Abstract
Objective:
The Medifast 5 & 1 Plan (MD) is a portion-controlled, nutritionally-balanced, low-fat weight-loss plan. We studied the effects of MD compared with a reduced-energy, food-based diet (FB) on body weight, waist circumference, fat mass and other measures in adults.
Design:
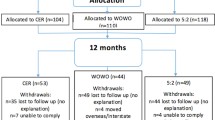
We conducted a two-parallel-arm, randomized, controlled trial comparing MD to FB over 52 weeks. A total of 120 men and women aged 19–65 years with BMI ⩾35 and ⩽50 kg m−2 were randomized to MD (n=60) or FB (n=60). Follow-up included a 26-week weight-loss phase and 26-week weight-maintenance phase. Anthropometric, body composition, biochemical and appetite/satiety measures were performed at baseline and at 26 and 52 weeks. An intention-to-treat, linear mixed models analysis was the primary analysis.
Results:
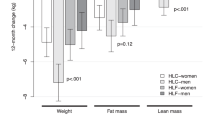
Fifty MD subjects (83.3%) and 45 FB subjects (75.0%) completed the study on assigned treatment. At 26 weeks, race-adjusted mean weight loss was 7.5 kg in MD subjects vs 3.8 kg in FB subjects (P=0.0002 for difference); reduction in waist circumference was 5.7 cm in MD vs 3.7 cm in FB (P=0.0064); and fat mass loss was 6.4 kg in MD vs 3.7 kg in FB (P=0.0011). At 52 weeks, the corresponding reductions were 4.7 vs 1.9 kg (P=0.0004); 5.0 vs 3.6 cm (P=0.0082); and 4.1 vs 1.9 kg (P=0.0019) in MD and FB subjects, respectively.
Conclusion:
In obese adults, MD resulted in significantly greater reductions in body weight and fat compared with an FB diet for 1 year after randomization.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Flegal KM, Carroll MD, Kit BK, Ogden CL . Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999-2010. JAMA 2012; 307: 491–497.
Ogden CL, Carroll MD, Kit BK, Flegal KM . Prevalence of obesity in the United States, 2009-2010. Centers for Disease Control and Prevention, Washington, DC, Department of Health and Human Services. c2012—(cited June 5 2012). NCHS Data Brief No. 82, January 2012. Available from http://www.cdc.gov/nchs/data/databriefs/db82.pdf.
Fontaine KR, Redden DT, Wang C, Westfall AO, Allison DB . Years of life lost due to obesity. JAMA 2003; 289: 187–193.
Klein S, Burke LE, Bray GA, Blair S, Allison DB, Pi-Sunyer X et alAmerican Heart Association Council on Nutrition, Physical Activity, and Metabolism. Clinical implications of obesity with specific focus on cardiovascular disease: a statement for professionals from the American Heart Association Council on Nutrition, Physical Activity, and Metabolism: endorsed by the American College of Cardiology Foundation. Circulation 2004; 110: 2952–2967.
Astrup A . Macronutrient balances and obesity: the role of diet and physical activity. Public Health Nutr 1999; 2: 341–347.
Green SM . Obesity: prevalence, causes, health risks and treatment. Br J Nurs 1997; 6: 1181–1185.
Keith SW, Redden DT, Katzmarzyk PT, Boggiano MM, Hanlon EC, Benca RM et al. Putative contributors to the secular increase in obesity: exploring the roads less traveled. Int J Obes (Lond) 2006; 30: 1585–1594.
Bray GA . Lifestyle and pharmacological approaches to weight loss: efficacy and safety. J Clin Endocrinol Metab 2008; 93: S81–S88.
Dansinger ML, Tatsioni A, Wong JB, Chung M, Balk EM . Meta-analysis: the effect of dietary counseling for weight loss. Ann Intern Med 2007; 147: 41–50.
Davis LM, Coleman C, Kiel J, Rampolla J, Hutchisen T, Ford L et al. Efficacy of a meal replacement diet plan compared to a food-based diet plan after a period of weight loss and weight maintenance: a randomized controlled trial. Nutr J 2010; 9: 11.
Young LR, Nestle M . Expanding portion sizes in the US marketplace: implications for nutrition counseling. J Am Diet Assoc 2003; 103: 231–234.
Derogatis LR, Melisaratos N . The Brief Symptom Inventory: an introductory report. Psychol Med. 1983; 13: 595–605.
Garner DM, Garfinkel PE . The Eating Attitudes Test: an index of the symptoms of anorexia nervosa. Psychol Med 1979; 9: 273–279.
Flint A, Raben A, Blundell JE, Astrup A . Reproducibility, power and validity of visual analogue scales in assessment of appetite sensations in single test meal studies. Int J Obes 2000; 24: 38–48.
Kenward MG, Roger JH . Small sample inference for fixed effects from restricted maximum likelihood. Biometrics 1997; 53: 983–997.
Fitzgibbon ML, Tussing-Humphreys LM, Porter JS, Martin IK, Odoms-Young A, Sharp LK . Weight loss and African-American women: a systematic review of the behavioural weight loss intervention literature. Obes Rev 2012; 13: 193–213.
Schulz KF, Altman DG, Moher D, The CONSORT Group. CONSORT 2010 statement: updated guidelines for reporting parallel group randomized trials. Ann Int Med 2010; 152: 726–732.
Cheskin LJ, Mitchell AM, Jhaveri AD, Mitola AH, Davis LM, Lewis RA et al. Efficacy of meal replacements versus a standard food-based diet for weight loss in type 2 diabetes. Diabetes Educ 2008; 34: 118–127.
Ditschuneit HH, Flechtner-Mors M . Value of structured meals for weight management: risk factors and long-term weight maintenance. Obes Res 2001; 9: 284S–289S.
Heymsfield SB, van Mierlo CAJ, Knaap van der HCM, Heo M, Frier HI . Weight management using a meal replacement strategy: meta and pooling analysis from six studies. Int J Obes Relat Metab Disord 2003; 27: 537–549.
Metzner CE, Folberth-Vogele A, Bitterlich N, Lemperle M, Schafer S, Alteheld B et al. Effect of a conventional energy-restricted modified diet with or without meal replacement on weight loss and cardiometabolic risk profile in overweight women. Nutr Metab 2011; 8: 64.
Noakes M, Foster PR, Keogh JB, Clifton PM . Meal replacements are as effective as structured weight-loss diets for treating obesity in adults with features of metabolic syndrome. J Nutr 2004; 134: 1894–1899.
Rothacker DQ, Staniszewski BA, Ellis PK . Liquid meal replacement vs traditional food: a potential model for women who cannot maintain eating habit change. J Am Diet Assoc 2001; 101: 345–347.
Ashley JM, St Jeor ST, Schrage JP, Perumean-Chaney SE, Gilbertson MC, McCall NL et al. Weight control in the physician’s office. Arch Intern Med 2001; 161: 1599–1604.
Ditschuneit HH, Flechtner-Mors M, Johnson TD, Adler G . Metabolic and weight-loss effects of a long-term dietary intervention in obese patients. Am J Clin Nutr 1999; 69: 198–204.
Leenen R, van der Kooy K, Mayboom S, Seidell JC, Deurenberg P, Weststrate JA . Relative effects of weight loss and dietary fat modification on serum lipid levels in the dietary treatment of obesity. J Lipid Res 1993; 34: 2183–2191.
Yip I, Go VLW, DeShields S, Saltsman P, Bellman M, Thames G et al. Liquid meal replacements and glycemic control in obese type 2 diabetes patients. Obes Res 2001; 9: 341S–347S.
Acknowledgements
The authors acknowledge Medifast, Inc., Owings Mills, MD, for their support of the study. We acknowledge the Metabolism/Human Physiology Core Laboratory of the Nutrition Obesity Research Center, Diabetes Research and Training Center, and Center for Clinical and Translational Science at UAB (grant nos. UL1RR025777, P60DK079626, and P30DK56336, respectively) and those who performed the glucose and hs-CRP assays: Maryellen Williams and Cindy Zeng. We also acknowledge the Bioanalytical Redox Biology Core of the Diabetes Research and Training Center at UAB and those who performed the LPO assay: Gin Chuang, Kelley Johnston, and Douglas R. Moellering, PhD. Finally, we thank Kathryn Kaiser, PhD, for her critical review of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Dr Allison has, anticipates, or has had financial interests with the following: Paul, Weiss, Rifkin, Wharton & Garrison, LLP; Kraft Foods, Inc., and Nutrition Impact, LLC and has received consulting fees from Medifast. The remaining authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
Shikany, J., Thomas, A., Beasley, T. et al. Randomized controlled trial of the Medifast 5 & 1 Plan for weight loss. Int J Obes 37, 1571–1578 (2013). https://doi.org/10.1038/ijo.2013.43
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2013.43
Keywords
This article is cited by
-
Commercial Weight Loss Programs in the Management of Obesity: an Update
Current Obesity Reports (2021)
-
Comparative effectiveness of a portion-controlled meal replacement program for weight loss in adults with and without diabetes/high blood sugar
Nutrition & Diabetes (2017)
-
Predictors of weight loss in young adults who are over-weight or obese and have psychosocial problems: a post hoc analysis
BMC Family Practice (2016)
-
Effectiveness of a Medifast meal replacement program on weight, body composition and cardiometabolic risk factors in overweight and obese adults: a multicenter systematic retrospective chart review study
Nutrition Journal (2015)