Abstract
Context:
No long-term studies have compared centrally acting drugs for treating obesity.
Objective:
To compare the efficacy and safety of diethylpropion (DEP), fenproporex (FEN), mazindol (MZD), fluoxetine (FXT) and sibutramine (SIB) in promoting weight loss.
Design and Setting:
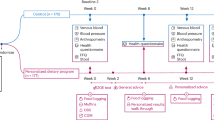
A prospective, randomized, placebo (PCB)-controlled study conducted at a single academic institution.
Patients:
A total of 174 obese premenopausal women.
Intervention:
Participants randomly received DEP 75 mg (n=28), FEN 25 mg (n=29), MZD 2 mg (n=29), SIB 15 mg (n=30), FXT 20 mg (n=29) or PCB (n=29) daily over 52 weeks. Diet and physical activity were encouraged.
Main Outcome Measures:
The primary endpoints were changes in body weight and the proportion of women who achieved at least 5% weight loss by week 52 in the intent-to-treat population. Other measurements included anthropometry, safety, metabolic and cardiovascular parameters.
Results:
Weight loss was greater than PCB (−3.1±4.3 kg) with DEP (−10.0±6.4 kg; P<0.001), SIB (−9.5±5.9 kg; P<0.001), FEN (−7.8±6.9 kg; P<0.01) and MZD (−7.4±4.9 kg; P<0.01) but not with FXT (−2.5±4.1 kg). Ten (33.3%) women lost⩾5% of their initial weight with PCB, compared with 20 (71.4%; P<0.001) with DEP, 20 (69%; P<0.02) with FEN, 21 (72.4%; P<0.01) with MZD, 22 (73.3%; P<0.001) with SIB and 10 (35.5%) with FXT. Each medically treated group experienced more adverse events compared with PCB (P<0.001). Compared with PCB, constipation was more prevalent with DEP, SIB and MZD (P<0.01); anxiety was more prevalent with DEP (P=0.01); and irritability occurred more frequently with DEP and FEN (P=0.02). Significant improvements in the depression and anxiety scores, binge-eating episodes and quality of life correlated with weight loss.
CONCLUSION:
The centrally acting drugs DEP, FEN, MZD and SIB were more effective than PCB in promoting weight loss in obese premenopausal women, with a satisfactory benefit–risk profile.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bray GA . Medical consequences of obesity. J Clin Endocrinol Metab 2004; 89: 2583–2589.
Berrington de, Gonzalez A, Hartge P, Cerhan JR, Flint AJ, Hannan L et al. Body-mass index and mortality among 1.46 million white adults. N Engl J Med 2010; 363: 2211–2219.
Unick JL, Beavers D, Jakicic JM, Kitabchi AE, Knowler WC, Wadden TA et al. Effectiveness of lifestyle interventions for individuals with severe obesity and type 2 diabetes: results from the Look AHEAD trial. Diabetes Care 2011; 34: 2152–2157.
Sjöström L . Review of the key results from the Swedish Obese Subjects (SOS) trial: a prospective controlled intervention study of bariatric surgery. J Intern Med 2013; 273: 219–234.
Caterson ID, Finer N, Coutinho W, Van Gaal LF, Maggioni AP, Torp-Pedersen C et al. Maintained intentional weight loss reduces cardiovascular outcomes: results from the Sibutramine Cardiovascular OUTcomes (SCOUT) trial. Diabetes Obes Metab 2012; 14: 523–530.
Sumithran P, Prendergast LA, Delbridge E, Purcell K, Shulkes A, Kriketos A et al. Long-term persistence of hormonal adaptations to weight loss. N Engl J Med 2011; 365: 1597–1604.
Haddock CK, Poston WS, Dill PL, Foreyt JP, Ericsson M . Pharmacotherapy for obesity: a quantitative analysis of four decades of published randomized clinical trials. Int J Obes Relat Metab Disord 2002; 26: 262–273.
Li Z, Maglione M, Tu W, Mojica W, Arterburn D, Shugarman LR et al. Meta-analysis: pharmacologic treatment of obesity. Ann Intern Med 2005; 142: 532–546.
Wilding JP . Treatment strategies for obesity. Obes Rev 2007; 8 (Suppl 1): 137–144.
Bray GA . Lifestyle and pharmacological approaches to weight loss: efficacy and safety. J Clin Endocrinol Metab 2008; 93 (11 Suppl 1): S81–S88.
Colman E . Anorectics on trial: a half century of federal regulation of prescription appetite suppressants. Ann Intern Med 2005; 143: 380–385.
Borsini F, Bendotti C, Carli M, Poggesi E, Samanin R . The roles of brain noradrenaline and dopamine in the anorectic activity of diethylpropion in rats: a comparison with d-amphetamine. Res Commun Chem Pathol Pharmacol 1979; 26: 3–11.
Santamaría A, Arias HR . Neurochemical and behavioral effects elicited by bupropion and diethylpropion in rats. Behav Brain Res 2010; 211: 132–139.
Cercato C, Roizenblatt VA, Leança CC, Segal A, Lopes Filho AP et al. A randomized double-blind placebo-controlled study of the long-term efficacy and safety of diethylpropion in the treatment of obese subjects. Int J Obes (Lond) 2009; 33: 857–865.
Cody JT, Valtier S, Stillman S . Amphetamine and fenproporex levels following multidose administration of fenproporex. J Anal Toxicol 1999; 23: 187–194.
Halpern A, Mancini MC . Treatment of obesity: an update on anti-obesity medications. Obes Rev 2003; 4: 25–42.
Inoue S . Clinical studies with mazindol. Obes Res 1995; 3 (Suppl 4): 549S–552S.
Wellman PJ . Systemic mazindol reduces food intake in rats via suppression of meal size and meal number. J Psychopharmacol 2008; 22: 532–535.
Araújo JR, Martel F . Sibutramine effects on central mechanisms regulating energy homeostasis. Curr Neuropharmacol 2012; 10: 49–52.
Bray GA, Blackburn GL, Ferguson JM, Greenway FL, Jain AK, Mendel CM et al. Sibutramine produces dose-related weight loss. Obes Res 1999; 7: 189–198.
Aigner M, Treasure J, Kaye W, Kasper S, WFSBP Task Force on Eating Disorders. World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the pharmacological treatment of eating disorders. World J Biol Psychiatry 2011; 12: 400–443.
Goldstein DJ, Rampey AH Jr, Enas GG, Potvin JH, Fludzinski LA, Levine LR . Fluoxetine: a randomized clinical trial in the treatment of obesity. Int J Obes Relat Metab Disord 1994; 18: 129–135.
Serretti A, Mandelli L . Antidepressants and body weight: a comprehensive review and meta-analysis. J Clin Psychiatry 2010; 71: 1259–1272.
James WP, Caterson ID, Coutinho W, Finer N, Van Gaal LF, Maggioni AP et al. Effect of sibutramine on cardiovascular outcomes in overweight and obese subjects. N Engl J Med 2010; 363: 905–917.
Beck AT, Steer RA, Ball R, Ranieri W . Comparison of Beck Depression Inventories-IA and -II in psychiatric outpatients. J Pers Assess 1996; 67: 588–597.
Beck AT, Epstein N, Brown G, Steer RA . An inventory for measuring clinical anxiety: psychometric properties. J Consult Clin Psychol 1988; 56: 893–897.
Freitas S, Lopes CS, Coutinho W, Appolinario JC . Translation and adaptation into Portuguese of the Binge-Eating Scale. Rev Bras Psiquiatr 2001; 23: 215–220.
Ware JE Jr, Sherbourne CD . The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care 1992; 30: 473–483.
WHOQOL Group. The World Health Organization Quality of Life assessment (WHOQOL): position paper from the World Health Organization. Soc Sci Med 1995; 41: 1403–1409.
Kolotkin RL, Head S, Hamilton M, Tse CK . Assessing impact of weight on quality of life. Obes Res 1995; 3: 49–56.
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC . Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985; 28: 412–419.
Lachin JM . Statistical considerations in the intent-to-treat principle. Control Clin Trials 2000; 21: 167–189.
Coutinho W . The first decade of sibutramine and orlistat: a reappraisal of their expanding roles in the treatment of obesity and associated conditions. Arq Bras Endocrinol Metabol 2009; 53: 262–270.
Gadde KM, Allison DB, Ryan DH, Peterson CA, Troupin B, Schwiers ML et al. Effects of low-dose, controlled-release, phentermine plus topiramate combination on weight and associated comorbidities in overweight and obese adults (CONQUER): a randomised, placebo-controlled, phase 3 trial. Lancet 2011; 377: 1341–1352.
Gray DS, Fujioka K, Devine W, Bray GA . A randomized double-blind clinical trial of fluoxetine in obese diabetics. Int J Obes Relat Metab Disord 1992; 16 (Suppl 4): S67–S72.
Wise SD . Clinical studies with fluoxetine in obesity. Am J Clin Nutr 1992; 55 (Suppl 1): 181S–184S.
Smith SR, Weissman NJ, Anderson CM, Sanchez M, Chuang E, Stubbe S et al. Multicenter, placebo-controlled trial of lorcaserin for weight management. N Engl J Med 2010; 363: 245–256.
Greenway FL, Fujioka K, Plodkowski RA, Mudaliar S, Guttadauria M, Erickson J et al. Effect of naltrexone plus bupropion on weight loss in overweight and obese adults (COR-I): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2010; 376: 595–605.
Kim SH, Lee YM, Jee SH, Nam CM . Effect of sibutramine on weight loss and blood pressure: a meta-analysis of controlled trials. Obes Res 2003; 11: 1116–1123.
Ioannides-Demos LL, Piccenna L, McNeil JJ . Pharmacotherapies for obesity: past, current, and future therapies. J Obes 2011; 2011: 179674.
Acknowledgements
SIB and FXT were generously provided by Medley Pharmaceuticals. FEN and DEP were generously provided by Aché Pharmaceuticals. MZD and PCB were prepared by Apparenza Drugstore, which was also responsible for packing all medications in identical bottles.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Suplicy, H., Boguszewski, C., dos Santos, C. et al. A comparative study of five centrally acting drugs on the pharmacological treatment of obesity. Int J Obes 38, 1097–1103 (2014). https://doi.org/10.1038/ijo.2013.225
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2013.225
Keywords
This article is cited by
-
Depression and obesity among females, are sex specificities considered?
Archives of Women's Mental Health (2021)
-
Counterfeit formulations: analytical perspective on anorectics
Forensic Toxicology (2021)
-
Personality type, eating behaviour and suicide risk in women in treatment for obesity
Eating and Weight Disorders - Studies on Anorexia, Bulimia and Obesity (2021)
-
Current and investigational anti-obesity drugs help reduce weight and offer additional benefits, but more effective options are needed
Drugs & Therapy Perspectives (2020)
-
Pharmacogenetics of amfepramone in healthy Mexican subjects reveals potential markers for tailoring pharmacotherapy of obesity: results of a randomised trial
Scientific Reports (2019)


