Abstract
Objective:
FSP27 KO mice showed enhanced expression of mitochondrial genes, increased mitochondrial activity and smaller lipid droplets. Here, we aimed to investigate lipid droplet protein (CIDEC/FSP27 and perilipinA (PLIN1)) gene expression in human adipose tissue in association with obesity, insulin resistance and mitochondrial gene expression.
Design and subjects:
In cohort 1, CIDEC/FSP27, PLIN1, adipogenic (FASN, ACACA, PPARG, GLUT4) and mitochondrial (PPARGC1A, PPARGC1B, TFAM, MT-CO3) gene expression were analyzed in 171 adipose tissue samples (88 visceral adipose tissue (VAT) and 83 subcutaneous adipose tissue (SAT) depots) and in a time course experiment in human subcutaneous and visceral preadipocytes using real-time PCR. In cohort 2, the effects of bariatric surgery-induced weight loss were also evaluated in six caucasian morbidly obese women. Additionally, in cohort 2 FSP27 and PLIN1 protein levels were measured using western blotting.
Results:
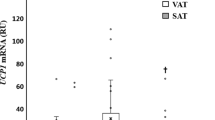
CIDEC/FSP27 (1.03±0.52 vs 0.49±0.23 relative gene expression unit (R.U.), P<0.0001) and PLIN1 (1.32±0.82 vs 0.63±0.42 R.U., P<0.0001) gene were significantly more expressed in SAT than in VAT. In VAT, CIDEC/FSP27 and PLIN1 gene expression decreased with body mass index, percent fat mass, fasting glucose, fasting insulin, HOMA and were positively associated with adipogenic (PPARG, GLUT4, FASN and ACACA) and mitochondrial biogenesis (PPARGC1A, PPARGC1B, TFAM and MT-CO3)-related genes. Mitochondrial gene expression increased during adipocyte differentiation in parallel to FSP27 and PLIN1 and other adipogenic genes. After bariatric surgery-induced weight loss, PLIN1 and CIDEC/FSP27 gene and protein expression in SAT increased significantly in parallel to adipogenic and mitochondrial genes.
Conclusion:
These findings suggest a positive functional interaction between CIDEC/FSP27, PLIN1 and mitochondrial biogenesis-related genes in human adipose tissue.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Rajala MW, Scherer PE . Minireview: the adipocyte—at the crossroads of energy homeostasis, inflammation, and atherosclerosis. Endocrinology 2003; 144: 3765–3773.
Wolins NE, Brasaemle DL, Bickel PE . A proposed model of fat packaging by exchangeable lipid droplet proteins. FEBS Lett 2006; 580: 5484–5491.
Puri V, Konda S, Ranjit S, Aouadi M, Chawla A, Chouinard M et al. Fat-specific protein 27, a novel lipid droplet protein that enhances triglyceride storage. J Biol Chem 2007; 282: 34213–34218.
Puri V, Virbasius JV, Guilherme A, Czech MP . RNAi screens reveal novel metabolic regulators: RIP140, MAP4k4 and the lipid droplet associated fat specific protein (FSP) 27. Acta Physiol (Oxford) 2008; 192: 103–115.
Toh SY, Gong J, Du G, Li JZ, Yang S, Ye J et al. Up-regulation of mitochondrial activity and acquirement of brown adipose tissue-like property in the white adipose tissue of Fsp27 deficient mice. PLoS One 2008; 3: e2890.
Sawada T, Miyoshi H, Shimada K, Suzuki A, Okamatsu-Ogura Y, Perfield JW 2nd et al. Perilipin overexpression in white adipose tissue induces a brown fat-like phenotype. PLoS One 2010; 5: e14006.
Puri V, Ranjit S, Konda S, Nicoloro SM, Straubhaar J, Chawla A et al. Cidea is associated with lipid droplets and insulin sensitivity in humans. Proc Natl Acad Sci USA 2008; 105: 7833–7838.
Rubio-Cabezas O, Puri V, Murano I, Saudek V, Semple RK, Dash S et al. Partial lipodystrophy and insulin resistant diabetes in a patient with a homozygous nonsense mutation in CIDEC. EMBO Mol Med 2009; 1: 280–287.
Kern PA, Di Gregorio G, Lu T, Rassouli N, Ranganathan G . Perilipin expression in human adipose tissue is elevated with obesity. J Clin Endocrinol Metab 2004; 89: 1352–1358.
Wang Y, Sullivan S, Trujillo M, Lee MJ, Schneider SH, Brolin RE et al. Perilipin expression in human adipose tissues: effects of severe obesity, gender, and depot. Obes Res 2003; 11: 930–936.
Ray H, Pinteur C, Frering V, Beylot M, Large V . Depot-specific differences in perilipin and hormone-sensitive lipase expression in lean and obese. Lipids Health Dis 2009; 8: 58.
Nielsen TS, Kampmann U, Nielsen RR, Jessen N, Orskov L, Pedersen SB et al. Reduced mRNA and protein expression of perilipin A and G0/G1 switch gene 2 (G0S2) in human adipose tissue in poorly controlled type 2 diabetes. J Clin Endocrinol Metab 2012; 97: E1348–E1352.
Gandotra S, Le Dour C, Bottomley W, Cervera P, Giral P, Reznik Y et al. Perilipin deficiency and autosomal dominant partial lipodystrophy. N Engl J Med 2011; 364: 740–748.
Gandotra S, Lim K, Girousse A, Saudek V, O'Rahilly S, Savage DB . Human frame shift mutations affecting the carboxyl terminus of perilipin increase lipolysis by failing to sequester the adipose triglyceride lipase (ATGL) coactivator AB-hydrolase-containing 5 (ABHD5). J Biol Chem 2011; 286: 34998–35006.
Guidone C, Manco M, Valera-Mora E, Iaconelli A, Gniuli D, Mari A et al. Mechanisms of recovery from type 2 diabetes after malabsorptive bariatric surgery. Diabetes 2006; 55: 2025–2031.
Ibrahim MM . Subcutaneous and visceral adipose tissue: structural and functional differences. Obes Rev 2010; 11: 11–18.
Hardy OT, Perugini RA, Nicoloro SM, Gallagher-Dorval K, Puri V, Straubhaar J et al. Body mass index-independent inflammation in omental adipose tissue associated with insulin resistance in morbid obesity. Surg Obes Relat Dis 2011; 7: 60–67.
O’Connell J, Lynch L, Cawood TJ, Kwasnik A, Nolan N, Geoghegan J et al. The relationship of omental and subcutaneous adipocyte size to metabolic disease in severe obesity. PLoS One 2010; 5: e9997.
Sadie-Van Gijsen H, Crowther NJ, Hough FS, Ferris WF . Depot-specific differences in the insulin response of adipose-derived stromal cells. Mol Cell Endocrinol 2010; 328: 22–27.
Virtanen KA, Lonnroth P, Parkkola R, Peltoniemi P, Asola M, Viljanen T et al. Glucose uptake and perfusion in subcutaneous and visceral adipose tissue during insulin stimulation in nonobese and obese humans. J Clin Endocrinol Metab 2002; 87: 3902–3910.
Nishino N, Tamori Y, Tateya S, Kawaguchi T, Shibakusa T, Mizunoya W et al. FSP27 contributes to efficient energy storage in murine white adipocytes by promoting the formation of unilocular lipid droplets. J Clin Invest 2008; 118: 2808–2821.
Matsusue K, Kusakabe T, Noguchi T, Takiguchi S, Suzuki T, Yamano S et al. Hepatic steatosis in leptin-deficient mice is promoted by the PPARgamma target gene Fsp27. Cell Metab 2008; 7: 302–311.
Shimizu M, Takeshita A, Tsukamoto T, Gonzalez FJ, Osumi T . Tissue-selective, bidirectional regulation of PEX11 alpha and perilipin genes through a common peroxisome proliferator response element. Mol Cell Biol 2004; 24: 1313–1323.
Wilson-Fritch L, Burkart A, Bell G, Mendelson K, Leszyk J, Nicoloro S et al. Mitochondrial biogenesis and remodeling during adipogenesis and in response to the insulin sensitizer rosiglitazone. Mol Cell Biol 2003; 23: 1085–1094.
Håkansson J, Eliasson B, Smith U, Enerbäck S . Adipocyte mitochondrial genes and the forkhead factor FOXC2 are decreased in type 2 diabetes patients and normalized in response to rosiglitazone. Diabetol Metab Syndr 2011; 3: 32.
Soronen J, Laurila PP, Naukkarinen J, Surakka I, Ripatti S, Jauhiainen M et al. Adipose tissue gene expression analysis reveals changes in inflammatory, mitochondrial respiratory and lipid metabolic pathways in obese insulin-resistant subjects. BMC Med Genomics 2012; 5: 9.
Girousse A, Langin D . Adipocyte lipases and lipid droplet-associated proteins: insight from transgenic mouse models. Int J Obes (Lond) 2011; 36: 581–594.
Kim JY, van de Wall E, Laplante M, Azzara A, Trujillo ME, Hofmann SM et al. Obesity-associated improvements in metabolic profile through expansion of adipose tissue. J Clin Invest 2007; 117: 2621–2637.
Acknowledgements
We acknowledge the clinical help of Oscar Rovira. This work was partially supported by research grants from the Ministerio de Economía y Competitividad (PI11-00214). CIBEROBN Fisiopatología de la Obesidad y Nutrición is an initiative from the Instituto de Salud Carlos III from Spain.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declared no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
Moreno-Navarrete, J., Ortega, F., Serrano, M. et al. CIDEC/FSP27 and PLIN1 gene expression run in parallel to mitochondrial genes in human adipose tissue, both increasing after weight loss. Int J Obes 38, 865–872 (2014). https://doi.org/10.1038/ijo.2013.171
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2013.171
Keywords
This article is cited by
-
Energetic metabolic reprogramming in Jurkat DFF40-deficient cancer cells
Molecular and Cellular Biochemistry (2022)
-
FSP27 and Links to Obesity and Diabetes Mellitus
Current Obesity Reports (2019)
-
New insights into apolipoprotein A5 in controlling lipoprotein metabolism in obesity and the metabolic syndrome patients
Lipids in Health and Disease (2018)
-
The Effects of Bariatric Surgery-Induced Weight Loss on Adipose Tissue in Morbidly Obese Women Depends on the Initial Metabolic Status
Obesity Surgery (2016)
-
An integrative systems genetics approach reveals potential causal genes and pathways related to obesity
Genome Medicine (2015)