypersaline microbial mats. The common features of these niches indicate that 2-methylhopanoids are enriched in sessile microbial communities inhabiting environments low in oxygen and fixed nitrogen with high osmolarity. Our results support the earlier conclusion that 2-methylhopanoids are not reliable biomarkers for cyanobacteria or any other taxonomic group, and raise the new hypothesis that, instead, they are indicators of a specific environmental niche.
Similar content being viewed by others
Introduction
Morphological and molecular fossils left by microorganisms in ancient sedimentary rocks can provide a valuable window into the early history of life on Earth. Yet due to challenges inherent in working with billion-year-old samples, the interpretation of these fossils has often been contentious (Schopf and Packer 1987; Walter et al., 1992; Brasier et al., 2002, 2004). In this context, organic biomarkers have received attention due to their potential to provide more specific information about the composition of ancient microbial communities (Brocks and Pearson, 2005). Hopanes and steranes are among the more prominent classes of these biomarkers (Rohmer, 2010). These molecules can unambiguously be interpreted as the diagenetic remains of hopanoids and steroids, polycyclic triterpenoids found in the membranes of numerous organisms today (Rohmer et al., 1984; Ourisson et al., 1987). However, ambiguity regarding their distribution and function in modern bacteria clouds our ability to interpret their fossils in the rock record.
Hopanoids structurally resemble steroids, but unlike steroids, they are primarily made by bacteria and do not require oxygen for their biosynthesis (Ourisson et al., 1987; Fischer et al., 2005; Rashby et al., 2007). Although hopanoids exhibit structural diversity of their side chains, much of this diversity is lost through diagenesis, resulting mainly in the preservation of their hydrocarbon skeletons, hopanes. Important exceptions to this are methyl groups at C-2 or C-3, which can be preserved. In fact, 2-methylhopanes have a rich history in the fossil record, found at discrete times and locations as far back as 2.7 billion years (Brocks et al., 1999), although this latter finding is under scrutiny (Rasmussen et al., 2008). The varied distribution of 2-methylhopanes in the more ‘recent’ rock record (that is, million-year timescales), showing peaks in abundance correlated with ocean anoxic events, suggests their production may be linked to particular environmental triggers (Knoll et al., 2007).
Until recently, 2-methylhopanes were viewed as biomarkers for cyanobacteria and their main energy-generating metabolism, oxygenic photosynthesis (Summons et al., 1999). However, the finding of conditional 2-methylhopanoid (2-MeBHP) production by the anoxygenic phototroph Rhodopseudomonas palustris called this interpretation into question (Rashby et al., 2007). From genomic data and culture-based work, it is now clear that only a minority of cyanobacteria (that is, 13% of all sequenced cyanobacterial species and 19% of all sequenced cyanobacterial genera) have the gene, hpnP, that encodes the enzyme responsible for methylating hopanoids at C-2 (Supplementary Figure S1); (Talbot et al., 2008; Welander et al., 2010). Moreover, other bacterial species possess hpnP including many from a subclade of alphaproteobacteria and an acidobacterium (Talbot et al., 2007a; Welander et al., 2010).
Despite the known distribution of hpnP among cultured organisms, uncertainty remains about which producers of 2-MeBHPs are environmentally relevant and whether there is a common ecology that correlates with the production of these molecules. Given that 2-MeBHPs are produced by diverse bacteria, we asked two questions: (1) what is the potential for 2-MeBHP production by different organisms in modern environments, and (2) might 2-MeBHP producers inhabit common ecological niche(s)? Here we assess the distribution of hpnP in various environments, and find a statistically significant correlation with modern habitats that support plant–microbe interactions. We discuss the implications of these results for interpreting the ancient 2-methylhopane record.
Materials and methods
Distribution of hpnP in cyanobacterial genomes
The abundance of hpnP genes among cyanobacteria was calculated using finished cyanobacterial genomes on the Joint Genomes Institute’s Integrated Microbial Genomes (IMG) database (Markowitz et al., 2011). Additionally, we analyzed this data set condensed to the genus level to reduce bias (Supplementary Figure S1).
Distribution of hpnP in metagenomes
To retrieve HpnP sequences from public metagenomes, HpnP from R. palustris TIE-1 (NC_011004.1) was used as a query against the NCBI metagenomic proteins database, IMG/M, CAMERA and myMGDB. All hits with an e-value equal to or less than 1 × 10−50 were subjected to phylogenetic analysis (Markowitz et al., 2011; Sun et al., 2011). Approximately 20% of hits retrieved clustered phylogenetically with known HpnP sequences, whereas all others clustered with sequences known not to be hpnP. Only sequences that cluster with known hpnP genes were identified as hpnP (Supplementary Data Set S1). All searches were completed in December 2012. Descriptions of surveyed metagenomes appear in Supplementary Data Set S2.
Sequences were identified as gyrB, psbC and shc if they had an e-value equal to or less than 1 × 10−20. Escherichia coli gyrB (NC_000913.2), Nostoc punctiforme psbC (YP_001866969.1) and R. palustris TIE-1 shc (NC_011004.1) were used as query sequences. This e-value was used because it captured known diversity of the genes without retrieving related sequences with other functions. Sequences of shc were determined to be cyanobacterial or alphaproteobacterial by top BLASTP hit (Table 1).
Clone library and sample preparation
DNA samples from Yellowstone National Park hot springs were collected and prepared as previously reported (Osburn et al., 2011). All other samples were extracted with the UltraClean Soil DNA Isolation Kit (MoBio, Carlsbad, CA, USA). DNA samples were stored at −20 °C. Information on samples can be found in Supplementary Table 1.
Nested PCR primers for hpnP were designed based on the conserved amino-acid motifs (A/G)FMPPQ and (S/T)GII(L/M)G, for the first pair, and (A/V)(L/I)GGPS and GIETP(E/D), for the second. The resulting primers were hpnP_1 F 5′-GSB TTY ATG CCD CCB CAR GG-3′, hpnP_1 R 5′-TCN ARK CCV AKR ATR ATN CC-3′, hpnP_2 F 5′-GYB VTB GGH GGN CCN TCN GT-3′ and hpnP_2 R 5′-TCN GGN GTY TCD ATN CC-3′, respectively (Supplementary Figure S3A). To amplify hpnP, Promega PCR master mix (Promega, Madison, WI, USA) and cycles of 95 °C for 2 min, 95 °C denaturation for 30 s, 50 °C for 30 s and 72 °C for 1 min for 35 cycles, followed by 72 °C for 3 min were used. An aliquot of the first PCR (1 μl) was used as a template for the second. We validated this method by testing the primer sets on diverse 2-MeBHP-producing organisms as well as some that do not make 2-MeBHPs (Supplementary Figure S3C).
PCR products from the second reaction were extracted with the Montage gel extraction kit (Millipore, Billerica, MA, USA) and cloned with the TOPO TA Cloning Kit for sequencing using One Shot Top10 electrocompetent cells (Invitrogen, Carlsbad, CA, USA). Twenty-four to 100 clones per sample were amplified by PCR and restriction digested with AluI (New England Biolabs, Ipswich, MA, USA). Amplicons with unique digestion patterns were sequenced (Retrogen, San Diego, CA, USA). Sequences were trimmed to remove contaminating vector and poor quality regions, and then translated in Geneious 5.6.5. Representative sequences of 95% identity clusters were picked using CD-HIT and used to make phylogenetic trees (Huang et al., 2010). Sequences have been deposited in GenBank under the accession numbers KC603770 thru KC603846.
Rarefaction curves
The number of unique restriction digestion patterns was used as a proxy for hpnP diversity. To generate rarefaction curves, the species observed metric was used in the alpha diversity package of QIIME 1.5.0 (Caporaso et al., 2010). Data sets were rarefied 100 times in 2 step increments (Supplementary Figure S5). To compare richness of hpnP between clone libraries, data sets were rarefied to 25 clones to avoid depth of sampling bias and averaged (Supplementary Figure S5). Guerrero Negro sample 4 was not included in this analysis because of low sampling coverage (Supplementary Table 1).
Alignment and phylogeny construction
All alignments were made using the MAFFT v6.859b l-ins-i algorithm (Katoh and Toh, 2008). Reference HpnP alignments comprise all HpnP sequences retrieved from NCBI as of December 2012 and hpnP from Phormidium luridum UTEX 426. Full-length P. luridum UTEX 426 hpnP was obtained by inverse PCR using the degenerate hpnP PCR primers as a probe. Outgroup sequences were picked from the sister clade of HpnP (Welander et al., 2010). The reference alignment was trimmed in Gblocks 0.91b with relaxed parameters (Talavera and Castresana, 2007). Environmental HpnP sequences were then added to the reference HpnP alignments using the seed option in MAFFT. Phylogenetic trees were made by PhyML v3.0 using the LG model with aLRT supports and modified in iTOL (Guindon et al., 2010; Letunic and Bork, 2007).
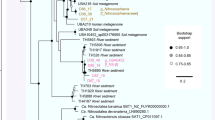
HpnP types (cyanobacterial, alphaproteobacterial or unknown) from metagenomic and clone library sequences were classified as such when they grouped phylogenetically with reference HpnP sequences of the same type (Figure 1; Supplementary Figures S2 and S4). Unknown hpnP groups were defined by a lack of reference sequences. Metagenomic hpnP hits with long branches were examined for recombination events using Recombination Analysis Tool, but none were found (Etherington et al., 2005).
HpnP diversity from metagenomes and its correlation with plant–microbe interactions. Metagenomic databases were searched for hpnP-like sequences. Sequences that could be phylogenetically classified as members of the HpnP family appear in this maximum likelihood phylogeny with HpnP sequences from genomes. The colored ranges on the tree’s branches indicate alphaproteobacterial, cyanobacterial or acidobacterial clades of HpnP determined by HpnP sequences from reference genomes (Supplementary Figure S2). Metagenomic sequences that fall within one of these clades are classified as belonging to that taxon. In the inner ring surrounding the tree, HpnP sequences from metagenomes are colored by environment of origin, either hot spring, terrestrial, freshwater or marine. In the middle ring, HpnP sequences from genomes are colored to indicate the presence of nifD, moxF or both genes in the same genome. The outer ring indicates that the organism the sequence derives from was isolated from a plant-associated environment, has an established plant interaction or that a metagenomic sequence is from a plant-associated environment (soil or rhizosphere, insect waste dumps, wood compost). The light gray background corresponds to no data in the inner two rings (for example, genomes do not have an environment of origin color) or no plant association found in the outer ring; the outgroup was not included in the analysis. aLRT support values and leaf names are shown in Supplementary Figure S2. The scale bar is a measure of evolutionary distance equaling 0.1 substitutions per site.
hpnP correlation with plants
The initial observation that many hpnP-containing organisms and metagenomes were plant associated used the following criteria for a positive plant association: the organism was isolated from a plant-associated environment, had a known plant interaction established in the literature or the metagenome was from a plant-derived environment (that is, soil and rhizosphere, wood compost and insect fungal gardens, as these are maintained by leaf-cutting ants) based on available metadata (Figure 1 outer ring).
To assess the significance of the relationship between hpnP or shc and plant-associated organisms or environments, we used the hypergeometric test to evaluate if there was non-random overlap between organisms or environments that have hpnP or shc and those that are plant associated (Supplementary Table 2). Plant association for metagenomes was determined as described above. When addressing this analysis among organisms, we included in our analysis all finished bacterial genomes condensed to the species level on IMG. Organisms were counted as plant associated if a plant species was listed under ‘host name’ in the description of the genome. As filling out this data field is voluntary, we would expect more false negatives than false positives. In an attempt to reduce the number of false negatives, we mined Pubmed for abstracts describing plant associations among our list of organisms. To conduct an unbiased search, we used the following Boolean expression: two-word name of the species AND host plant OR plant host OR plant-microbe OR root-coloniz* OR plant-associat* NOT pathogen, where * allows for multiple endings. Abstract hits were manually annotated and positive hits were combined with the plant-associated list from IMG.
The hypergeometric test was also used to assess an hpnP correlation with nifD and moxF in finished bacterial genomes from IMG (Supplementary Table 3). Genomes were found to have nifD or moxF if they returned an e-value less than or equal to 1 × 10−50 when using Nostoc sp. PCC 7120 nifD (gi 4376092) and Methylobacterium extorquens AM1 moxF (YP_002965446) as queries. Similar results for nifD were also obtained for nifH.
Hopanoid analysis
Select samples were targeted for analysis by liquid chromatography-mass spectrometry for a limited number of hopanoids. These samples appear in Figure 2 with either an identifier, if hopanoids were analyzed, or blank, if hopanoids were not analyzed. We also attempted to quantify hopanoids, specifically unextended hopanoids that cannot be identified by liquid chromatography-mass spectrometry, using standard GC-MS techniques (Sessions et al., 2013), but we were unable to unambiguously detect hopanoids due to a high background. Samples were extracted as previously reported (Sessions et al., 2013).
Distribution of HpnP types in clone libraries and hopanoids from various environments. HpnP sequences generated from each sample were classified by HpnP type in a phylogenetic tree (Supplementary Figure S4). The presence of hpnP or an hpnP type is indicated in blue. Total abundances of hpnP types are reported below each environment. A red outline highlights spatial and temporal heterogeneity. Sample descriptions appear in Supplementary Table 1. Presence of hopanoids was determined for select samples by LC-MS. Abbreviations: Ia, bacteriohopanetetrol; IIa, 2-methylbacteriohopanetetrol; Ib, bacteriohopanepentol; IIb, 2-methylbacteriohopanepentol; Ic, aminobacteriohopnetriol; Id, anhydrobacteriohopanetetrol; Ie, adenosylhopane; and n.d., not detected. Other functionalized hopanoids, including aminohopanoids, were not detected. Methylated hopanoids (IIa and IIb) are highlighted in bold type.
Methylated and non-methylated bacteriohopanepolyols were identified by liquid chromatography-mass spectrometry as previously described (Welander et al., 2012). Lipids were acetylated by incubating total lipid extract (1 mg) from each sample in a 1:1 (v:v, 250 μl) mixture of acetic anhydride (Sigma-Aldrich, St. Louis, MO, USA) and pyridine (Sigma-Aldrich) for 1 h at 70 °C. Acetylated total lipid extracts were dried down under a stream of N2 and resuspended in methanol (1 ml) for a final total lipid extract concentration of 1 μg μl−1. Subsequently, each sample (5 μl) were loaded onto the liquid chromatography-mass spectrometry for analysis, a 1200 Series HPLC (Agilent Technologies, Santa Clara, CA, USA) equipped with an autosampler and a binary pump linked to a Q-TOF 6520 mass spectrometer (Agilent Technologies) via an atmospheric pressure chemical ionization interface (Agilent Technologies) operated in positive ion mode. The analytical procedure was adapted from (Talbot et al., 2001). A Poroshell 120 EC-C18 column (2.1 × 150 mm, 2.7 μm; Agilent Technologies), set at 30 °C, was eluted isocratically first with MeOH/water (95:5, v-v) for 2 min at a flow rate of 0.15 ml min−1, then using a linear gradient up to 20% (v) of isopropyl alcohol over 18 min at a flow rate of 0.19 ml min−1, and isocratic for 10 min. The linear gradient was then set to 30% (v) of isopropyl alcohol at 0.19 ml min−1 over 10 min and maintained for 5 min. The column was subsequently eluted using a linear gradient up to 80% isopropyl alcohol (v) over 1 min at a flow rate of 0.15 ml min−1 and isocratic for 14 min. Finally, the column was eluted with MeOH/water (95:5, v-v) at 0.15 ml min−1 for 5 min. The atmospheric pressure chemical ionization parameters were as follows: gas temperature 325 °C, vaporizer temperature 350 °C, drying gas (N2) flow 6 l min−1, nebulizer (N2) flow 30 l min−1, capillary voltage 1200 V, corona needle 4 μA, fragmentor 150 V. Data were recorded by scanning from m/z 100–1600. Bacteriohopanepolyols were identified on the basis of accurate mass measurements of their protonated molecular ions, fragmentation patterns in MS-MS mode and by comparison of relative retention time and the mass spectra with published data (Talbot, Squier, et al., 2003; Talbot, Summons, et al., 2003; Talbot et al., 2007b).
Results and discussion
Environmental distribution of hpnP
To identify potential biological sources of environmental 2-MeBHPs, we followed a two-pronged approach: (1) we searched all available metagenomes for the presence of hpnP (Figure 1; Table 1; Supplementary Figure S2) and (2) we generated clone libraries of hpnP sequences from diverse environments (Figure 2; Supplementary Table 1). In the first survey, 59 metagenomes were found to have hpnP, which resulted in the identification of 139 partial hpnP sequences (Supplementary Figure S2 and Supplementary Data Set S1). We also searched the same metagenomes for shc, which encodes squalene hopene cyclase, the enzyme that catalyzes the first step in hopanoid biosynthesis. For the second approach, we designed degenerated PCR primers that amplify all known diversity of hpnP (Supplementary Figure S3) and were used to retrieve 76 unique hpnP sequences from 62 samples (Supplementary Figure S4). Due to the potential for PCR bias, we did not infer the abundance of any particular hpnP type within a given library. However, we did estimate the abundance of a particular hpnP type in an environment by counting the number of samples from that environment that contained that specific hpnP type (Figure 2). Although we cannot exclude the possibility that horizontal gene transfer confounds our taxonomic assignment of hpnP sequences, there is no evidence of recent transfer events in the phylogeny of hpnP (Welander et al., 2010).
Based on both metagenomic and clone library data, we found that cyanobacterial hpnP copies are not ubiquitous in most modern habitats, constituting only 4% of metagenomic hpnP sequences (Figure 2; Table 1). Consistent with this finding, we found low abundances of metagenomic cyanobacterial shc sequences in all environments as seen previously (Table 1); (Pearson et al., 2007, 2009; Pearson and Rusch 2009). These data suggest that not only are cyanobacteria minor producers of 2-MeBHPs, but that they do not contribute substantially to hopanoid production in general. However, we cannot rule out the possibility that rare members of a community may be disproportionately active hopanoid producers.
Furthermore, some environmental hpnP sequences could not be identified as being from one of the known clades of 2-MeBHP produces. These sequences, which can be confidently identified as hpnP by homology, may represent new taxa of hpnP-containing organisms; alternatively, as the database of hpnP sequences from cultured organisms grows, we may be able to assign these sequences to previously identified clades (Figure 1, Supplementary Figures S2 and S4). Among clone library samples, the two unknown groups of hpnP segregate by environment of origin, with unknown group 1 found in most environments and unknown group 2 primarily in hot springs. Fourteen out of 36 hot spring clone libraries contained unknown hpnP sequences from group 2, nearly identical to the abundance of cyanobacterial hpnP sequences, 15 out of 36. Additionally, hpnP sequences belonging to unknown group 1 were found in 11 out of 12 pond samples (Figure 2). These observations indicate that potential novel groups of 2-MeBHP-producing bacteria may have a major role in 2-MeBHP production in hot springs and freshwater environments.
Acknowledging that comparative metagenomic analysis can be biased by the samples that have been sequenced and the methods used to sequence them, we nevertheless found a robust pattern in the available data: the majority (63%) of metagenomic hpnP sequences belong to terrestrial habitats, such as soil, the rhizosphere, insect fungal gardens and wood compost (Figure 1). This finding is not due to deeper sequencing of terrestrial environments. Using the abundance of gyrB to approximate the number of bacteria, because it is found in single copy in all bacterial genomes, we estimate that ∼1% of terrestrial bacteria have hpnP whereas less than 0.4% of bacteria in other environments surveyed have hpnP (Table 1); (Biers et al., 2009). In comparing hpnP richness between clone library data sets, rhizosphere and pond samples were found to contain more unique hpnP genes than the other environments tested (Supplementary Figure S5). Interestingly, 15% of terrestrial bacteria contain shc, whereas other habitats contain less than 7% of bacteria with shc (Table 1), implying that the same argument may be true for hopanoids in general. Taken together, these data suggest that terrestrial and freshwater environments harbor the majority of hpnP and shc diversity in modern ecosystems and should be considered likely sources of modern and possibly ancient (2-methyl)hopanoids. This finding is congruent with the presence of (2-methyl)hopanoids in terrestrial and freshwater settings (Talbot and Farrimond, 2007; Cooke et al., 2008; Pearson et al., 2009; Xu et al., 2009) and observations across a land–sea transect where (2-methyl)hopanoids appeared to be partially terrestrial in origin (Sáenz et al., 2011).
Paradoxically, the sedimentary context of the 2-methylhopane fossil record suggests ancient 2-MeBHP producers inhabited shallow, tropical marine environments (Knoll et al., 2007). It is noteworthy that our culture-independent search for hpnP sequences in all marine metagenomes, highly biased towards open water, coastal and estuarine samples, did not identify any cyanobacterial hpnP sequences. This was not due to a lack of cyanobacteria, as 13% of bacteria in marine metagenomes were estimated to be cyanobacteria based on the abundance of psbC, which encodes a component of the photosynthetic machinery present at one copy per cyanobacterial genome (Table 1); (Mulkidjanian et al., 2006). Of those hpnP sequences identified, 93% were alphaproteobacterial and 7% were of unknown origin. These sequences derived from coastal waters, estuary sediments, Ace Lake (Antarctica) and deep-sea hydrothermal vents (Table 1). These data suggest that 2-MeBHPs in some depositionally relevant modern marine environments (that is coastal water and estuaries) most likely derive from alphaproteobacteria.
To assess the capacity for 2-MeBHP production in other marine habitats that are preserved in the rock record, we analyzed hpnP clone libraries from Guerrero Negro hypersaline mats and Highborne Cay stromatolites (Hoehler et al., 2001; Dupraz and Visscher, 2005). Among Guerrero Negro samples that contained hpnP, three out of four had cyanobacterial hpnP genes whereas an equal number also contained alphaproteobacterial or unknown hpnP copies. Similarly, in Highborne Cay, within the two samples found to have hpnP, both cyanobacterial and alphaproteobacterial copies of the gene were present (Figure 2). Although it is interesting that these habitats are the only marine environments where we find cyanobacterial hpnP genes, 2-MeBHP production cannot definitely be attributed to cyanobacteria in these environments. This result contrasts with a study of hpnP diversity in Hamelin Pool Shark Bay, Australia, another locality for stromatolite deposition, where mainly cyanobacterial hpnP was recovered (Garby et al., 2012). It remains to be determined if this inconsistency is due to an inherent difference between these locales or due to differences in sampling and methodology; for example, the hpnP PCR primers used in this study were on average more degenerate than those used in Garby et al. (2012).
In microbial ecology, it is well known that microheterogeneity can exist over small spatial scales (Hunt et al., 2008). In assessing our data, we observed spatial and temporal differences in hpnP diversity (Figure 2). These differences were evident even among samples with similar geochemistry, but the causes of variation are unknown. Related to this but at the level of lipid production, the presence of 2-MeBHPs did not always correlate with the presence of hpnP (Figure 2). Although the lack of 2-MeBHPs may have been due to the detection limit of our analysis, two additional factors may explain this observation: first, 2-MeBHP production can depend on the growth condition (Rashby et al., 2007); second, the presence of 2-MeBHPs, but not hpnP, may have resulted from bacteria no longer present assuming the degradation rate of hopanoids is considerably slower than the disappearance of DNA. Therefore, the presence of hpnP does not require that a community is making 2-MeBHPs but indicates that it has the capacity to do so when the environment calls for it.
hpnP is correlated with plants
Using two independent methods, we have shown that diverse microbes have the genetic potential to produce 2-MeBHPs in a multitude of modern environments tested, making it difficult to use 2-MeBHPs as unambiguous biomarkers for any particular taxonomic group. This leaves open the question of whether 2-MeBHPs instead reflect a deeper underlying physiological function for the organisms that produce them. Notably, 43% of organisms with hpnP and 63% of hpnP sequences from metagenomes are plant associated, defined as bacteria that form commensal or mutualistic symbiotic interactions with land plants. Notable examples include Bradyrhizobium spp., Methylobacterium spp. and Nostoc spp. (Figure 1); (Knani et al., 1994; Bravo et al., 2001; Meeks, 2009). To assess whether this observation was non-random, we used the hypergeometric test calculated with the number of plant-associated bacteria estimated at the species level to alleviate bias in the data set. We also performed this test with shc-containing bacteria and metagenomes. In all cases, we found the enrichment between (meta)genomes containing hpnP or shc and those that are plant associated to be significant (P-values<10−6): 46% and 51% of hpnP-containing bacteria and metagenomes are plant associated, as well as 24 and 30% of those containing shc, in contrast to only 9% of bacteria (Supplementary Table 2). There is thus a preferential association of 2-MeBHP production, and to a lesser extent hopanoid production, with plant symbiosis.
To expand on this, we investigated the possibility that metabolisms used to establish plant–microbe interaction might be correlated to hpnP. It is common for plant symbionts to provide fixed nitrogen to plants or utilize methanol, a byproduct of plant metabolism, as a carbon source (Bravo et al., 2001; Gourion et al., 2006; Meeks, 2009). We found that 74% of hpnP-containing bacteria had either nifD or moxF, which encode proteins necessary for nitrogen fixation and methanol utilization, respectively (Figure 1, middle ring). Using the hypergeometric test, we found hpnP to be significantly overrepresented among bacteria containing nifD or moxF (P-values<10−6; Supplementary Table 3). As the presence of nifD or moxF is not exclusive to plant symbionts, these numbers are likely overestimates, whereas the percentage of hpnP-containing bacteria that are plant associated is an underestimate of the number of hpnP-containing bacteria that are plant associated because not all organisms have been tested for the ability to form symbioses.
Consistent with this finding, the hpnP gene in R. palustris TIE-1 is regulated by an extracytoplasmic function transcription factor conserved in alphaproteobacteria that has a role in establishing plant symbioses in Bradyrhizobium japonicum and Methylobacterium extorquens (Gourion et al., 2006, 2009). This factor induces hpnP expression in response to osmotic stress in R. palustris (Kulkarni et al., 2013); osmolyte production by plants in the rhizosphere is well documented, and in some cases has been shown to promote plant–microbe symbioses (Miller and Wood, 1996; Khamar et al., 2010).
Geobiological implications
Although the presence of hpnP significantly correlates with plant-associated bacteria in modern environments, the 2-methylhopane record predate the rise of land plants (Clarke et al., 2011). Thus, ancient symbioses clearly cannot explain the presence of 2-methylhopanes in the remote past. A possible explanation for why today we find the capacity for 2-MeBHP production enriched in habitats containing microbe–plant associations is that the capacity to make 2-MeBHPs is selected by particular environmental conditions present in these habitats that are similar to those in the ancient depositional context. Specifically, we note that many modern environments containing 2-MeBHP producers comprise sessile microbial communities that have suboxia or anoxia, high osmolarity and limited fixed nitrogen; these same parameters have also been used to describe the depositional context of 2-methylhopanes. For example, increased 2-methylhopane indices have been measured in ancient sedimentary rocks recording ocean anoxic events, which may have favored nitrogen fixing organisms (Knoll et al., 2007) and in sessile microbial mat and stromatolites, which contain high osmolytes in the form of extracellular polysaccharides and excreted small molecules (Summons et al., 1999). Although none of the described niche parameters are solely responsible for the presence of hpnP or 2-MeBHPs, and 2-MeBHPs are not required for occupancy of this niche, their combination appears to be correlated with the capacity for 2-MeBHP production. Determining the underlying cellular role for 2-MeBHPs given the described niche is necessary to provide a more definitive interpretation for ancient 2-methylhopanes. In conclusion, our ecological data demonstrate that 2-MeBHPs cannot be used as taxonomic biomarkers for any particular group but suggest 2-MeBHPs may be diagnostic for the confluence of particular environmental parameters.
Accession codes
References
Biers EJ, Sun S, Howard EC . (2009). Prokaryotic genomes and diversity in surface ocean waters: interrogating the global ocean sampling metagenome. Appl Environ Microbiol 75: 2221–2229.
Brasier M, Green O, Lindsay J, Steele A . (2004). Earth’s oldest (approximately 3.5 Ga) fossils and the ‘early Eden hypothesis’: questioning the evidence. Orig Life Evol Biosph 34: 257–269.
Brasier M, Green OR, Jephcoat AP, Kleppe AK, Van Kranendonk MJ, Lindsay JF et al (2002). Questioning the evidence for Earth’s oldest fossils. Nature 416: 76–81.
Bravo J, Perzl M, Härtner T, Kannenberg EL, Rohmer M . (2001). Novel methylated triterpenoids of the gammacerane series from the nitrogen-fixing bacterium Bradyrhizobium japonicum USDA 110. Eur J Biochem 268: 1323–1331.
Brocks JJ, Logan GA, Buick R, Summons RE . (1999). Archean molecular fossils and the early rise of eukaryotes. Science 285: 1033–1036.
Brocks JJ, Pearson A . (2005). Building the biomarker tree of life. Rev Mineral Geochem 59: 233–258.
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK et al (2010). QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7: 335–336.
Clarke JT, Warnock RCM, Donoghue PCJ . (2011). Establishing a time-scale for plant evolution. New Phytol 192: 266–301.
Cooke MP, Talbot HM, Farrimond P . (2008). Bacterial populations recorded in bacteriohopanepolyol distributions in soils from Northern England. Org Geochem 39: 1347–1358.
Dupraz C, Visscher PT . (2005). Microbial lithification in marine stromatolites and hypersaline mats. Trends Microbiol 13: 429–438.
Etherington GJ, Dicks J, Roberts IN . (2005). Recombination Analysis Tool (RAT): a program for the high-throughput detection of recombination. Bioinformatics 21: 278–281.
Fischer WW, Summons RE, Pearson A . (2005). Targeted genomic detection of biosynthetic pathways: anaerobic production of hopanoid biomarkers by a common sedimentary microbe. Geobiology 3: 33–40.
Garby TJ, Walter MR, Larkum AW, Neilan BA . (2012). Diversity of cyanobacterial biomarker genes from the stromatolites of Shark Bay, Western Australia. Environ Microbiol 15: 1464–1475.
Gourion B, Rossignol M, Vorholt JA . (2006). A proteomic study of Methylobacterium extorquens reveals a response regulator essential for epiphytic growth. Proc Natl Acad Sci USA 103: 13186–13191.
Gourion B, Sulser S, Frunzke J, Francez-Charlot A, Stiefel P, Pessi G et al (2009). The PhyR-sigma(EcfG) signalling cascade is involved in stress response and symbiotic efficiency in Bradyrhizobium japonicum. Mol Microbiol 73: 291–305.
Guindon S, Dufayard J-F, Lefort V, Anisimova M, Hordijk W, Gascuel O . (2010). New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 3.0. Syst Biol 59: 307–321.
Hoehler TM, Bebout BM, Des Marais DJ . (2001). The role of microbial mats in the production of reduced gases on the early Earth. Nature 412: 324–327.
Huang Y, Niu B, Gao Y, Fu L, Li W . (2010). CD-HIT Suite: a web server for clustering and comparing biological sequences. Bioinformatics 26: 680–682.
Hunt DE, David LA, Gevers D, Preheim SP, Alm EJ, Polz MF . (2008). Resource partitioning and sympatric differentiation among closely related bacterioplankton. Science 320: 1081–1085.
Katoh K, Toh H . (2008). Recent developments in the MAFFT multiple sequence alignment program. Brief Bioinform 9: 286–298.
Khamar HJ, Breathwaite EK, Prasse CE, Fraley ER, Secor CR, Chibane FL et al (2010). Multiple roles of soluble sugars in the establishment of Gunnera-Nostoc endosymbiosis. Plant Physiol 154: 1381–1389.
Knani M, Corpe WA, Rohmer M . (1994). Bacterial hopanoids from pink-pigmented facultative methylotrophs (PPFMs) and from green plant surfaces. Microbiology 140: 2755–2759.
Knoll AH, Summons RE, Waldbauer JR, Zumberge JE . (2007). The geological succession of primary producers in the oceans. In The Evolution of Primary Producers in the SeaFalkowski P, Knoll AH (ed) Academic Press: Boston, pp 133–164.
Kulkarni G, Wu C-H, Newman DK . (2013). The general stress response factor EcfG regulates expression of the C-2 hopanoid methylase HpnP in Rhodopseudomonas palustris TIE-1. J Bacteriol 195: 2490–2498.
Letunic I, Bork P . (2007). Interactive Tree Of Life (iTOL): an online tool for phylogenetic tree display and annotation. Bioinformatics 23: 127–128.
Markowitz VM, Chen I-MA, Chu K, Szeto E, Palaniappan K, Grechkin Y et al (2011). IMG/M: the integrated metagenome data management and comparative analysis system. Nucleic Acids Res 40: D123–D129.
Meeks JC . (2009). Physiological adaptations in nitrogen-fixing nostoc–plant symbiotic associations. In Microbiology Monographs: Prokaryotic Symbionts in PlantsPawlowski K (ed) Vol. 8. Springer-Verlag: Munster, Germany, pp 181–205.
Miller KJ, Wood JM . (1996). Osmoadaptation by rhizosphere bacteria. Annu Rev Microbiol 50: 101–136.
Mulkidjanian AY, Koonin E V, Makarova KS, Mekhedov SL, Sorokin A, Wolf YI et al (2006). The cyanobacterial genome core and the origin of photosynthesis. Proc Natl Acad Sci USA 103: 13126–13131.
Osburn MR, Sessions AL, Pepe-Ranney C, Spear JR . (2011) Hydrogen-isotopic variability in fatty acids from Yellowstone National Park hot spring microbial communities. Geochim Cosmochim Acta 75: 4830–4845.
Ourisson G, Rohmer M, Poralla K . (1987). Prokaryotic hopanoids and other polyterpenoid sterol surrogates. Annu Rev Microbiol 41: 301–333.
Pearson A, Flood Page SR, Jorgenson TL, Fischer WW, Higgins MB . (2007). Novel hopanoid cyclases from the environment. Environ Microbiol 9: 2175–2188.
Pearson A, Leavitt WD, Sáenz JP, Summons RE, MC-M Tam, Close HG . (2009). Diversity of hopanoids and squalene-hopene cyclases across a tropical land-sea gradient. Environ Microbiol 11: 1208–1223.
Pearson A, Rusch DB . (2009). Distribution of microbial terpenoid lipid cyclases in the global ocean metagenome. ISME J 3: 352–363.
Rashby SE, Sessions AL, Summons RE, Newman DK . (2007). Biosynthesis of 2-methylbacteriohopanepolyols by an anoxygenic phototroph. Proc Natl Acad Sci USA 104: 15099–15104.
Rasmussen B, Fletcher IR, Brocks JJ, Kilburn MR . (2008). Reassessing the first appearance of eukaryotes and cyanobacteria. Nature 455: 1101–1104.
Rohmer M . (2010). Handbook of hydrocarbon and lipid microbiology. In Chemistry and Physics of LipidsTimmis KN (ed). Springer Berlin Heidelberg: Berlin, Heidelberg.
Rohmer M, Bouvier-Nave P, Ourisson G . (1984). Distribution of hopanoid triterpenes in prokaryotes. J Gen Microbiol 130: 1137–1150.
Sáenz JP, Eglinton TI, Summons RE . (2011). Abundance and structural diversity of bacteriohopanepolyols in suspended particulate matter along a river to ocean transect. Org Geochem 42: 774–780.
Schopf JW, Packer B . (1987). Early Archean (3.3-billion to 3.5-billion-year-old) microfossils from Warrawoona Group, Australia. Science 237: 70–73.
Sessions AL, Zhang L, Welander PV, Doughty D, Summons RE, Newman DK . (2013). Identification and quantification of polyfunctionalized hopanoids by high temperature gas chromatography–mass spectrometry. Org Geochem 56: 120–130.
Summons RE, Jahnke LL, Hope JM, Logan GA . (1999). 2-Methylhopanoids as biomarkers for cyanobacterial oxygenic photosynthesis. Nature 400: 554–557.
Sun S, Chen J, Li W, Altintas I, Lin A, Peltier S et al (2011). Community cyberinfrastructure for advanced microbial ecology research and analysis: the CAMERA resource. Nucleic Acids Res 39: D546–D551.
Talavera G, Castresana J . (2007). Improvement of phylogenies after removing divergent and ambiguously aligned blocks from protein sequence alignments. Syst Biol 56: 564–577.
Talbot HM, Farrimond P . (2007). Bacterial populations recorded in diverse sedimentary biohopanoid distributions. Org Geochem 38: 1212–1225.
Talbot HM, Rohmer M, Farrimond P . (2007a). Rapid structural elucidation of composite bacterial hopanoids by atmospheric pressure chemical ionization liquid chromatography/ion trap mass spectrometry. Rapid Commun Mass Spectrometry 21: 880–892.
Talbot HM, Rohmer M, Farrimond P . (2007b). Structural characterisation of unsaturated bacterial hopanoids by atmospheric pressure chemical ionisation liquid chromatography/ion trap mass spectrometry. Rapid Commun Mass Spectrom 21: 1613–1622.
Talbot HM, Squier AH, Keely BJ, Farrimond P . (2003). Atmospheric pressure chemical ionisation reversed-phase liquid chromatography/ion trap mass spectrometry of intact bacteriohopanepolyols. Rapid Commun Mass Spectrom 17: 728–737.
Talbot HM, Summons RE, Jahnke L, Cockell CS, Rohmer M, Farrimond P . (2008). Cyanobacterial bacteriohopanepolyol signatures from cultures and natural environmental settings. Org Geochem 39: 232–263.
Talbot HM, Summons RE, Jahnke L, Farrimond P . (2003). Characteristic fragmentation of bacteriohopanepolyols during atmospheric pressure chemical ionisation liquid chromatography/ion trap mass spectrometry. Rapid Commun Mass Spectrom 17: 2788–2796.
Talbot HM, Watson DF, Murrell JC, Carter JF, Farrimond P . (2001). Analysis of intact bacteriohopanepolyols from methanotrophic bacteria by reversed-phase high-performance liquid chromatography-atmospheric pressure chemical ionisation mass spectrometry. J Chromatogr, A 921: 175–185.
Walter MR, Grotzinger JP, Schopf JW . (1992). Proterozoic stromatolites. In The Proterozoic Biosphere: A Multidisciplinary StudySchopf JW, Klien C (eds). Cambridge University Press: Cambridge, pp 253–260.
Welander PV, Coleman ML, Sessions AL, Summons RE, Newman DK . (2010). Identification of a methylase required for 2-methylhopanoid production and implications for the interpretation of sedimentary hopanes. Proc Natl Acad Sci USA 107: 8537–8542.
Welander PV, Doughty DM, Wu C-H, Mehay S, Summons RE, Newman DK . (2012). Identification and characterization of Rhodopseudomonas palustris TIE-1 hopanoid biosynthesis mutants. Geobiology 10: 163–177.
Xu Y, Cooke MP, Talbot HM, Simpson MJ . (2009). Bacteriohopanepolyol signatures of bacterial populations in Western Canadian soils. Org Geochem 40: 79–86.
Acknowledgements
We acknowledge members of the Newman lab for constructive comments on the manuscript. This work was supported by grants from the Howard Hughes Medical Institute to DKN and a NASA award (NNX12AD93G) to DKN, ALS and RES. Research access to Yellowstone hot springs was granted to JRS from the Yellowstone Center for Resources. We thank V Orphan, D Des Marais, the NASA Ames Research Center and ESSA Exportadora del Sal, SA, de CV, Guerrero Negro, Baja, California Sur, Mexico for samples of Guerrero Negro microbial mats. We are grateful to E Allen, J Valliere, D Caron and A Lie for help with sample collection. JNR was supported by an NSF graduate fellowship, MLC by an Agouron Institute postdoctoral fellowship and PVW by a NASA Astrobiology Institute postdoctoral fellowship. DKN is an HHMI Investigator.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies this paper on The ISME Journal website
Rights and permissions
About this article
Cite this article
Ricci, J., Coleman, M., Welander, P. et al. Diverse capacity for 2-methylhopanoid production correlates with a specific ecological niche. ISME J 8, 675–684 (2014). https://doi.org/10.1038/ismej.2013.191
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ismej.2013.191
Keywords
This article is cited by
-
Genetics re-establish the utility of 2-methylhopanes as cyanobacterial biomarkers before 750 million years ago
Nature Ecology & Evolution (2023)
-
Distribution of methylhopanes in some Niger Delta oils
Arabian Journal of Geosciences (2019)
-
Cyanobacterial photosynthesis under sulfidic conditions: insights from the isolate Leptolyngbya sp. strain hensonii
The ISME Journal (2018)
-
Hopanoid lipids: from membranes to plant–bacteria interactions
Nature Reviews Microbiology (2018)
-
2α-Methylhopane: Indicator for Oil–Source Correlation in the Pearl River Mouth Basin, China
Aquatic Geochemistry (2017)