Abstract
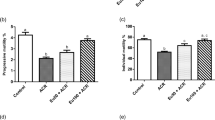
Although several studies on toxic effect of aspartame metabolite have been studied, controversial reports over the use of aspartame owing to the fact that it releases methanol as one of its metabolite during metabolism exist. This present study is proposed to investigate whether aspartame (40 mg kg−1 b.wt) administration for 90 days could induce oxidative stress and alter antioxidant status of epididymal sperm in Wistar strain male albino rats. To mimic the human methanol metabolism, methotrexate (MTX)-treated rats were included to study the effects of aspartame. Oral intubations of FDA approved 40 mg kg−1 b.wt aspartame were given daily for 90 days to Wistar strain male albino rats and studied along with controls and MTX-treated controls. Sperm count, viability, morphology, morphometry and motility were assessed. A significant decrease in sperm function of aspartame treated animals was observed when compared with the control and MTX control. The free radical generation were observed in epididymal sperm by assessing the scavenging enzymes, enzymatic and non-enzymatic antioxidants. Result suggest that there was a significant increase glutathione-s-transferase (GST), with a significant decrease in reduced glutathione (GSH), superoxide dismutase activity (SOD), glutathione peroxidase levels (GPx), catalase activity (CAT) and glutathione reductase concentration. The increase in free radicals generation could have ultimately caused the lipid peroxidation mediated damages on the testis. Aspartame treated animals also revealed the reduced space in seminiferous tubules, which resulted in reduced Leydig cells when compared with control in histopathology. These findings demonstrate that aspartame metabolites could be a contributing factor for development of oxidative stress in the epididymal sperm.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Jeganathan PS, Namasivayam A . Methanol induced biogenic amine changes in discrete areas of rat brain: role of simultaneous ethanol administration. Indian J Physiol Pharmacol 1998; 32: 1–10.
Soffritti M, Belpoggi F, Padovani M, Lauricia M, Dagli Spositi D, Minardi F . Life-time carcinogenicity biossay of toluene given by stomach tube to Sprague-Dawley rats. Eur J Oncol 2004; 9: 91–102.
McMartin KE, Martin-Amat G, Noker PE, Tephly TR . Lack of a role for formaldehyde in methanol poisoning in the monkey. Biochem Pharm 1978; 28: 645–649.
Lee EW, Garner CD, Terzo TS . Animal model for the study of methanol toxicity comparison of folate-reduced rat responses with published monkey data. J Toxicol Environ Health 1994; 41: 71–82.
Iyyaswamy A, Rathinasamy S . Effect of chronic exposure to aspartame on oxidative stress in the brain of albino rats. J Biosci 2012; 37: 679–688.
Ashok I, Sheeladevi R . Biochemical responses and mitochondrial mediated activation of apoptosis on long-term effect of aspartame in rat brain. Redox Biol 2014; 2: 820–831.
Parthasarathy JN, Ramasundaram SK, Sundaramahalingam M, Rathinasamy SD . Methanol induced oxidative stress in rat lymphoid organs. J Occup Health 2006; 48: 20–27.
Ashok I, Sheeladevi R, Dapkupar W . Effect of long-term aspartame (artificial sweetener) on anxiety, locomotor activity and emotionality behavior in Wistar Albino rats. Biomed Prev Nutr 2014; 4: 39–43.
Ashok I, Sheeladevi R . Oxidant stress evoked damages in rat hepatocyte leading to triggered NOS levels on long term consumption of aspartame. J Food Drug Anal 2015; 23: 679–691.
Bergstrom BP, Cummings DR, Skaggs TA . Aspartame decreases evoked extracellular dopamine levels in the rat brain: an in vivo voltammetry study. Neuropharmacology 2007; 53: 967–974.
Ashok I, Sheeladevi R, Wankhar D . Long term effect of aspartame (Artificial sweetener) on membrane homeostatic imbalance and histopathology in the rat brain. Free Radic Antioxid 2013; 3: S42–S49.
Ashok I, Sheeladevi R, Dapkupar W . Acute effect of aspartame (artificial sweetener) induced oxidative stress in the brain regions of wistar albino rats. J Biomed Res 2014; 28: 1–7.
Gonzalez-Quevedo A, Obregon F, Urbina M, Rousso T, Lima L . Effect of chronic methanol administration on amino acids and monoamines in retina, optic nerve, and brain of the rat. Toxicol Appl Pharmacol 2002; 185: 77–84.
Tabor HW . A method for determination of formiminoglutamic acid in urine. J Clin Investig 1962; 37: 824–828.
Marklund S, Marklund G . Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 1974; 47: 469–474.
Sinha AK . Colorimetric assay of catalase. Anal Biochem 1972; 47: 389–394.
Rotruck JT . Selenium: biochemical role as a component of glutathione peroxi- dase. Science 1973; 179: 588–590.
Habig WH, Palst MJ, Jakoby WB . Glutathione-S-transferase the first enzymatic step in mercapturic formation. J Biol Chem 1973; 249: 7130.
Charles EM, Robert GL . Hepatic glutathione reductase Purification and general kinetic properties. J Biol Chem 1962; 237: 1589–1595.
Omaye ST, Amball JDT, Sauberlich HE . Selected method for the determination of ascorbic acid in animal cells, tissue and fluid. Methods Enzymol 1979; 62: 1–11.
Moron MS, Depierre JW, Mannervik B . Levels of glutathione, glutathione re- ductase and glutathione S-transferase activities in rat lung and liver. Biochim Biophys Acta 1979; 582: 67–78.
Ohkawa H, Ohishi M, Yagi K . Assay for lipid peroxidase in animal tissues by thiobarbituric acid reaction. Anal Biochem 1970; 95: 351–358.
Atessahin A, Karahan I, Turk G, Gur S, Yilmaz S, Cerbibasi AO . Protective role of lycopene on cisplatin-induced changes in sperm characteristics testicular damage and oxidative stress in rats. Reprod Toxicol 2006; 21: 42–47.
Wyrobek AJ, Gordon LA, Burkhart JG, Francis MW, Kapp RWJ, Letz G et al. An evaluation of the mouse sperm morphology test and other sperm tests in nonhuman mammals. A report of the U.S. Environmental Protection Agency Gene-Tox Program. Mutation Res 1983; 115: 1–72.
Hafez ESS Techniques of Human Andrology. Elsevier / North-Holland Biomedical Press: Amsterdam, 1977 p 471.
WHO Laboratory Manual for Examination of Human Semen and Sperm-Cervical Mucus Interaction. 4th edn. Published on behalf of the World Health Organization by Cambridge University Press: Cambridge, UK, 1999.
Goodman J, Tephly TR . The role of hepatic microbody and soluble oxidases in the peroxidation of methanol in the rat and monkey. Mol Pharmacol 1968; 4: 492–501.
Ganong. Ganong’s Review of Medical Physiology (24 ed.). TATA McGRAW HILL. pp. 419–420. ISBN 978-1-25-902753-6.
Nandi S, Banerjee PP, Zirkin BR . Germ cell apoptosis in the testes of Sprague Dawley rats following testosterone withdrawal by ethane 1, 2 dimethanesulfonate administration: relationship of Fas. Biol Reprod 1999; 61: 70–75.
Gil-Guzman E, Ollero M, Lopez MC, Sharma RK, Alvarez JG, Thomas AJ Jr . Differential production of reactive oxygen species by subjects of human spermatozoa at different stages of maturation. Hum Reprod 2001; 16: 1922–1930.
Young KA, Zirkin BR, Nelson RJ . Short photoperiod induces testicular apoptosis in the white-footed mouse (peromyscus leucopus). Endocrinology 1999; 140: 1331–1339.
Sikka SC . Relative impact of oxidative stress on male reproductive function. Curr Med Chem 2001; 8: 851–862.
Armstrong JS, Rajasekaran M, Chamulitrat W, Gatti P, Hellstrom WJ, Sikka SC . Characterization of reactive oxygen species induced effects on human spermatozoa movement and energy metabolism. Free Radic Biol Med 1999; 26: 869–880.
Duru NK, Morshedi M, Ochninger S . Effects of hydrogen peroxide on DNA and plasma membrane integrity of human spermatozoa. Fertil Steril 2000; 74: 1200–1207.
Syntin P, Robaire B . Sperm structural and motility changes during aging in the brown Norway rat. J Androl 2001; 22: 235–244.
Giorgio M, Trinei M, Migliaccio E, Pelicci PG . Hydrogen peroxide: a metabolic by-product or a common mediator of ageing signals? Nat Rev Mol Cell Biol 2007; 8: 722–728.
De Lamirande E, Gagnon C . Reactive oxygen species and human spermatozoa: effects on the motility of intact spermatozoa and on sperm axonemes. J Androl 1992; 13: 368–378.
Jones R, Mann T, Shreins RJ . Peroxidative breakdown of phospholipids in human spermatozoa: spermicidal effects of fatty acid peroxides and protective action of seminal plasma. Fertil Steril 1979; 31: 531–537.
Weir CP, Robaire B . Spermatozoa have decreased antioxidant enzymatic capacity and increased reactive oxygen species production during aging in the brown Norway rat. J Androl 2007; 28: 229–240.
Mari M, Wu D, Nieto N, Cederbaum AI . CYP2E1-dependent toxicity and upregulation of antioxidant genes. J Biomed Sci 2001; 8: 52–55.
Maher P . The effects of stress and aging on glutathione metabolism. Ageing Res Rev 2005; 4: 288–314.
Lenzi A, Cualosso F, Gandini L, Lombardo F, Dondero F . Placebo controlled, double-blind cross over trial of glutathione therapy, in male infertility. Hum Reprod 1993; 9: 2050.
Calvin HI, Cooper GW, Wallace E . Evidence that selenium in rat sperm is associated with a cystine rich structural protein of the mitochondrial capsule. Gamete Res 1981; 4: 139–149.
Rommerts FFG . Testosterone: an overview of biosynthesis transport metabolism and action. In: Nieschlag E, Hehre HM (eds). Testosterone Action Deficiency Substitution. Springler-Verlag: Berlin, 1990, pp 1–22.
Acknowledgements
The author is grateful to the valuable suggestion offered by Dr NJ Parthasarathy and Co-authors. The author is grateful to the valuable help given by L. Sundareswaran, Wankupar Wankhar is acknowledged. We thank Mr. GS. Sravan for his language check on this manuscript. The financial assistance provided by the Indian Council of Medical Research (ICMR) File. No. 3 / 1 / 2 / 29 / Nut. / 2012 / Dated 29-09-2013 for Senior Research Fellow is gratefully acknowledged. I acknowledge University of Madras for providing the infrastructure to conduct the research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Ashok, I., Poornima, P., Wankhar, D. et al. Oxidative stress evoked damages on rat sperm and attenuated antioxidant status on consumption of aspartame. Int J Impot Res 29, 164–170 (2017). https://doi.org/10.1038/ijir.2017.17
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijir.2017.17