Abstract
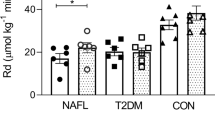
Supervised exercise reduces liver fat and improves endothelial function, a surrogate of cardiovascular disease (CVD) risk, in nonalcoholic fatty liver disease (NAFLD). We hypothesised that after a 16-week supervised exercise program, patients would maintain longer-term improvements in cardiorespiratory fitness, liver fat and endothelial function. Ten NAFLD patients (5/5 males/females, age 51±13 years, body mass index 31±3 kg m−2 (mean±s.d.)) underwent a 16-week supervised moderate-intensity exercise intervention. Biochemical markers, cardiorespiratory fitness (VO2peak), subcutaneous, visceral and liver fat (measured by magnetic resonance imaging and spectroscopy respectively) and brachial artery flow-mediated dilation (FMD) were assessed at baseline, after 16 weeks of supervised training and 12 months after ending supervision. Despite no significant change in body weight, there were significant improvements in VO2peak (6.5 ml kg−1 min−1 (95% confidence interval 2.8, 10.1); P=0.003), FMD (2.9% (1.5, 4.2); P=0.001), liver transaminases (P<0.05) and liver fat (−10.1% (−20.6, 0.5); P=0.048) immediately after the 16-week supervised training. Nevertheless, 12 months after ending supervision, VO2peak (0.9 ml kg−1 min−1 (−3.3, 5.1); P=0.65), FMD (−0.07% (−2.3, 2.2); P=0.95), liver transaminases (P>0.05) and liver fat (1.4% (−13.0, 15.9); P=0.83) were not significantly different from baseline. At 12 months following cessation of supervision, exercise-mediated improvements in liver fat and other cardiometabolic variables had reversed with cardiorespiratory fitness at baseline levels. Maintenance of high cardiorespiratory fitness and stability of body weight are critical public health considerations for the treatment of NAFLD (Clinicaltrials.gov identifier: NCT01834300).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Adams LA, Lymp JF, St Sauver J, Sanderson SO, Lindor KD, Feldstein A et al. The natural history of nonalcoholic fatty liver disease: a population-based cohort study. Gastroenterology 2005; 129: 113–121.
Targher G, Day CP, Bonora E . Risk of cardiovascular disease in patients with nonalcoholic fatty liver disease. N Engl J Med 2010; 363: 1341–1350.
Harrison SA, Day CP . Benefits of lifestyle modification in NAFLD. Gut 2007; 56: 1760–1769.
Pugh CJ, Spring VS, Kemp GJ, Richardson P, Shojaee-Moradie F, Umpleby AM et al. Exercise training reverses endothelial dysfunction in nonalcoholic fatty liver disease. Am J Physiol Heart Circ Physiol 2014; 307: H1298–H1306.
Cuthbertson DJ, Shojaee-Moradie F, Sprung VS, Jones H, Pugh CJ, Richardson P et al. Dissociation between exercise-induced reduction in liver fat and changes in hepatic and peripheral glucose homeostasis in obese patients with non-alcoholic fatty liver disease. Clin Sci (Lond) 2015; 130: 93–104.
Hallsworth K, Thoma C, Hollingsworth KG, Cassidy S, Anstee QM, Day CP et al. Modified high-intensity interval training reduces liver fat and improves cardiac function in non-alcoholic fatty liver disease: a randomised controlled trial. Clin Sci (Lond) 2015; 129: 1097–1105.
Sprung VS, Cuthbertson DJ, Pugh CJ, Aziz N, Kemp GJ, Daousi C et al. Exercise training in polycystic ovarian syndrome enhances flow-mediated dilation in the absence of changes in fatness. Med Sci Sports Exerc 2013; 45: 2234–2242.
Thomas EL, Hamilton G, Patel N, O'Dwyer R, Dore CJ, Goldin RD et al. Hepatic triglyceride content and its relation to body adiposity: a magnetic resonance imaging and proton magnetic resonance spectroscopy study. Gut 2005; 54: 122–127.
Woodman RJ, Playford DA, Watts GF, Cheetham C, Reed C, Taylor RR et al. Improved analysis of brachial artery ultrasound using a novel edge-detection software system. J Appl Physiol 2001; 91: 929–937.
Lesser IA, Dick T, Gasevic D, Mackey DC, Leipsic JA, Lear SA . The association between physical activity and liver fat after five years of follow-up in a primary prevention multi-ethnic cohort. Prev Med 2014; 67: 199–203.
Keating SE, Hackett DA, Parker HM, O'Connor HT, Gerofi JA, Sainsbury A et al. Effect of aerobic exercise training dose on liver fat and visceral adiposity. J Hepatol 2015; 63: 174–182.
Inaba Y, Chen JA, Bergmann SR . Prediction of future cardiovascular outcomes by flow-mediated vasodilatation of brachial artery: a meta-analysis. Int J Cardiovasc Imaging 2010; 26: 631–640.
Haufe S, Haas V, Utz W, Birkenfeld AL, Jeran S, Böhnke J et al. Long-lasting improvements in liver fat and metabolism despite body weight regain after dietary weight loss. Diabetes Care 2013; 36: 3786–3792.
Ong JP, Pitts A, Younossi ZM . Increased overall mortality and liver-related mortality in non-alcoholic fatty liver disease. J Hepatol 2008; 49: 608–612.
Haus JM, Solomon TP, Kelly KR, Fealy CE, Kullman EL, Scelsi AR et al. Improved hepatic lipid composition following short-term exercise in nonalcoholic fatty liver disease. J Clin Endocrinol Metab 2013; 98: E1181–E1188.
Acknowledgements
We thank all patients for their participation in the study. This study was funded by the European Federation for the Study of Diabetes (EFSD).
Author contributions
GJK, PR, AMU, DJG, NTC, HJ and DJC: conception and design of research; CJAP, VSS, FS-M, HJ and DJC performed experiments; CJAP, VSS, GJK, FS-M, AMU, DJG, NTC, HJ and DJC analysed data; CJAP, VSS, GJK, PR, FS-M, AMU, DJG, NTC, HJ and DJC interpreted results of experiments; CJAP, VSS, PR, HJ and DJC prepared figures; CJAP, HJ and DJC drafted manuscript; CJAP, VS.S, GJK, PR, FS-M, AMU, DJG, NTC, HJ and DJC edited and revised manuscript; CJAP, VSS, GJK, PR, FS-M, AMU, DJG, NTC, HJ and DJC approved final version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Pugh, C., Sprung, V., Jones, H. et al. Exercise-induced improvements in liver fat and endothelial function are not sustained 12 months following cessation of exercise supervision in nonalcoholic fatty liver disease. Int J Obes 40, 1927–1930 (2016). https://doi.org/10.1038/ijo.2016.123
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2016.123
This article is cited by
-
Lifestyle intervention for metabolic dysfunction-associated fatty liver disease: a 24-h integrated behavior perspective
Hepatology International (2024)
-
Relation Between Endothelial Dysfunction and Exercise Training-Mediated Adaptation in Cardiovascular Risk Factors, Cardiorespiratory Fitness, and Vascular Health in Humans: A secondary analysis
Journal of Science in Sport and Exercise (2024)
-
High-Intensity Interval Training is Safe, Feasible and Efficacious in Nonalcoholic Steatohepatitis: A Randomized Controlled Trial
Digestive Diseases and Sciences (2023)
-
Exercise in the Management of Metabolic-Associated Fatty Liver Disease (MAFLD) in Adults: A Position Statement from Exercise and Sport Science Australia
Sports Medicine (2023)
-
The efficacy of flaxseed and hesperidin on non-alcoholic fatty liver disease: an open-labeled randomized controlled trial
European Journal of Clinical Nutrition (2021)