Abstract
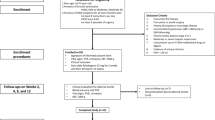
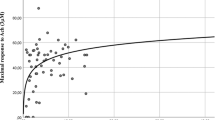
End-stage renal disease (ESRD) affects sexual function. The etiology of sexual dysfunction in ESRD is multifactorial and hypogonadism is not uncommon. In a cohort of 96 men with ESRD, scores on International Index of Erectile Function (IIEF) correlated with bioavailable testosterone (T; P<0.01). We further evaluated the effects of administration of T on ED in hypogonadal men on hemodialysis. Nine men with ED and hypogonadism (baseline bioavailable T <3.82 nmol l−1), received 5 g T-gel (1% (10 mg g−1) per day treatment for 6 months. The IIEF and frequency of sexual intercourse per week were used to assess treatment efficacy. T administration significantly increased serum T levels to the normal range, and was associated with statistically significant improvements of the scores of the IIEF domains (erectile function from 7.6±8.0 to 12.8±7.8; intercourse satisfaction from 3.4±4.5 to 4.8±3.1; orgasmic function from 4.8±3.1 to 3.4±2.9; sexual desire from 3.4±2.9 to 4.4±0.6; overall sexual satisfaction from 2.6±1.3 to 3.8±1.0). But scores of the IIEF did not normalize. Frequency of weekly sexual intercourse increased from 0.4±0.4 to 1.1±0.8 (P<0.05) after 6 months. There were no adverse effects. We conclude that restoring T levels to normal in men with ESRD improved scores on the IIEF and frequency of weekly sexual intercourse.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bellinghieri G, Santoro D, Mallamace A, Savica V . Sexual dysfunction in chronic renal failure. J Nephrol 2008; 21 (Suppl 13): S113–S117.
Anantharaman P, Schmidt RJ . Sexual function in chronic kidney disease. Adv Chronic Kidney Dis 2007; 14: 119–125.
Rosas SE, Joffe M, Franklin E, Strom BL, Kotzker W, Brensinger C et al. Prevalence and determinants of erectile dysfunction in hemodialysis patients. Kidney Int 2001; 59: 2259–2266.
Ali ME, Abdel-Hafez HZ, Mahran AM, Mohamed HZ, Mohamed ER, El-Shazly AM et al. Erectile dysfunction in chronic renal failure patients undergoing hemodialysis in Egypt. Int J Impot Res 2005; 17: 180–185.
Cerqueira J, Moraes M, Glina S . Erectile dysfunction: prevalence and associated variables in patients with chronic renal failure. Int J Impot Res 2002; 14: 65–71.
Palmer BF . Sexual dysfunction in men and women with chronic kidney disease and end-stage kidney disease. Adv Ren Replace Ther 2003; 10: 48–60.
Brockenbrough AT, Dittrich MO, Page ST, Smith T, Stivelman JC, Bremner WJ . Transdermal androgen therapy to augment EPO in the treatment of anemia of chronic renal disease. Am J Kidney Dis 2006; 47: 251–262.
Johansen KL . Treatment of hypogonadism in men with chronic kidney disease. Adv Chronic Kidney Dis 2004; 11: 348–356.
Johansen KL . Testosterone metabolism and replacement therapy in patients with end-stage renal disease. Semin Dial 2004; 17: 202–208.
Carrero JJ, Qureshi AR, Parini P, Arver S, Lindholm B, Barany P et al. Low serum testosterone increases mortality risk among male dialysis patients. J Am Soc Nephrol 2009; 20: 613–620.
Lawrence IG, Price DE, Howlett TA, Harris KP, Feehally J, Walls J . Correcting impotence in the male dialysis patient: experience with testosterone replacement and vacuum tumescence therapy. Am J Kidney Dis 1998; 31: 313–319.
Chatterjee R, Wood S, McGarrigle HH, Lees WR, Ralph DJ, Neild GH . A novel therapy with testosterone and sildenafil for erectile dysfunction in patients on renal dialysis or after renal transplantation. J Fam Plann Reprod Health Care 2004; 30: 88–90.
Singh AB, Norris K, Modi N, Sinha-Hikim I, Shen R, Davidson T et al. Pharmacokinetics of a transdermal testosterone system in men with end stage renal disease receiving maintenance hemodialysis and healthy hypogonadal men. J Clin Endocrinol Metab 2001; 86: 2437–2445.
Rosen RC, Riley A, Wagner G, Osterloh IH, Kirkpatrick J, Mishra A . The International Index of Erectile Function (IIEF): a multidimensional scale for assessment of erectile dysfunction. Urology 1997; 49: 822–830.
Cappelleri JC, Rosen RC, Smith MD, Mishra A, Osterloh IH . Diagnostic evaluation of the erectile function domain of the International Index of Erectile Function. Urology 1999; 54: 346–351.
Swerdloff RS, Wang C, Cunningham G, Dobs A, Iranmanesh A, Matsumoto AM et al. Long-term pharmacokinetics of transdermal testosterone gel in hypogonadal men. J Clin Endocrinol Metab 2000; 85: 4500–4510.
Lew-Starowicz M, Gellert R . The sexuality and quality of life of hemodialyzed patients—ASED multicenter study. J Sex Med 2009; 6: 1062–1071.
Saad F, Grahl AS, Aversa A, Yassin AA, Kadioglu A, Moncada I et al. Effects of testosterone on erectile function: implications for the therapy of erectile dysfunction. BJU Int 2007; 99: 988–992.
Aversa A, Isidori AM, Greco EA, Giannetta E, Gianfrilli D, Spera E et al. Hormonal supplementation and erectile dysfunction. Eur Urol 2004; 45: 535–538.
Traish AM, Goldstein I, Kim NN . Testosterone and erectile function: from basic research to a new clinical paradigm for managing men with androgen insufficiency and erectile dysfunction. Eur Urol 2007; 52: 54–70.
Traish A, Kim N . The physiological role of androgens in penile erection: regulation of corpus cavernosum structure and function. J Sex Med 2005; 2: 759–770.
Traish AM, Guay AT . Are androgens critical for penile erections in humans? Examining the clinical and preclinical evidence. J Sex Med 2006; 3: 382–404; discussion 404–387.
Shabsigh R, Rajfer J, Aversa A, Traish AM, Yassin A, Kalinchenko SY et al. The evolving role of testosterone in the treatment of erectile dysfunction. Int J Clin Pract 2006; 60: 1087–1092.
Bellinghieri G, Santoro G, Santoro D, Lo Forti B, Savica V, Favazzi P et al. Ultrastructural changes of corpora cavernosa in men with erectile dysfunction and chronic renal failure. Semin Nephrol 2004; 24: 488–491.
Karakitsos D, Patrianakos AP, De Groot E, Boletis J, Karabinis A, Kyriazis J et al. Androgen deficiency and endothelial dysfunction in men with end-stage kidney disease receiving maintenance hemodialysis. Am J Nephrol 2006; 26: 536–543.
Dunkel L, Raivio T, Laine J, Holmberg C . Circulating luteinizing hormone receptor inhibitor(s) in boys with chronic renal failure. Kidney Int 1997; 51: 777–784.
Handelsman DJ, Liu PY . Androgen therapy in chronic renal failure. Baillieres Clin Endocrinol Metab 1998; 12: 485–500.
Toorians AW, Janssen E, Laan E, Gooren LJ, Giltay EJ, Oe PL et al. Chronic renal failure and sexual functioning: clinical status versus objectively assessed sexual response. Nephrol Dial Transplant 1997; 12: 2654–2663.
Tas A, Ersoy A, Ersoy C, Gullulu M, Yurtkuran M . Efficacy of sildenafil in male dialysis patients with erectile dysfunction unresponsive to erythropoietin and/or testosterone treatments. Int J Impot Res 2006; 18: 61–68.
Seibel I, Poli De Figueiredo CE, Teloken C, Moraes JF . Efficacy of oral sildenafil in hemodialysis patients with erectile dysfunction. J Am Soc Nephrol 2002; 13: 2770–2775.
Bancroft J . The endocrinology of sexual arousal. J Endocrinol 2005; 186: 411–427.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cangüven, Ö., Aykose, G., Albayrak, S. et al. Efficacy of testosterone gel in the treatment of erectile dysfunction in hypogonadal hemodialysis patients: a pilot study. Int J Impot Res 22, 140–145 (2010). https://doi.org/10.1038/ijir.2009.55
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijir.2009.55