Abstract
An epilithic microbial community was launched into low Earth orbit, and exposed to conditions in outer space for 548 days on the European Space Agency EXPOSE-E facility outside the International Space Station. The natural phototroph biofilm was augmented with akinetes of Anabaena cylindrica and vegetative cells of Nostoc commune and Chroococcidiopsis. In space-exposed dark controls, two algae (Chlorella and Rosenvingiella spp.), a cyanobacterium (Gloeocapsa sp.) and two bacteria associated with the natural community survived. Of the augmented organisms, cells of A. cylindrica and Chroococcidiopsis survived, but no cells of N. commune. Only cells of Chroococcidiopsis were cultured from samples exposed to the unattenuated extraterrestrial ultraviolet (UV) spectrum (>110 nm or 200 nm). Raman spectroscopy and bright-field microscopy showed that under these conditions the surface cells were bleached and their carotenoids were destroyed, although cell morphology was preserved. These experiments demonstrate that outer space can act as a selection pressure on the composition of microbial communities. The results obtained from samples exposed to >200 nm UV (simulating the putative worst-case UV exposure on the early Earth) demonstrate the potential for epilithic colonization of land masses during that time, but that UV radiation on anoxic planets can act as a strong selection pressure on surface-dwelling organisms. Finally, these experiments have yielded new phototrophic organisms of potential use in biomass and oxygen production in space exploration.
Similar content being viewed by others
Introduction
The environment of outer space is an extreme environment that contains a concatenation of different stressors such as ionizing radiation, desiccation, high ultraviolet (UV) radiation levels and extreme temperature fluctuations. The response of organisms to these stressors has been the subject of study for a number of decades (Horneck et al., 2010; Olsson-Francis and Cockell, 2010a, 2010b), and it is clear that although vacuum and low temperatures can be tolerated by a number of different organisms, the high UV flux kills exposed organisms rapidly (Horneck, 1993; Pogoda de la Vega et al., 2007). Without atmospheric protection, the full solar spectrum is detrimental because the action spectrum for UV damage increases by orders of magnitude in the UVC (200–280 nm) and shorter wavelengths (Munakata et al., 1991; Wehner and Horneck, 1995a, 1995b).
In a previous experiment, biofilms of phototrophs were exposed to conditions in low Earth orbit (LEO) for 10 days (Olsson-Francis et al., 2010) to isolate extremophilic phototrophs of potential use in biomass or oxygen provision in space exploration. The experiment showed that one species of cyanobacterium survived the exposure conditions, but that all the other organisms were killed.
As many of the stressors experienced in space can be replicated in the laboratory, a relevant question is what advantages are gained from launching organisms into space. Laboratory apparatus cannot exactly reproduce the combined stressors experienced in the space environment. In particular, it is difficult to replicate the spectral quality of the extraterrestrial UV radiation spectrum in the laboratory, even using solar simulators (Sayre et al., 1990). Exposures over long time periods are difficult to achieve. Ultimately, the best way to examine the survival of microorganisms in outer space is to carry out experiments in space.
The radiation conditions in outer space can be used to investigate environmental stressors in the Earth's past. On the early Earth, when there was a lack of a UV-protecting ozone shield during the Archean Eon (although there may have been brief periods of oxygen production; Anbar et al., 2007), surface-dwelling organisms might have been exposed to a UV flux containing wavelengths greater than 200 nm (lower wavelengths are screened by CO2), compared with wavelengths greater than ∼290 nm experienced on the Earth today (Rettberg et al., 1998; Cockell, 2000). Space experiments can be used to investigate the fate of organisms exposed to these worst-case radiation regimens, by using the natural extraterrestrial solar spectrum artificially cut off at 200 nm. The UV radiation flux on the surface of present-day Mars, with low dust loading in the atmosphere, also contains short wavelengths greater than 200 nm, so extraterrestrial UV radiation exposure experiments reveal the fate of contaminants released onto the surface of that planet from spacecraft (Cockell et al., 2000; Patel et al., 2003).
The exposure of natural microbial communities to space conditions may also allow the isolation of new taxa of microorganisms that might be useful in space applications. Rock-dwelling and weathering microorganisms, particularly phototrophs, might be used in geomicrobiological applications, for example, for breaking down rocks into soil or producing oxygen and biomass (Liu et al., 2008; Cockell, 2010; Olsson-Francis and Cockell, 2010a, 2010b).
In this experiment, we examined the survival of a community of phototrophs under space conditions to: (1) investigate outer space as a microbial selection pressure; (2) investigate the fate of phototrophs under elevated UV fluxes experienced on the early Earth and (3) isolate novel microorganisms for use in space applications.
Materials and methods
Samples
Samples of rocks with epilithic phototrophic biofilms were collected from coastal limestone cliffs at Beer, Devon, United Kingdom (50°41.50′N, 3°08.19′W). The cliffs are comprised of Cretaceous limestone. Rocks were collected from the upper greensand layer, which is located on the lower parts of the cliff. We selected this community because we hypothesized that the exposure to periodic desiccation, saltwater (during high tide), freshwater (during rain events at low tide), temperature fluctuations, solar radiation and the generally nutrient-poor environment of the limestone surface would select for a community of extremophilic organisms, some of which might be capable of tolerating the desiccation and temperature fluctuations associated with outer space. The organisms inhabit the surface and form an epilithic covering. Rocks were collected from the cliffs and cut into blocks with an upper surface area of approximately 1 cm2.
The rocks were augmented with three extreme-tolerant phototrophs. Anabaena cylindrica, a filamentous cyanobacterium, forms environmentally resistant resting states (akinetes) that are known to confer resistance to long-term desiccation (Adams and Carr, 1981). Nostoc commune is a desiccation-resistant filamentous cyanobacterium (Potts, 1994). Chroococcidiopsis is an extreme-tolerant cyanobacterium that naturally inhabits the interior and surface of desert rocks. It has tolerance to desiccation, nutrient starvation and ionizing radiation (Billi and Grilli Caiola, 1996a, 1996b).
Anabaena cylindrica was grown in BG-11 medium (Rippka et al., 1979; Olsson-Francis et al., 2010) at 25 °C, under a natural sunlight and day/night cycle. The akinetes were induced by transferring log phase cells into an iron limited (no iron) BG-11 medium, followed by three washes in the same medium. After 35 days of growth, the akinetes were harvested by allowing them to settle to the bottom of the flask. They were washed and resuspended in sterile H2O. The akinetes were stored in a refrigerator at 4 °C until required. A total of 100 μl of akinetes (3 × 104 cells ml−1) were added to the surface of the samples. Nostoc commune (PCC7524) was grown in BG-11 medium at 21 °C under natural sunlight and day/night cycle. A volume of 100 μl was mixed into the akinetes at a concentration of 3 × 105 filaments ml−1 (this is a lower approximation of viable cell numbers because filaments consist of one or more cells. In this study, filaments ranged between one and approximately 20 cells). Chroococcidiopsis 029 (culture collection of microorganisms from extreme environments) was cultured as previously described (Cockell et al., 2005). Half a milliliter of cells were mixed with the previous two organisms to a final cell concentration of 1 × 106 cells ml−1 (this is a lower approximation as some cells are tetrads and multi-cell clusters within a sheath). Augmented cells were distributed evenly on the rock surface. In all cases, cell numbers were quantified by direct counting of diluted samples under bright-field microscopy.
Sample preparation
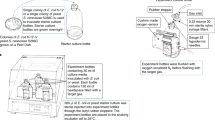
The rock samples were attached to 0.6-cm diameter quartz glass disks and fixed into the European Space Agency EXPOSE-E (EuTEF; European Technology Exposure Facility). The technical details of this facility have been described previously (Rabbow et al., 2009). A total of 32 samples were tested. In all, 16 samples were within sample containers evacuated to space conditions (vacuum) during the experiment. Four of these samples were exposed to 100% of the UV radiation >110 nm using MgF2 cut-off filters, four to 0.1% UV radiation >110 nm using additional MgF2 neutral density filters. Eight samples were dark controls, which were in corresponding positions underneath the UV-exposed samples. A total of 16 samples were within conditions designed to simulate conditions on the early Earth. The sample containers contained 103 Pa CO2. Four of these samples were exposed to 100% of the UV radiation >200 nm using quartz cut-off filters, four to 0.1% UV radiation >200 nm using additional quartz neutral density filters. Eight samples were dark controls, which were in corresponding positions underneath the UV-exposed samples.
Control rocks with and without augmented cyanobacteria were also stored in our laboratory in Milton Keynes, UK during the course of the experiment in darkness, ambient atmosphere and at 21 °C.
Space exposure
A Space shuttle (Space Shuttle Atlantis, mission STS 122; NASA, Washington, DC, USA) was used to launch the samples into low Earth orbit at a height of 340–367 km on 7 February 2008. The samples were transferred from the cargo bay of the spacecraft to the exterior of the European Space Agency Columbus module (European Space Agency, Paris, France) of the International Space Station during an astronaut space walk. The lids and valves of the EXPOSE-E facility were opened, and the samples were exposed to space conditions beginning on 20 February 2008 for a total of 548 days. After the exposure, the samples were retrieved by an astronaut and transferred to the cargo bay of the Space Shuttle Discovery (Mission STS 128). Samples were then transported from outer space to Edwards Airforce Base, California, on 12 September 2009, and sent to the Open University, Milton Keynes, UK for analysis. During the space exposure, samples not under neutral density filters, but exposed to >200 nm were exposed to between 4.40 × 105 kJm−2 and 6.47 × 105 kJm−2 (200–400 nm) UV radiation depending on the exact sample location. During the space exposure, the temperatures of the samples remained below 40 °C. However, on 20 March 2009, the samples experienced a temperature of 59.6 °C (for less than 1 hr), and a low temperature of −21.7 °C was measured on 25 March 2008. Samples also experienced a minimum temperature of at least −25 °C during an intermittent inactivation period for the EuTEF platform including EXPOSE-E from 1 September to 13 November 2008.
Ground controls
A set of replicate samples was exposed at the same time to similar conditions as the flight experiments at the DLR (Deutschen Zentrum für Luft- und Raumfahrt), Germany. The vacuum-exposed conditions in the flight experiment were simulated by evacuation to 1.7 × 10−3 Pa. Samples with a CO2 atmosphere were pressurized at 103 Pa, identical to space flight experiments. Temperatures were varied to match temperature conditions experienced by the flight experiments. A SOL2000 solar simulator was used to simulate UV exposure (which produces wavelengths greater than 200 nm), and samples were exposed to UV irradiances such that total fluences in the ground experiments matched fluences in the flight experiments. The spectral quality of the lamp is provided in Onofri et al. (2008).
Bright-field microscopy
Bright-field microscopy was used to examine the biofilms after the space exposure. Small parts of the biofilm, including the natural community and augmented organisms, were removed from the rock with a sterile blade under aseptic conditions. The biofilm section was transferred to the surface of a glass microscope slide, rehydrated and examined on a Leica DMRP fluorescence microscope (Leica Microsystems, Wetzlar, Germany). Autofluorescence of cyanobacteria was determined with a Leica N2.1 cube (Leica Microsystems), using an excitation filter with a bandpass of 515–560 nm and a long-band emission filter (>590 nm).
Scanning electron microscopy
The structure of the biofilm in controls and UV-exposed samples was examined by scanning electron microscopy. Small sections of rocks were prepared by carefully breaking the samples and mounting them on aluminum stubs with two-sided carbon tape. They were gold coated (15–20 nm thickness) and examined in secondary electron mode at a 20 kV accelerating voltage and 7–15 mm working distance using a Quanta 3D dual beam FIBSEM (FEI, Hillsboro, OR, USA).
Raman spectroscopy
Raman spectroscopy was used to investigate the fate of the phototroph carotenoids in the surface layer. Raman was carried out using a Horiba Jobin Yvon LabRAM (Horiba Jobin Yvon, Stanmore, UK), and samples were excited using a 514.5 nm (green) laser at 0.7 mW. The footprint area of analysis was a 2 μm diameter spot. Spectra were the mean of five separate spectra, each acquired over 20 s to increase the signal to noise ratio. Wavenumber values were accurate to ±1 cm−1. Data were gathered in the program LabSpec (Horiba Jobin Yvon, Stanmore, UK). At least 10 different spots on the surface of each sample were examined.
Microbial survival study
Following exposure, each of the rocks from the space exposure experiments and the ground-controls were split aseptically and a half was incubated in 5 ml of BG-11 medium at 25 °C, under a 16/8 h day light cycle (LEEC Incubator, model PL2, Nottingham, UK). Cyanobacterial and algal growth was routinely monitored using the Leica DMRP microscope. We also enriched four control rocks, which were retained in our laboratory during the experimental period. After 8 weeks of growth, microorganisms were isolated on BG-11 agar plates (1% agar, Bacteriological Agar No. 1, Oxoid, Basingstoke, UK).
Molecular analysis
For analysis of the indigenous microbial community in the samples, rocks were collected from the field site aseptically and stored in sterile plastic bags (Whirlpak, Fisher Scientific, Loughborough, UK) at −80 °C. For DNA extraction, three of the collected rocks were crushed, as previously described (Herrera and Cockell, 2007). The powdered rock was grounded in a sterile pestle and mortar containing liquid nitrogen. DNA was extracted from 5 g of the crushed rock, using the PowerMax Soil DNA Isolation Kit (MoBio Laboratories, Cambridge, UK) according to the manufacturer's instructions.
PCR was conducted on the extracted DNA using the bacterial-specific 16S rRNA gene primers pA (5′-GTT TGA TCC TGG CTC AG-3′; E. coli nucleotide positions 11–28) and Com2 (5′-TCA ATT CCT TTG AGT TT-3′; E. coli nucleotide positions 907-924), which cover the V1–V5 region of the 16S rRNA gene (Bruce et al., 1992; Schwieger and Tebbe, 1998). Algal sequences were amplified using algal primers p23srV_f1 (5′-GGA CAG AAA GCA CCT-3′) and P23srV_r1 (5′-TCA GCC TGT TAT CCC TAG AG-3′) (Sherwood and Presting, 2007), which cover the 23S plastid domain V region. The PCR products were extracted and purified from a 0.8% (w/v) agarose gel (Invitrogen, Paisley, UK) using the GenElute Gel Extraction kit (Sigma–Aldrich, Poole, UK). The purified product was ligated at 4 °C, with the pCR4-TOPO vector. Chemical transformation was conducted with the OneShot TOP10 chemically competent Escherichia coli from the TOPO-TA cloning kit (Invitrogen).
The 16S rDNA and 23S rDNA inserts were sequenced using the T7 and T3 universal primers. Putative chimeras were identified with the Chimera check program, Bellerophon, of the Greengenes website (http://greengenes.lbl.gov). The sequences were phylogenetically classified using the Classifier in the Ribosomal Data Project II (Cole et al., 2009) and the nearest sequences were identified in the GenBank database using the BLASTN program (Altschul et al., 1990). On the basis of these results, the sequences were aligned with ClustalX (Thompson et al., 1997). The phylogenetic trees were constructed using the neighbour-joining method in MEGA4 (Saitou and Nei, 1987; Tamura et al., 2007). Analysis of the cyanobacterial components of the rock was presented previously (Olsson-Francis et al., 2010).
Isolate identification
Molecular and morphological techniques were used to identify the microbial isolates. A Leica DMRP microscope was used to morphologically distinguish between the algae and cyanobacteria. Identification of isolates was confirmed by PCR with the bacterial or algal primers. PCR was carried out directly from colonies using DNA template, which was prepared by dispersing a colony in 50 μl of 10 mM TE buffer, pH 7.5 and freeze-thawing at −20 °C. The PCR products were cleaned using the GenElute Gel Extraction kit (Sigma–Aldrich) and the DNA sequencing of the PCR products was carried out directly (McLab, San Francisco, CA, USA).
Nucleotide sequence accession numbers
All of the sequences obtained from the clone libraries and the isolates were deposited in the GenBank database under accession numbers HQ917700-HQ917849.
Results
Samples: visual and bright-field observations
Dark control samples returned from space exhibited no obvious differences under visual inspection compared with non-flight controls. Similarly, samples exposed to >110 nm and >200 nm radiation, but under a 0.1% neutral density filter, showed no visible differences to dark and non-flight controls (Figure 1). However, the surface of samples exposed to 100% UV radiation >110 nm and >200 nm were visibly bleached. Under bright-field microscopy, no obvious alterations were observed in the surface layers in dark controls and those under 0.1% neutral density filters (dark control sample shown in Figure 2a). Samples exposed to 100% UV radiation >110 nm or >200 nm UV radiation had developed a brown coloration on their surfaces (Figure 2b). These cells did not autofluoresce, although their morphology was preserved. Cells underneath the brown surface layer exhibited no obvious alteration in color, and they autofluoresced with intensity similar to the controls.
Micrographs of space flight dark control cells and cells exposed to UV radiation >200 nm (CO2 atmosphere). (a) Bright-field micrograph of surface biofilm of dark control sample (scale bar 20 μm). (b) Bright-field micrograph of surface biofilm of UV-exposed sample. Brown cells in upper section of micrograph correspond to exposed surface layer, green cells below them are cells protected under the surface layer (scale bar 20 μm). (c) Secondary scanning electron microscopy image of surface dark control sample showing biofilm (detachment of biofilm was observed in all samples and is a result of desiccation) (scale bar 25 μm). Cells are bound together by polysaccharide (d) scanning electron microscopy of sample exposed to 100% UV radiation >200 nm showing detached biofilm (scale bar 25 μm).
Scanning electron microscopy
The integrity of the cell biofilms was preserved in both control and UV-exposed samples (Figures 2c and d). Individual surface cells in all samples preserved their morphology under scanning electron microscopy, consistent with bright-field microscopy.
Raman spectroscopy
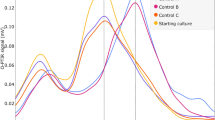
Raman spectra of the surface of the biofilms in darkened (control) conditions exhibited the distinctive features of carotenoids. This spectrum was identical in all dark controls and laboratory non-flight controls (Figure 3). Major peaks observed corresponded to the C-CH3 rocking mode at 1005 cm−1, the C–C bond at 1157 cm−1, C-CH3 bond at 1191 cm−1 and 1212 cm−1 and the C=C bond at 1522 cm−1, at similar wavenumbers to those observed in other carotenoids (de Oliveira et al., 2009), for example, β-carotene (Arcangeli and Cannistraro, 2000). In samples exposed to unfiltered (100%) UV flux with either a >110 nm or >200 nm cut-off filter, the carotenoid features were completely absent and the spectra displayed a generic fluorescence background signal (Figure 3). However, in samples on which the UV flux had been reduced to 0.1% using a neutral density filter, carotenoid signatures were observed.
Microbial clone libraries and culturing
Clone libraries
The results from clone library analysis are shown in Figures 4a (algae), 4b (cyanobacteria; full cyanobacterial tree described in Olsson-Francis et al., 2010) and Figure 5 (bacteria).
Phototroph phylogenetic trees. (a) Neighbour-joining tree of the algal community of the limestone rock using the plastid 23S domain V plastid rDNA sequence data (clone library sequences in bold). The clones are represented as phylotypes (defined as 97% sequence similarity). The percentage of clones in each phylotype is shown in parentheses. Isolates that survived in the space flight samples are underlined. The scale bar corresponds to 0.02 changes per nucleotide. The percentage of bootstrap replicates (1000 replicates) resulting in the same cluster is given near the respective nodes for bootstrap values higher than 80%. Phormidium articulatum was used as an outgroup. (b) 16S rDNA phylogenetic tree of the isolates from the cyanobacterial community of the limestone rocks (for the full community analysis see Olsson-Francis et al., 2010). The neighbour-joining tree was constructed using nucleotide positions 112–770 (E. coli numbering). The scale bar corresponds to 0.02 changes per nucleotide. The percentage of bootstrap replicates (1000 replicates) resulting in the same cluster is given near the respective nodes for bootstrap values higher than 80%. The isolate that survived in the space flight sample is underlined. E. coli was used as an outgroup.
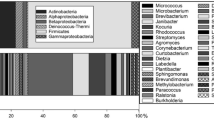
The non-cyanobacterial bacterial phylogenetic tree (cyanobacterial wedge shown in Figure 4b). The neighbour-joining tree was constructed using nucleotide positions 30–902 (E. coli numbering). Phylotypes obtained from the rock clone library are shown in bold. The two isolates examined in this study are underlined. The scale bar corresponds to 0.02 changes per nucleotide. The percentage of bootstrap replicates (1000 replicates) resulting in the same cluster is given near the respective nodes for bootstrap values higher than 80%. Aquifex sp. was used as an outgroup.
Culturable organisms
In this study, nine culturable cyanobacteria and two algae (Table 1) were isolated (Figures 4a and b). During the isolation of cyanobacteria and algae in BG-11, two non-cyanobacterial prokaryotes were isolated from the natural biofilm that had distinctive colony color (Table 1; Figure 5) and survived in the ground-based or space flight experiments. One, an α-proteobacteria isolate, was most closely affiliated to Geminicoccus roseus, an aerobic phototrophic bacterium (Foesel et al., 2007) and formed pink colonies. The other, a Bacteroidetes isolate, most closely affiliated with Gramella, a genus involved in marine hydrocarbon degradation in microbial assemblages (Bauer et al., 2006) and formed cream colonies. In addition, two algae also grew in control enrichment cultures (Figure 4a), but were not isolated and did not survive any of the space or ground-simulation experiments.
Ground simulation
In the ground simulation experiments neither A. cylindrica nor N. commune survived in any of the UV-exposed samples, but cells of these species were cultured from dark control samples, although both species survived dark conditions better under 103 Pa CO2 than evacuation (Table 1). Chroococcidiopsis was cultured from all samples. In all, 5 of the 9 cyanobacterial isolates from the natural community survived in dark, two of which were also cultured from UV-exposed samples (Leptolyngbya OU_13 and Gloeocapsa OU_20) (Table 1). Both algal isolates survived dark conditions, one of which (Chlorella sp.) was cultured from UV-exposed samples (Table 1). Both non-cyanobacterial prokaryotes, OU_22 (Bacteroidetes) and OU_21 (α-proteobacteria), survived in dark and UV-exposed conditions (Table 1).
Space exposure experiments
Following 548 days in low Earth orbit, viable A. cylindrica cells were cultured from a dark control (under 103 Pa CO2). N. commune was not cultured from any samples. Chroococcidiopsis was cultured from all samples. Only one of the culturable cyanobacterial species from the natural biofilm survived. It was recovered from dark control samples, but from none of the UV-exposed samples (Table 1). The isolate was the previously described Gloeocapsa OU_20 that survived 10 day exposure to conditions in low Earth orbit (Olsson-Francis et al., 2010). The two culturable algal species from the natural biofilm (OU_26 and OU_27) survived dark control conditions. Only the Chlorella sp. (OU_26) survived in the UV-exposed conditions and was cultured from samples exposed to a 0.1% flux under >110 nm and >200 nm (Table 1). The two non-cyanobacterial prokaryotes survived dark conditions and 0.1% UV flux >200 nm (Table 1).
Organisms in ground-control experiments generally exhibited greater survival than space-exposed samples. Five cyanobacterial species (compared with one after space exposure) and both non-cyanobacterial prokaryotes (compared with one after space exposure) survived in ground-based dark conditions. One cyanobacterial isolate (OU_13) and one non-cyanobacterial prokaryote (OU_22) survived the UV-exposed conditions in ground-experiments that their space-exposed counterparts did not.
Discussion
A diversity of microorganisms has been exposed to conditions in outer space, but few natural communities have been examined. In this experiment, we studied the effects of prolonged (548 days) conditions in low Earth orbit on a natural community of rock-dwelling phototrophs augmented with A. cylindrica akinetes and extreme-tolerant vegetative cells of Nostoc commune and Chroococcidiopsis.
By using cut-off filters at defined wavelengths, the extraterrestrial UV radiation flux can be used to simulate past UV environments. The early Earth, during the time when life first arose, is thought to have been anoxic. In the worst-case scenario without an ozone shield and no other shielding, UV radiation wavelengths greater than 200 nm might have penetrated to the surface, capable of causing at least three orders of magnitude more damage to DNA (Rettberg et al., 1998; Cockell and Horneck, 2001). In our experiments, only the Chroococidiopsis survived in samples exposed to the >200 nm unattenuated UV flux. When this flux was reduced to 0.1%, one cyanobacterium from the natural biofilm (Gloeocapsa OU_20), one alga (Chlorella sp.) and the α-proteobacterium isolate survived. Chlorella is known to be UV tolerant (Mehta and Hawxby, 1977; Hsu and Hsu, 1998; Lüttge and Büdel, 2010), although this is the first time a member of this genus has been investigated under an extraterrestrial UV flux.
The sample sizes we studied were small because of logistical constraints in launching organisms into Earth orbit, so caution must be exercised in interpreting the results. For example, we do not know precisely the abundance of cells of each isolate of the natural community on each sample. However, these data show that in a worst-case scenario, early Earth UV fluxes would have been extremely detrimental to surface-dwelling epilithic organisms, particularly those in an inactive state.
The detrimental effects of these UV fluxes are confirmed by the analysis of the surface layer, which showed that the unattenuated UV flux not only killed organisms, but also bleached the chlorophyll and accessory pigments and destroyed carotenoids, as observed by bright-field microscopy and Raman spectroscopy. The observations are consistent with previous data. For example, a fluence of 5.76 kJ m−2 UV-B radiation caused plasmolysis of a species of Chlorella cells and destruction of the thylakoids (Juan et al., 2005). At fluences of greater than 15 kJ m−2, the viability of Nostoc cells was rapidly reduced and accessory pigments bleached (Aráoz and Häder, 1997; Aráoz et al., 1998). Although organisms beneath the altered surface layer did not exhibit obvious alteration by bright-field microscopy, the data suggest that the detrimental effects of UV radiation can penetrate to the natural community covered by the augmented cyanobacteria. UV radiation damage could have been caused either directly to cell components such as DNA, which did not manifest itself as destruction of accessory pigments or carotenoids, or indirectly by the production of damaging radicals in the surface layer that subsequently destroyed the subsurface layers (Hansen et al., 2009).
The greater survival of organisms in dark controls compared with UV exposure, both in ground-simulation and space exposure experiments, is consistent with data obtained with other organisms such as Bacillus subtilis, which were rapidly killed by extraterrestrial UV radiation, but not when shielded (Horneck, 1993; Rettberg et al., 2002; Schuerger et al., 2003).
We also observed generally greater survival of organisms in ground-based experiments than in space-exposed samples. This observation might be explained by the different radiation environments. The quality of the UV fluxes is different in space than the ground-based UV lamp. Extraterrestrial UV fluxes have higher fluxes of short-wavelength UV radiation, which are likely to have been more detrimental to the UV-exposed samples. However, this does not explain the difference between the dark control samples. One explanation is that the space-exposed samples were affected by ionizing radiation. The total ionizing dose during the experiments was determined using an ionizing radiation dosimeter within the EXPOSE-E facility as 240 mGy (TP Dachev, Space Research Institute, Sofia, Bulgaria, personal communication). The ionizing radiation resistance of cyanobacteria has not been well explored, but Chroococcidiopsis is known to be resistant up to 10 kGy (Billi et al., 2000). In experiments in which we exposed the natural rock community to doses of 5 kGy using a 60Co source, only Gloeocapsa OU_20 survived (unpublished data). The lower tolerance of the other components may have contributed to their loss of viability even at the doses received in this experiment, depending upon their dose-response functions.
Despite these results, cells of Chroococcidiopsis did survive under all UV-exposed conditions in ground and space-based experiments. The results are unlikely to be explained by UV radiation resistance alone. In a study using mono-layers of the same strain of Chroococcidiopsis, cells were killed in <1 day by the UV fluxes in the Atacama Desert (Cockell et al., 2008). Similarly, Chroococcidiopsis exposed to a simulated (>200 nm) Martian UV flux was killed in less than 30 min (Cockell et al., 2005). In the Atacama study, a small subpopulation of cells survived, which was attributed to self-shielding by clumps of cells in areas of an imperfect monolayer. Self-shielding might have contributed to cell survival in the work reported in this study, suggested by the presence of unbleached cells under the brown altered surface layer. Other factors may have contributed to survival. Chroococcidiopsis is a polyextremophile, tolerant of multiple combined stressors including desiccation, ionizing radiation and temperature excursions (Grilli Caiola et al., 1993; Billi et al., 2000; Billi and Grilli Caiola, 1996a, 1996b), all three of which were experienced by the samples during this experiment. Finally, the high cell numbers of Chroococccidiopsis might have contributed. The organism had approximately 25 times greater number of cells than A. cylindrica on the rock surface, although the number of viable cells was similar to Nostoc commune, given the filamentous characteristic of the latter organism.
Applied to the worst-case UV radiation scenario on the early Earth or similar anoxic planets, the result shows that although UV fluxes would have been detrimental and a strong selection pressure on epilithic communities, some factors would have allowed organisms to survive. They include tolerance of extreme environmental conditions, the ‘matting’ habit, whereby cells are shielded by surface layers (Sagan, 1973; Margulis et al., 1976; Olson and Pierson, 1986; Pierson et al., 1993), and high cell numbers. Either individually or in combination, these factors would have allowed the surfaces of rocks to be colonized, despite the destruction of biomolecules in the surface layers.
The work reported in this study also revealed new microorganisms of potential use in space exploration. Phototrophs have a number of uses, for example, in oxygen production in life support systems, the amelioration of planetary regolith (surface material) and the extraction of useful elements from rocks (Cockell, 2010). The data show that some phototrophs can survive under space conditions, but that high UV fluxes or the combined stressors of space conditions even under dark conditions (including temperature fluctuations) kill most organisms. Consistent with earlier results from the exposure of the same biofilm to 10 days in low Earth orbit, we isolated the cyanobacterium (Gloeocapsa OU_20) (Olsson-Francis et al., 2010). The EXPOSE-E experiment reported in this study extends the known survival of this organism to conditions in low Earth orbit to 548 days. Our experiments also resulted in the identification of two novel algal species, neither of which was isolated in the previous 10 day experiment. One explanation may be heterogeneities in the abundance or presence of the organisms on the rock surface, so that in the 10 day experiment they were either not present on the samples, or their population was not sufficiently abundant for a sub-population of cells to survive. Nevertheless, our results show that space exposure experiments can result in the identification of novel, potentially useful, eukaryotic fast-growing phototrophs. The two algal species closely affiliate to genera (Chlorella and Rosenvingiella), known to have representatives that can tolerate environmental extremes (Belcher, 1969; Broady, 1996; Rindi et al., 2004). The two novel non-cyanobacterial prokaryotic isolates belong to genera not previously characterized for tolerance to extreme environmental conditions.
In conclusion, we have shown that prokaryotic and eukaryotic phototrophs can survive conditions in low Earth orbit for a year and a half and that these conditions act as a selective pressure on communities. Our data show that if the early Earth was exposed to high UV radiation (>200 nm), it would have been an important selection pressure on epilithic communities. However, some organisms could have survived the unattenuated flux in an inactive state for considerable lengths of time. Future work must investigate the physiological capabilities of the novel extremophiles isolated from the rocks and their potential practical uses.
Accession codes
References
Adams DG, Carr NG . (1981). Heterocyst differentiation and cell division in the cyanobacterium Anabaena cylindrica: effect of high light intensity. J Cell Sci 49: 341–352.
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ . (1990). Basic local alignment search tool. J Mol Biol 215: 403–410.
Anbar AD, Duan Y, Lyons TW, Arnold GL, Kendall B, Creaser RA et al. (2007). A whiff of oxygen before the great oxidation event? Science 317: 1903–1906.
Aráoz R, Häder DP . (1997). Ultraviolet radiation induces both degradation and synthesis of phycobilisomes in Nostoc sp: a spectroscopic and biochemical approach. FEMS Microbiol Ecol 23: 301–313.
Aráoz R, Shelton M, Lebert M, Häder D-P . (1998). Differential behaviour of two cyanobacterium species to UV radiation. Artificial UV radiation induces phycoerythrin synthesis. J Photochem Photobiol 44: 175–183.
Arcangeli C, Cannistraro S . (2000). In situ Raman microspectroscopy identification and localisation of carotenoids: approach to monitoring of UV-B irradiation stress on Antarctic fungus. Biopolymers 57: 179–186.
Bauer M, Kube M, Teeling H, Richter M, Lombardot T, Allers E et al. (2006). Whole genome analysis of the marine Bacteroidetes Gramella forsetii reveals adaptations to degradation of polymeric organic matter. Environ Microbiol 8: 2201–2213.
Belcher JH . (1969). Prasiococcus calcarius (Boye Petersen) Vischer in the South Sandwich Islands. Br Phycol J 4: 119–120.
Billi D, Friedmann EI, Hofer KG, Grilli Caiola M, Ocampo-Friedmann R . (2000). Ionizing radiation resistance in the desiccation-tolerant cyanobacterium Chroococcidiopsis. Applied Environ Microbiol 66: 1489–1492.
Billi D, Grilli Caiola M . (1996a). Effects of nitrogen limitation and starvation on Chroococcidiopsis sp. (Chroococcales). New Phytol 133: 563–571.
Billi D, Grilli Caiola M . (1996b). Effects of nitrogen and phosphorus deprivation on Chroococcidiopsis sp. (Chroococcales) Algal Studies 83: 93–105.
Broady PA . (1996). Diversity, distribution and dispersal of Antarctic terrestrial algae. Biodivers Conserv 5: 1307–1335.
Bruce KD, Hiorns WD, Hobman JL, Osborn AM, Strike P, Ritchie DA . (1992). Amplification of DNA from native populations of soil bacteria by using the polymerase chain reaction. Appl Environ Microbiol 58: 3413–3416.
Cockell CS . (2000). The ultraviolet history of the terrestrial planets–implications for biological evolution. Planet Space Sci 48: 203–214.
Cockell CS . (2010). Geomicrobiology beyond Earth - microbe-mineral interactions in space exploration and settlement. Trends Microbiol 18: 308–314.
Cockell CS, Catling D, Davis WL, Kepner RN, Lee PC, Snook K et al. (2000). The ultraviolet environment of Mars, biological implications past, present and future. Icarus 146: 343–359.
Cockell CS, Horneck G . (2001). The ultraviolet climate of Earth, past and present–theoretical and space-based observations. Photochem Photobiol 73: 447–451.
Cockell CS, McKay CP, Warren-Rhodes KA, Horneck G . (2008). Ultraviolet radiation-induced limitation to epilithic microbial growth in arid deserts-dosimetric experiments in the hyperarid core of the Atacama Desert. J Photochem Photobiol 90: 79–87.
Cockell CS, Schuerger AC, Billi D, Friedmann EI, Panitz C . (2005). Effects of a simulated martian UV flux on the cyanobacterium, Chroococcidiopsis sp. 029. Astrobiology 5: 127–140.
Cole JR, Wang Q, Cardenas E, Fish J, Chai B, Farris RJ et al. (2009). The Ribosomal Database Project: improved alignments and new tools for rRNA analysis. Nucleic Acids Res 37: D141–D145.
De Oliveira VE, Castro HV, Edwards HGM, de Oliveira LF . (2009). Carotenes and carotenoids in natural biological samples: a Raman spectroscopic analysis. J Raman Spectro 41: 642–650.
Foesel BU, Göβner A, Drake HL, Schramm A . (2007). Geminicoccus roseus gen. nov., sp. nov., an aerobic phototrophic Alphaproteobacterium isolated from a marine aquaculture biofilter. Syst Appl Microbiol 30: 581–586.
Grilli Caiola M, Ocampo-Friedmann R, Friedmann EI . (1993). Cytology of long-term desiccation in the desert cyanobacterium Chroococcidiopsis (Chroococcales). Phycologia 32: 315–322.
Hansen AA, Jensen LL, Kristoffersen T, Mikkelsen K, Merrison J, Finster KW et al. (2009). Effects of long-term simulated martian conditions on a freeze-dried and homogenized bacterial permafrost community. Astrobiology 9: 229–240.
Herrera A, Cockell CS . (2007). Exploring microbial diversity in volcanic environments: a review of methods in DNA extraction. J Microbiol Methods 70: 1–12.
Horneck G . (1993). Responses of Bacillus subtilis spores to space environment: results from experiments in space. Origin Life Evol Biosph 23: 37–52.
Horneck G, Klaus DM, Mancinelli RL . (2010). Space microbiology. Microbial Mol Biol Rev 74: 121–156.
Hsu S-J, Hsu B-D . (1998). Flow cytometry of Chlorella after dehydration stress. Plant Sci 134: 163–169.
Juan Y, Xuexi T, Peiju Z, Jiyuan T, Shuanglin D . (2005). Physiological and ultrastructural changes of Chlorella sp. induced by UVB radiation. Prog Nat Sci 15: 678–683.
Liu YD, Cockell CS, Wang G, Hu CX, Chen L, De Philippis R . (2008). Control of lunar and Martian dust—experimental insights from artificial and natural cyanobacterial and algal crusts in the desert of Inner Mongolia, China. Astrobiology 8: 75–86.
Lüttge U, Büdel B . (2010). Resurrection kinetics of photosynthesis in desiccation-tolerant terrestrial green algae (Chlorophyta) on tree bark. Plant Biol 12: 437–444.
Margulis L, Walker JCG, Rambler M . (1976). Reassessment of roles of oxygen and ultraviolet light in Precambrian evolution. Nature 264: 620–624.
Mehta R, Hawxby K . (1977). Use of ultraviolet radiation to achieve bacteria-free algal culture. Proc Okla Acad Sci 57: 54–60.
Munakata N, Saito M, Hieda K . (1991). Inactivation action spectra of Bacillus subtilis spores in extended ultraviolet wavelengths (50–300 nm) obtained with synchrotron radiation. Photochem Photobiol 54: 761–768.
Olson JM, Pierson BK . (1986). Photosynthesis 3.5 thousand million years ago. Photosynthesis Res 9: 251–259.
Olsson-Francis K, Cockell CS . (2010a). Use of cyanobacteria in in-situ resource use in space applications. Planet Space Sci 58: 1279–1285.
Olsson-Francis K, Cockell CS . (2010b). Experimental methods for studying microbial survival in extraterrestrial environments. J Microbiol Methods 80: 1–13.
Olsson-Francis K, de la Torre R, Cockell C . (2010). Isolation of novel extreme-tolerant cyanobacteria from a rock-dwelling microbial community by using exposure to low Earth orbit. Appl Environ Microbiol 76: 2115–2121.
Onofri S, Barreca D, Selbmann, Isola D, Rabbow E, Horneck G et al. (2008). Resistance of Antarctic black fungi and cryptoendolithic communities to simulated space and Martian conditions. Stud Microl 61: 99–109.
Patel MR, Bérces A, Kolb C, Lammer H, Rettberg P, Zarnecki JC et al. (2003). Seasonal and diurnal variations in Martian surface ultraviolet irradiation : biological and chemical implications for the Martian regolith. Int J Astrobiol 2: 21–34.
Pierson BK, Mitchell HK, Ruff-Roberts AL . (1993). Chloroflexus aurantiacus and ultraviolet radiation : implications for archean shallow-water stromatolites. Origin Life Evol Biosph 23: 243–260.
Pogoda de la Vega U, Rettberg P, Reitz G . (2007). Simulation of the environmental climate conditions on martian surface and its effect on Deinococcus radiodurans. Adv Space Res 40: 1672–1677.
Potts M . (1994). Desiccation tolerance of prokaryotes. Microbiol Mol Biol Rev 58: 755–805.
Rabbow E, Horneck G, Rettberg P, Schott JU, Panitz C, L’Affilitto A et al. (2009). EXPOSE, an astrobiological exposure facility on the International Space Station–from proposal to flight. Origin Life Evol Biosph 39: 581–598.
Rettberg P, Eschweiler U, Strauch K, Reitz G, Horneck G, Wänke H et al. (2002). Survival of microorganisms in space protected by meteorite material: results of the experiment EXOBIOLOGIE of the PERSEUS mission. Adv Space Res 30: 1539–1545.
Rettberg P, Horneck G, Strauch W, Facius R, Seckmeyer G . (1998). Simulation of planetary UV radiation climate on the example of the early Earth. Adv Space Res 22: 335–339.
Rindi F, McIvor L, Guiry D . (2004). The prasiolales (Chlorophyta) of Atlantic Europe: an assessment based on morphological, molecular, and ecological data, including the characterization of Rosenvingiella radicans (Kützing) comb nov. J Phycol 40: 977–997.
Rippka R, Deruelles J, Waterbury JB, Herdman M, Stanier RY . (1979). Generic assignments, strain histories and properties of pure cultures of cyanobacteria. J Gen Microbiol 111: 1–61.
Sagan C . (1973). Ultraviolet radiation selection pressure on the earliest organisms. J Theor Biol 39: 195–200.
Saitou N, Nei M . (1987). The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4: 406–425.
Sayre RM, Cole C, Billhimer W, Stanfield J, Ley RD . (1990). Spectral comparison of solar simulators and sunlight. Photodermatol Photoimmul Photomed 7: 159–165.
Schuerger AC, Mancinelli RL, Kern RG, Rothschild LJ, McKay CP . (2003). Survival of Bacillus subtilis on spacecraft surfaces under simulated Martian environments: implications for the forward contamination of Mars. Icarus 165: 253–276.
Schwieger F, Tebbe CC . (1998). A new approach to utilize PCR-single-strand-conformation polymorphism for 16S rRNA gene-based microbial community analysis. Appl Environ Microbiol 64: 4870–4876.
Sherwood AR, Presting GG . (2007). Universal primers amplify a 23S rDNA plastid marker in eukaryotic algae and cyanobacteria. J Phycology 43: 605–608.
Tamura K, Dudley J, Nei M, Kumar S . (2007). MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24: 1596–1599.
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG . (1997). The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25: 4876–4882.
Wehner J, Horneck G . (1995a). Effects of vacuum UV and UVC radiation on dry E coli plasmid pUC19, I inactivation, lacZ mutation indication and strand breaks. J Photochem Photobiol B: Biol 28: 77–85.
Wehner J, Horneck G . (1995b). Effects of vacuum UV and UVC radiation on dry Escherichia coli plasmid pUC19: II. Mutational specificity at the lacZ gene. J Photochem Photobiol B: Biol 30: 171–177.
Acknowledgements
This work was supported by an STFC Grant (PP/E001408/1). We thank the European Space Agency for the flight opportunity. This work was conducted as part of the ADAPT experiment on EXPOSE-E.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cockell, C., Rettberg, P., Rabbow, E. et al. Exposure of phototrophs to 548 days in low Earth orbit: microbial selection pressures in outer space and on early earth. ISME J 5, 1671–1682 (2011). https://doi.org/10.1038/ismej.2011.46
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ismej.2011.46
Keywords
This article is cited by
-
Common loss of far-red light photoacclimation in cyanobacteria from hot and cold deserts: a case study in the Chroococcidiopsidales
ISME Communications (2023)
-
Molecular repertoire of Deinococcus radiodurans after 1 year of exposure outside the International Space Station within the Tanpopo mission
Microbiome (2020)
-
Photoprotective role of UV-screening pigment scytonemin against UV-B-induced damages in the heterocyst-forming cyanobacterium Nostoc sp. strain HKAR-2
Brazilian Journal of Botany (2020)
-
New ecosystems in the deep subsurface follow the flow of water driven by geological activity
Scientific Reports (2019)
-
Phototrophic biofilms: diversity, ecology and applications
Journal of Applied Phycology (2017)