Abstract
Objective:
To provide an overview of the Healthy Lifestyle in Europe by Nutrition in Adolescence Cross-Sectional Study (HELENA-CSS) design, with particular attention to its quality control procedures. Other important methodological aspects are described in detail throughout this supplement.
Design:
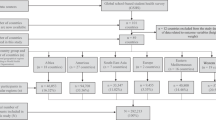
Description of the HELENA-CSS sampling and recruitment approaches, standardization and harmonization processes, data collection and analysis strategies and quality control activities.
Results:
The HELENA-CSS is a multi-centre collaborative study conducted in European adolescents located in urban settings. The data management systems, quality assurance monitoring activities, standardized manuals of operating procedures and training and study management are addressed in this paper. Various quality controls to ensure collection of valid and reliable data will be discussed in this supplement, as well as quantitative estimates of measurement error.
Conclusion:
The great advantage of the HELENA-CSS is the strict standardization of the fieldwork and the blood analyses, which precludes to a great extent the kind of immeasurable confounding bias that often interferes when comparing results from isolated studies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Dietz WH . Periods of risk in childhood for the development of adult obesity—what do we need to learn? J Nutr 1997; 127: 1884S–1886S.
Moreno LA, Mesana MI, Fleta J, Ruiz JR, González-Gross MM, Sarría A et al., the AVENA Study Group. Overweight, obesity and body fat composition in Spanish adolescents. The AVENA Study. Ann Nutr Metab 2005; 49: 71–76.
Silber TJ . Anorexia nervosa among children and adolescents. Adv Pediatr 2005; 52: 49–76.
Thane CW, Bates CJ, Prentice A . Risk factors for low iron intake and poor iron status in a national sample of British young people aged 4–18 years. Publ Health Nutr 2003; 6: 485–496.
Moreno LA, González-Gross MM, Kersting M, Molnár D, De Henauw S, Beghin L et al., on behalf of the HELENA Study Group. Assessing, understanding and modifying nutritional status, eating habits and physical activity in European adolescents. The HELENA Study. Publ Health Nutr 2008; 11: 288–299.
Matthys C, De Henauw S, Devos C, De Backer G . Estimated energy intake, macronutrient intake and meal pattern of Flemish adolescents. Eur J Clin Nutr 2003; 57: 366–375.
González-Gross M, Castillo MJ, Moreno LA, Nova E, González-Lamuño D, Pérez-Llamas F et al. Alimentación y valoración del estado nutricional de los adolescentes españoles (Estudio AVENA). Evaluación de riesgos y propuesta de intervención. I. Descripción metodológica del proyecto. Nutr Hosp 2003; 18: 15–28.
De Henauw S, Gottrand F, De Bourdeaudhuij I, Gonzalez-Gross M, Leclercq C, Kafatos A et al., on behalf of the HELENA Study Group. Nutritional status and lifestyles of adolescents in a public health perspective. The HELENA Project—Healthy Lifestyle in Europe by Nutrition in Adolescence. J Publ Health 2007; 15: 187–197.
Béghin L, Castera M, Manios Y, Gilbert C, Kersting M, De Henauw S et al., on behalf of the HELENA Study Group. Quality assurance of ethical issues and regulatory aspects relating to good clinical practices in the HELENA Cross-Sectional Study. Int J Obes (London) 2008; 32 (Suppl 5): S12–S18.
Moreno LA, Mesana MI, González-Gross M, Gil CM, Fleta J, Warnberg J et al. Anthropometric body fat composition reference values in Spanish adolescents. The AVENA Study. Eur J Clin Nutr 2006; 60: 191–196.
Cacciari E, Milani S, Balsamo A, Dammarco F, De Luca F, Chiarelli F et al. Italian cross-sectional growth charts for height, weight and BMI (6–20 y). Eur J Clin Nutr 2002; 56: 171–180.
Wärnberg J, Moreno LA, Mesana MI, Marcos A, the AVENA group. Inflammatory mediators in overweight and obese Spanish adolescents. The AVENA Study. Int J Obes Relat Metab Disord 2004; 28 (Suppl 3): S54–S58.
Wärnberg J, Nova E, Moreno LA, Romeo J, Mesana MI, Ruiz JR et al., the AVENA Study group. Inflammatory proteins are related to total and abdominal adiposity in a healthy adolescent population: the AVENA Study. Am J Clin Nutr 2006; 84: 505–512.
González-Gross M, Breidenassel C, Gómez S, Ferrari M, Béghin L, Spinneker A et al., on behalf of the HELENA Study Group. Sampling and processing of fresh blood samples within a European Multicenter Nutritional study: evaluation of biomarker stability during transport and storage. Int J Obes (London) 2008; 32 (Suppl 5): S66–S75.
Nagy E, Vicente-Rodriguez G, Manios Y, Beghin L, Iliescu C, Censi L et al., on behalf of the HELENA Study Group. Harmonization process and reliability assessment of anthropometric measurements in a multicenter study in adolescents. Int J Obes (London) 2008; 32 (Suppl 5): S58–S65.
Moreno LA, Joyanes M, Mesana MI, González-Gross M, Gil CM, Sarría A et al., and the AVENA Study Group. Harmonization of anthropometric measurements for a multicenter nutrition survey in Spanish adolescents. Nutrition 2003; 19: 481–486.
Iliescu C, Béghin L, Maes L, De Bourdeaudhuij I, Libersa C, Vereecken C, et al., on behalf of the HELENA Study Group. Socioeconomic questionnaire and clinical assessment in the HELENA Cross-Sectional Study: methodology. Int J Obes (London) 2008; 32 (Suppl 5): S19–S25.
Acknowledgements
The HELENA Study was carried out with the financial support of the European Community Sixth RTD Framework Programme (Contract FOOD-CT-2005-007034). The content of this paper reflects only the authors' views and the European Community is not liable for any use that may be made of the information contained therein. The researchers from the University of Zaragoza, Spain (LAM) are complementarily supported by FUNDACIÓN MAPFRE (Spain). We thank Ms Carine Vereecken, PhD, for her kind and constructive comments on this paper.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
Conflict of interest
Frédéric Gottrand has received consulting fees from Numico Clinical Nutrition, lecture fees from SMS and grant support from Danone Research. The remaining authors state no conflict of interest.
Rights and permissions
About this article
Cite this article
Moreno, L., De Henauw, S., González-Gross, M. et al. Design and implementation of the Healthy Lifestyle in Europe by Nutrition in Adolescence Cross-Sectional Study. Int J Obes 32 (Suppl 5), S4–S11 (2008). https://doi.org/10.1038/ijo.2008.177
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2008.177
Keywords
This article is cited by
-
Interplay of the Mediterranean diet and genetic hypertension risk on blood pressure in European adolescents: Findings from the HELENA study
European Journal of Pediatrics (2024)
-
Psychometric properties of the online Satisfaction with Life Scale in university students from a low-income region
Psicologia: Reflexão e Crítica (2023)
-
Association between PTPN1 polymorphisms and obesity-related phenotypes in European adolescents: influence of physical activity
Pediatric Research (2023)
-
Changes in physical activity patterns from adolescence to young adulthood: the BELINDA study
European Journal of Pediatrics (2023)
-
Clinical and physical characteristics of thinness in adolescents: the HELENA study
European Journal of Nutrition (2023)