Abstract
Objective:
Using a recombinant rat leptin antagonist, we investigated the effects of early postnatal leptin disruption on long-term leptin sensitivity and metabolic phenotype.
Design:
Three groups of 10 newborn female Wistar rats were injected subcutaneously with either saline (control) or leptin antagonist (at 2.5 or 7.5 μg g−1 day−1) from postnatal day 2 to day 13.
Results:
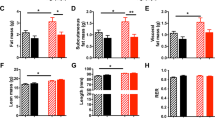
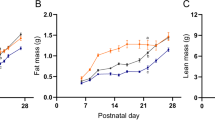
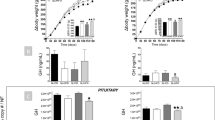
At weaning (day 28), antagonist-treated rats presented similar body weight (BW) compared to control animals. At 3 months of age, there was no significant change in BW, food intake and leptin or insulin levels between groups. Only a disturbed relationship between circulating insulin and glucose levels was observed in antagonist-treated animals. At 4 months of age, treated animals developed a leptin resistance appreciated by the lack of response to a 7-days leptin treatment (1 mg kg−1 day−1) in term of decrease in food intake and BW. At 8 months of age, following 3 months of high-energy diet, rlepm7.5 animals presented higher BW gain associated with increased body fatness and striking hyperleptinaemia as compared to control animals.
Conclusion:
The blockage of leptin action during the critical period of early life in rodents has long-term consequences by altering the capacity to respond to leptin during adulthood, thus predisposing the animals to obesity. These findings clearly demonstrate the physiological importance of the postnatal leptin surge for the optimal onset of the metabolic regulation, at least in rodents, and its implication in the prevention of unfavourable developmental programming.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ahima RS . Obesity: much silence makes a mighty noise. Gastroenterology 2007; 132: 2085–2086.
Roth J, Qiang X, Marban SL, Redelt H, Lowell BC . The obesity pandemic: where have we been and where are we going? Obes Res 2004; 12 (Suppl 2): 88S–101S.
Taylor PD, Poston L . Developmental programming of obesity in mammals. Exp Physiol 2007; 92: 287–298.
Gluckman PD, Hanson MA, Pinal C . The developmental origins of adult disease. Matern Child Nutr 2005; 1: 130–141.
Gluckman PD, Hanson MA, Beedle AS . Early life events and their consequences for later disease: a life history and evolutionary perspective. Am J Hum Biol 2007; 19: 1–19.
McMillen IC, Edwards LJ, Duffield J, Muhlhausler BS . Regulation of leptin synthesis and secretion before birth: implications for the early programming of adult obesity. Reproduction 2006; 131: 415–427.
Breier BH, Vickers MH, Ikenasio BA, Chan KY, Wong WP . Fetal programming of appetite and obesity. Mol Cell Endocrinol 2001; 185: 73–79.
Masuzaki H, Ogawa Y, Sagawa N, Hosoda K, Matsumoto T, Mise H et al. Nonadipose tissue production of leptin: leptin as a novel placenta-derived hormone in humans. Nat Med 1997; 3: 1029–1033.
Smith-Kirwin SM, O’Connor DM, De Johnston J, Lancey ED, Hassink SG, Funanage VL . Leptin expression in human mammary epithelial cells and breast milk. J Clin Endocrinol Metab 1998; 83: 1810–1813.
Aoki N, Kawamura M, Matsuda T . Lactation-dependent down regulation of leptin production in mouse mammary gland. Biochim Biophys Acta 1999; 1427: 298–306.
Bado A, Levasseur S, Attoub S, Kermorgant S, Laigneau JP, Bortoluzzi MN et al. The stomach is a source of leptin. Nature 1998; 394: 790–793.
Sobhani I, Bado A, Vissuzaine C, Buyse M, Kermorgant S, Laigneau JP et al. Leptin secretion and leptin receptor in the human stomach. Gut 2000; 47: 178–183.
Huang L, Li C . Leptin: a multifunctional hormone. Cell Res 2000; 10: 81–92.
Schwartz MW, Woods SC, Porte Jr D, Seeley RJ, Baskin DG . Central nervous system control of food intake. Nature 2000; 404: 661–671.
Harrold JA . Hypothalamic control of energy balance. Curr Drug Targets 2004; 5: 207–219.
Pinto S, Roseberry AG, Liu H, Diano S, Shanabrough M, Cai X et al. Rapid rewiring of arcuate nucleus feeding circuits by leptin. Science 2004; 304: 110–115.
Devaskar SU, Ollesch C, Rajakumar RA, Rajakumar PA . Developmental changes in ob gene expression and circulating leptin peptide concentrations. Biochem Biophys Res Commun 1997; 238: 44–47.
Ahima RS, Prabakaran D, Flier JS . Postnatal leptin surge and regulation of circadian rhythm of leptin by feeding. Implications for energy homeostasis and neuroendocrine function. J Clin Invest 1998; 101: 1020–1027.
Mistry AM, Swick A, Romsos DR . Leptin alters metabolic rates before acquisition of its anorectic effect in developing neonatal mice. Am J Physiol 1999; 277: R742–R747.
Ahima RS, Hileman SM . Postnatal regulation of hypothalamic neuropeptide expression by leptin: implications for energy balance and body weight regulation. Regul Pept 2000; 92: 1–7.
Proulx K, Richard D, Walker CD . Leptin regulates appetite-related neuropeptides in the hypothalamus of developing rats without affecting food intake. Endocrinology 2002; 143: 4683–4692.
Bouret SG, Draper SJ, Simerly RB . Formation of projection pathways from the arcuate nucleus of the hypothalamus to hypothalamic regions implicated in the neural control of feeding behavior in mice. J Neurosci 2004; 24: 2797–2805.
Bouret SG, Draper SJ, Simerly RB . Trophic action of leptin on hypothalamic neurons that regulate feeding. Science 2004; 304: 108–110.
Bouret SG, Simerly RB . Minireview: leptin and development of hypothalamic feeding circuits. Endocrinology 2004; 145: 2621–2626.
Jaquet D, Leger J, Tabone MD, Czernichow P, Levy-Marchal C . High serum leptin concentrations during catch-up growth of children born with intrauterine growth retardation. J Clin Endocrinol Metab 1999; 84: 1949–1953.
Fernandez-Twinn DS, Ozanne SE . Mechanisms by which poor early growth programs type-2 diabetes, obesity and the metabolic syndrome. Physiol Behav 2006; 88: 234–243.
Vickers MH, Breier BH, Cutfield WS, Hofman PL, Gluckman PD . Fetal origins of hyperphagia, obesity, and hypertension and postnatal amplification by hypercaloric nutrition. Am J Physiol Endocrinol Metab 2000; 279: E83–E87.
Vickers MH, Gluckman PD, Coveny AH, Hofman PL, Cutfield WS, Gertler A et al. Neonatal leptin treatment reverses developmental programming. Endocrinology 2005; 146: 4211–4216.
Salomon G, Niv-Spector L, Gussakovsky EE, Gertler A . Large-scale preparation of biologically active mouse and rat leptins and their L39A/D40A/F41A muteins which act as potent antagonists. Protein Expr Purif 2006; 47: 128–136.
Gertler A . Development of leptin antagonists and their potential use in experimental biology and medicine. Trends Endocrinol Metab 2006; 17: 372–378.
Holemans K, Caluwaerts S, Poston L, Van Assche FA . Diet-induced obesity in the rat: a model for gestational diabetes mellitus. Am J Obstet Gynecol 2004; 190: 858–865.
Godfrey KM, Lillycrop KA, Burdge GC, Gluckman PD, Hanson MA . Epigenetic mechanisms and the mismatch concept of the developmental origins of health and disease. Pediatr Res 2007; 61: 5R–10R.
Fall CH . The fetal and early life origins of adult disease. Indian Pediatr 2003; 40: 480–502.
Yajnik CS . Obesity epidemic in India: intrauterine origins? Proc Nutr Soc 2004; 63: 387–396.
Scarpace PJ, Matheny M, Zhang Y, Cheng KY, Tumer N . Leptin antagonist reveals an uncoupling between leptin receptor signal transducer and activator of transcription 3 signaling and metabolic responses with central leptin resistance. J Pharmacol Exp Ther 2007; 320: 706–712.
Zhang J, Matheny MK, Tumer N, Mitchell MK, Scarpace PJ . Leptin antagonist reveals that the normalization of caloric intake and the thermic effect of food after high-fat feeding are leptin dependent. Am J Physiol Regul Integr Comp Physiol 2007; 292: R868–R874.
Oliver P, Pico C, De Matteis R, Cinti S, Palou A . Perinatal expression of leptin in rat stomach. Dev Dyn 2002; 223: 148–154.
McFadin EL, Morrison CD, Buff PR, Whitley NC, Keisler DH . Leptin concentrations in periparturient ewes and their subsequent offspring. J Anim Sci 2002; 80: 738–743.
Casabiell X, Pineiro V, Tome MA, Peino R, Dieguez C, Casanueva FF . Presence of leptin in colostrum and/or breast milk from lactating mothers: a potential role in the regulation of neonatal food intake. J Clin Endocrinol Metab 1997; 82: 4270–4273.
Sanchez J, Oliver P, Miralles O, Ceresi E, Pico C, Palou A . Leptin orally supplied to neonate rats is directly uptaken by the immature stomach and may regulate short-term feeding. Endocrinology 2005; 146: 2575–2582.
Xiao XQ, Grove KL, Grayson BE, Smith MS . Inhibition of uncoupling protein expression during lactation: role of leptin. Endocrinology 2004; 145: 830–838.
Dundar NO, Anal O, Dundar B, Ozkan H, Caliskan S, Buyukgebiz A . Longitudinal investigation of the relationship between breast milk leptin levels and growth in breast-fed infants. J Pediatr Endocrinol Metab 2005; 18: 181–187.
Locke R . Preventing obesity: the breast milk-leptin connection. Acta Paediatr 2002; 91: 891–894.
Pico C, Oliver P, Sanchez J, Miralles O, Caimari A, Priego T et al. The intake of physiological doses of leptin during lactation in rats prevents obesity in later life. Int J Obes (Lond) 2007; 31: 1199–1209.
Acknowledgements
We are grateful to Dr Michel Parquet and Dr Michel Riottot (NMPA, IBAIC, Orsay) for their advice and help during animal experiments. We also thank all the team of the unit UEAR (INRA Jouy-en-Josas) and particularly Bernard Cayron, Christophe Auger and Blandine Barbier for their precious help in animal management.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Attig, L., Solomon, G., Ferezou, J. et al. Early postnatal leptin blockage leads to a long-term leptin resistance and susceptibility to diet-induced obesity in rats. Int J Obes 32, 1153–1160 (2008). https://doi.org/10.1038/ijo.2008.39
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2008.39
Keywords
This article is cited by
-
Programming of metabolism by adipokines during development
Nature Reviews Endocrinology (2023)
-
Early adversity promotes binge-like eating habits by remodeling a leptin-responsive lateral hypothalamus–brainstem pathway
Nature Neuroscience (2023)
-
Hormone levels in preterm and donor human milk before and after Holder pasteurization
Pediatric Research (2020)
-
Sex differences in major depression and comorbidity of cardiometabolic disorders: impact of prenatal stress and immune exposures
Neuropsychopharmacology (2019)
-
Cross-fostering reduces obesity induced by early exposure to monosodium glutamate in male rats
Endocrine (2017)