Abstract
Aims:
The pathogenesis of obesity remains incompletely understood. Drosophila have conserved neuroendocrine and digestion systems with human and become an excellent system for studying energy homeostasis. Here, we reported a novel obesity Drosophila model, in which expression of human protein, synphilin-1 (SP1), in neurons fosters positive energy balance.
Subjects and methods:
To further understand the actions of SP1 in energy balance control, the upstream activation sequence UAS/GAL4 system was used to generate human SP1 transgenic Drosophila. We characterized a human SP1 transgenic Drosophila by assessing SP1 expression, fat lipid deposition, food intake and fly locomotor activity to determine the major behavioral changes and their consequences in the development of the obesity-like phenotype.
Results:
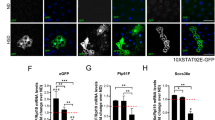
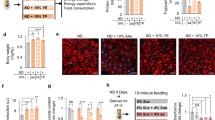
Overexpression of SP1 in neurons, but not peripheral cells, increased the body weight of flies compared with that of non-transgenic controls. SP1 increased food intake but did not affect locomotor activity. SP1 increased the levels of triacylglycerol, and the size of fat body cells and lipid droplets, indicating that SP1 increased lipid-fat disposition. Survival studies showed that SP1 transgenic flies were more resistant to food deprivation. SP1 regulated lipin gene expression that may participate in SP1-induced fat deposition and starvation resistance.
Conclusion:
These studies demonstrate that SP1 expression affects energy homeostasis in ways that enhance positive energy balance and provide a useful obesity model for future pathogenesis and therapeutic studies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ribeiro CS, Carneiro K, Ross CA, Menezes JR, Engelender S . Synphilin-1 is developmentally localized to synaptic terminals, and its association with synaptic vesicles is modulated by alpha-synuclein. J Biol Chem 2002; 277: 23927–23933.
Nagano Y, Yamashita H, Takahashi T, Kishida S, Nakamura T, Iseki E et al. Siah-1 facilitates ubiquitination and degradation of synphilin-1. J Biol Chem 2003; 278: 51504–51514.
Wakabayashi K, Engelender S, Yoshimoto M, Tsuji S, Ross CA, Takahashi H . Synphilin-1 is present in Lewy bodies in Parkinson’s disease. Ann Neurol 2000; 47: 521–523.
Engelender S, Kaminsky Z, Guo X, Sharp AH, Amaravi RK, Kleiderlein JJ et al. Synphilin-1 associates with alpha-synuclein and promotes the formation of cytosolic inclusions. Nat Genet 1999; 22: 110–114.
Smith WW, Margolis RL, Li X, Troncoso JC, Lee MK, Dawson VL et al. Alpha-synuclein phosphorylation enhances eosinophilic cytoplasmic inclusion formation in SH-SY5Y cells. J Neurosci 2005; 25: 5544–5552.
Chung KK, Zhang Y, Lim K L, Tanaka Y, Huang H, Gao J et al. Parkin ubiquitinates the alpha-synuclein-interacting protein, synphilin-1: implications for Lewy-body formation in Parkinson disease. Nat Med 2001; 7: 1144–1150.
Szargel R, Rott R, Engelender S . Synphilin-1 isoforms in Parkinson’s disease: regulation by phosphorylation and ubiquitylation. Cell Mol Life Sci 2008; 65: 80–88.
O’Farrell C, Murphy DD, Petrucelli L, Singleton AB, Hussey J, Farrer M et al. Transfected synphilin-1 forms cytoplasmic inclusions in HEK293 cells. Brain Res Mol Brain Res 2001; 97: 94–102.
Lee G, Tanaka M, Park K, Lee SS, Kim YM, Junn E et al. Casein kinase II-mediated phosphorylation regulates alpha-synuclein/synphilin-1 interaction and inclusion body formation. J Biol Chem 2004; 279: 6834–6839.
Avraham E, Szargel R, Eyal A, Rott R, Engelender S . Glycogen synthase kinase 3beta modulates synphilin-1 ubiquitylation and cellular inclusion formation by SIAH: implications for proteasomal function and Lewy body formation. J Biol Chem 2005; 280: 42877–42886.
Marx FP, Soehn AS, Berg D, Melle C, Schiesling C, Lang M et al. The proteasomal subunit S6 ATPase is a novel synphilin-1 interacting protein--implications for Parkinson’s disease. FASEB J 2007; 21: 1759–1767.
Alvarez-Castelao B, Castano JG . Synphilin-1 inhibits alpha-synuclein degradation by the proteasome. Cell Mol Life Sci 2011; 15: 2643–2654.
Smith WW, Liu Z, Liang Y, Masuda N, Swing DA, Jenkins NA et al. Synphilin-1 attenuates neuronal degeneration in the A53T {alpha}-synuclein transgenic mouse model. Hum Mol Genet 2010; 19: 2087–2098.
Li X, Liu Z, Tamashiro K, Shi B, Rudnicki DD, Ross CA et al. Synphilin-1 exhibits trophic and protective effects against Rotenone toxicity. Neuroscience 2010; 165: 455–462.
Giaime E, Sunyach C, Herrant M, Grosso S, Auberger P, McLean PJ et al. Caspase-3-derived C-terminal product of synphilin-1 displays antiapoptotic function via modulation of the p53-dependent cell death pathway. J Biol Chem 2006; 281: 11515–11522.
Li X, Tamashiro KL, Liu Z, Bello NT, Wang X, Aja S et al. A novel obesity model: synphilin-1-induced hyperphagia and obesity in mice. Int J Obes (Lond) 2011. e-pub ahead of print 13 December 2011 doi:10.1038/ijo.2011.235.
Baker KD, Thummel CS . Diabetic larvae and obese flies-emerging studies of metabolism in Drosophila. Cell Metab 2007; 6: 257–266.
Leopold P, Perrimon N . Drosophila and the genetics of the internal milieu. Nature 2007; 450: 186–188.
Markstein M, Pitsouli C, Villalta C, Celniker SE, Perrimon N . Exploiting position effects and the gypsy retrovirus insulator to engineer precisely expressed transgenes. Nat Genet 2008; 40: 476–483.
Ni JQ, Markstein M, Binari R, Pfeiffer B, Liu LP, Villalta C et al. Vector and parameters for targeted transgenic RNA interference in Drosophila melanogaster. Nat Methods 2008; 5: 49–51.
Al-Anzi B, Sapin V, Waters C, Zinn K, Wyman RJ, Benzer S . Obesity-blocking neurons in Drosophila. Neuron 2009; 63: 329–341.
Liu Z, Wang X, Yu Y, Li X, Wang T, Jiang H et al. A Drosophila model for LRRK2-linked parkinsonism. Proc Natl Acad Sci USA 2008; 105: 2693–2698.
Zhang H, Liu J, Li CR, Momen B, Kohanski RA, Pick L . Deletion of Drosophila insulin-like peptides causes growth defects and metabolic abnormalities. Proc Natl Acad Sci USA 2009; 106: 19617–19622.
Ja WW, Carvalho GB, Mak EM, de la Rosa NN, Fang AY, Liong JC et al. Prandiology of Drosophila and the CAFE assay. Proc Natl Acad Sci USA 2007; 104: 8253–8256.
Brand AH, Perrimon N . Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 1993; 118: 401–415.
Li T, Yang D, Sushchky S, Liu Z, Smith WW . Models for LRRK2-linked Parkinsonism. Parkinsons Dis 2011; 2011: 942412.
Markow TA . Perspective: female remating, operational sex ratio, and the arena of sexual selection in Drosophila species. Evolution 2002; 56: 1725–1734.
DiAngelo JR, Birnbaum MJ . Regulation of fat cell mass by insulin in Drosophila melanogaster. Mol Cell Biol 2009; 29: 6341–6352.
Csaki LS, Reue K . Lipins: multifunctional lipid metabolism proteins. Annu Rev Nutr 2010; 30: 257–272.
Harris TE, Finck BN . Dual function lipin proteins and glycerolipid metabolism. Trends Endocrinol Metab 2011; 22: 226–233.
Reue K . The lipin family: mutations and metabolism. Curr Opin Lipidol 2009; 20: 165–170.
Ugrankar R, Liu Y, Provaznik J, Schmitt S, Lehmann M . Lipin is a central regulator of adipose tissue development and function in Drosophila melanogaster. Mol Cell Biol 2011; 31: 1646–1656.
Goodman JM . Demonstrated and inferred metabolism associated with cytosolic lipid droplets. J Lipid Res 2009; 50: 2148–2156.
Beller M, Thiel K, Thul PJ, Jackle H . Lipid droplets: a dynamic organelle moves into focus. FEBS Lett 2010; 584: 2176–2182.
Harbison ST, Chang S, Kamdar KP, Mackay TF . Quantitative genomics of starvation stress resistance in Drosophila. Genome Biol 2005; 6: R36.
Reue K, Brindley DN . Thematic review series: glycerolipids. multiple roles for lipins/phosphatidate phosphatase enzymes in lipid metabolism. J Lipid Res 2008; 49: 2493–2503.
Finck BN, Gropler MC, Chen Z, Leone TC, Croce MA, Harris TE et al. Lipin 1 is an inducible amplifier of the hepatic PGC-1alpha/PPARalpha regulatory pathway. Cell Metab 2006; 4: 199–210.
Acknowledgements
We thank Dr Craig Montell for helpful discussion. We also thank Drs Bader Al-Anzi and Dr Youngseok Lee for kindly providing us the GAL4 driver flies. This work was supported by the National Institutes of Health, Grants: DK083410 to WWS, and the Paul R McHugh Chair for Motivated Behavior to THM.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Liu, J., Li, T., Yang, D. et al. Synphilin-1 alters metabolic homeostasis in a novel Drosophila obesity model. Int J Obes 36, 1529–1536 (2012). https://doi.org/10.1038/ijo.2012.111
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2012.111
Keywords
This article is cited by
-
Treating the Onset of Diabetes Using Probiotics Along with Prebiotic from Pachyrhizus erosus in High-Fat Diet Fed Drosophila melanogaster
Probiotics and Antimicrobial Proteins (2022)
-
Molecular and cytological analysis of widely-used Gal4 driver lines for Drosophila neurobiology
BMC Genetics (2020)
-
A tachykinin-like neuroendocrine signalling axis couples central serotonin action and nutrient sensing with peripheral lipid metabolism
Nature Communications (2017)
-
Glycerol-3-phosphate acyltransferase-1 upregulation by O-GlcNAcylation of Sp1 protects against hypoxia-induced mouse embryonic stem cell apoptosis via mTOR activation
Cell Death & Disease (2016)