Abstract
Background:
Both the glucocorticoid receptor (GR) and the mineralocorticoid receptor (MR) are expressed in adipose tissue and assumed to mediate cortisol actions on adipose tissue. The relative significance of the two receptors in mediating glucocorticoid regulation of adipogenesis and adipokine expression in human adipocytes has not been addressed.
Methods:
We investigated the differential roles of the GR and MR in mediating glucocorticoid actions on adipogenesis and adipokine production using RNA interference in primary cultures of human preadipocytes and adipocytes.
Results:
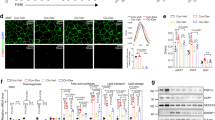
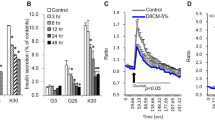
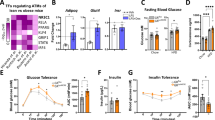
Both types of receptors are expressed, but levels of GR were several hundred fold higher than MR in both human preadipocytes and adipocytes. As expected, cortisol added during adipogenesis increased the differentiation of human preadipocytes. Silencing of GR, but not MR, blocked these proadipogenic actions of cortisol. In differentiated human adipocytes, addition of cortisol increased leptin and adiponectin, while suppressing interleukin-6 (IL-6), messenger RNA levels and protein secretion. Knockdown of GR by 65% decreased leptin and adiponectin while increasing IL-6 production. In addition, GR silencing blocked the effects of cortisol on adipokine expression. In contrast, although MR knockdown increased leptin, it did not affect adiponectin and IL-6 expression.
Conclusion:
Our data demonstrate that although both GR and MR have roles in regulating leptin expression, GR plays more important roles in mediating the actions of cortisol to regulate adipogenesis and adipokine production in human adipocytes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Peckett AJ, Wright DC, Riddell MC . The effects of glucocorticoids on adipose tissue lipid metabolism. Metabolism 2011; 60: 1500–1510.
Lee MJ, Gong DW, Burkey BF, Fried SK . Pathways regulated by glucocorticoids in omental and subcutaneous human adipose tissues: a microarray study. Am J Physiol Endocrinol Metab 2011; 300: E571–E580.
Lee MJ, Fried SK . Glucocorticoids antagonize tumor necrosis factor-alpha-stimulated lipolysis and resistance to the antilipolytic effect of insulin in human adipocytes. Am J Physiol Endocrinol Metab 2012; 303: E1126–E1133.
Gathercole LL, Morgan SA, Bujalska IJ, Hauton D, Stewart PM, Tomlinson JW . Regulation of lipogenesis by glucocorticoids and insulin in human adipose tissue. PLoS One 2011; 6: e26223.
Lee MJ, Pramyothin P, Karastergiou K, Fried SK . Deconstructing the roles of glucocorticoids in adipose tissue biology and the development of central obesity. Biochim Biophys Acta 2014; 1842: 473–481
Fried SK, Bunkin DA, Greenberg AS . Omental and subcutaneous adipose tissues of obese subjects release interleukin-6: depot difference and regulation by glucocorticoid. J Clin Endocrinol Metab 1998; 83: 847–850.
Bruun JM, Lihn AS, Madan AK, Pedersen SB, Schiott KM, Fain JN et al. Higher production of IL-8 in visceral vs. subcutaneous adipose tissue. Implication of nonadipose cells in adipose tissue. Am J Physiol Endocrinol Metab 2004; 286: E8–E13.
Hirata A, Maeda N, Nakatsuji H, Hiuge-Shimizu A, Okada T, Funahashi T et al. Contribution of glucocorticoid-mineralocorticoid receptor pathway on the obesity-related adipocyte dysfunction. Biochem Biophys Res Commun 2012; 419: 182–187.
Caprio M, Feve B, Claes A, Viengchareun S, Lombes M, Zennaro MC . Pivotal role of the mineralocorticoid receptor in corticosteroid-induced adipogenesis. FASEB J 2007; 21: 2185–2194.
Hoppmann J, Perwitz N, Meier B, Fasshauer M, Hadaschik D, Lehnert H et al. The balance between gluco- and mineralo-corticoid action critically determines inflammatory adipocyte responses. J Endocrinol 2010; 204: 153–164.
Hauner H, Schmid P, Pfeiffer EF . Glucocorticoids and insulin promote the differentiation of human adipocyte precursor cells into fat cells. J Clin Endocrinol Metab 1987; 64: 832–835.
Hauner H, Entenmann G, Wabitsch M, Gaillard D, Ailhaud G, Negrel R et al. Promoting effect of glucocorticoids on the differentiation of human adipocyte precursor cells cultured in a chemically defined medium. J Clin Invest 1989; 84: 1663–1670.
Lee MJ, Wu Y, Fried SK . A modified protocol to maximize differentiation of human preadipocytes and improve metabolic phenotypes. Obesity (Silver Spring) 2012; 20: 2334–2340.
Hauner H, Skurk T, Wabitsch M . Cultures of human adipose precursor cells. Methods Mol Biol 2001; 155: 239–247.
Wang JC, Derynck MK, Nonaka DF, Khodabakhsh DB, Haqq C, Yamamoto KR . Chromatin immunoprecipitation (ChIP) scanning identifies primary glucocorticoid receptor target genes. Proc Natl Acad Sci USA 2004; 101: 15603–15608.
Caprio M, Antelmi A, Chetrite G, Muscat A, Mammi C, Marzolla V et al. Antiadipogenic effects of the mineralocorticoid receptor antagonist drospirenone: potential implications for the treatment of metabolic syndrome. Endocrinology 2011; 152: 113–125.
Pantoja C, Huff JT, Yamamoto KR . Glucocorticoid signaling defines a novel commitment state during adipogenesis in vitro. Mol Biol Cell 2008; 19: 4032–4041.
Steger DJ, Grant GR, Schupp M, Tomaru T, Lefterova MI, Schug J et al. Propagation of adipogenic signals through an epigenomic transition state. Genes Dev 2010; 24: 1035–1044.
Yu G, Floyd ZE, Wu X, Hebert T, Halvorsen YD, Buehrer BM et al. Adipogenic differentiation of adipose-derived stem cells. Methods Mol Biol 2011; 702: 193–200.
Tomlinson JJ, Boudreau A, Wu D, Atlas E, Hache RJ . Modulation of early human preadipocyte differentiation by glucocorticoids. Endocrinology 2006; 147: 5284–5293.
Pairault J, Green H . A study of the adipose conversion of suspended 3T3 cells by using glycerophosphate dehydrogenase as differentiation marker. Proc Natl Acad Sci USA 1979; 76: 5138–5142.
Kuri-Harcuch W, Marsch-Moreno M . DNA synthesis and cell division related to adipose differentiation of 3T3 cells. J Cell Physiol 1983; 114: 39–44.
Schmidt W, Poll-Jordan G, Loffler G . Adipose conversion of 3T3-L1 cells in a serum-free culture system depends on epidermal growth factor, insulin-like growth factor I, corticosterone, and cyclic AMP. J Biol Chem 1990; 265: 15489–15495.
Entenmann G, Hauner H . Relationship between replication and differentiation in cultured human adipocyte precursor cells. Am J Physiol 1996; 270: C1011–C1016.
Engeli S, Bohnke J, Feldpausch M, Gorzelniak K, Heintze U, Janke J et al. Regulation of 11beta-HSD genes in human adipose tissue: influence of central obesity and weight loss. Obes Res 2004; 12: 9–17.
Veilleux A, Laberge PY, Morency J, Noel S, Luu-The V, Tchernof A . Expression of genes related to glucocorticoid action in human subcutaneous and omental adipose tissue. J Steroid Biochem Mol Biol 2010; 122: 28–34.
Lee MJ, Fried SK, Mundt SS, Wang Y, Sullivan S, Stefanni A et al. Depot-specific regulation of the conversion of cortisone to cortisol in human adipose tissue. Obesity (Silver Spring) 2008; 16: 1178–1185.
Guo C, Ricchiuti V, Lian BQ, Yao TM, Coutinho P, Romero JR et al. Mineralocorticoid receptor blockade reverses obesity-related changes in expression of adiponectin, peroxisome proliferator-activated receptor-gamma, and proinflammatory adipokines. Circulation 2008; 117: 2253–2261.
Hirata A, Maeda N, Hiuge A, Hibuse T, Fujita K, Okada T et al. Blockade of mineralocorticoid receptor reverses adipocyte dysfunction and insulin resistance in obese mice. Cardiovasc Res 2009; 84: 164–172.
Patsouris D, Neels JG, Fan W, Li PP, Nguyen MT, Olefsky JM . Glucocorticoids and thiazolidinediones interfere with adipocyte-mediated macrophage chemotaxis and recruitment. J Biol Chem 2009; 284: 31223–31235.
Kraus D, Jager J, Meier B, Fasshauer M, Klein J . Aldosterone inhibits uncoupling protein-1, induces insulin resistance, and stimulates proinflammatory adipokines in adipocytes. Horm Metab Res 2005; 37: 455–459.
Li P, Zhang XN, Pan CM, Sun F, Zhu DL, Song HD et al. Aldosterone perturbs adiponectin and PAI-1 expression and secretion in 3T3-L1 adipocytes. Horm Metab Res 2011; 43: 464–469.
Acknowledgements
This work was supported by NIH DK080448 and P30 DK046200 (to SKF). We thank Dr Monica Miller for assistance with adipose tissue sampling during surgery.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Lee, MJ., Fried, S. The glucocorticoid receptor, not the mineralocorticoid receptor, plays the dominant role in adipogenesis and adipokine production in human adipocytes. Int J Obes 38, 1228–1233 (2014). https://doi.org/10.1038/ijo.2014.6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2014.6
Keywords
This article is cited by
-
Perceived stress and diet quality in women of reproductive age: a systematic review and meta-analysis
Nutrition Journal (2020)
-
Influence of race on the effect of premature birth on salivary cortisol response to stress in adolescents
Pediatric Research (2020)
-
Early weaning leads to specific glucocorticoid signalling in fat depots of adult rats
Endocrine (2020)
-
Association of intracellular lipid accumulation in subcutaneous adipocyte precursors and plasma adipokines in bariatric surgery candidates
Lipids in Health and Disease (2019)
-
Metabolism-related microRNAs in maternal breast milk are influenced by premature delivery
Pediatric Research (2017)