Abstract
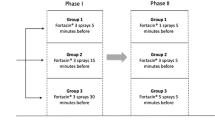
Premature ejaculation (PE) is thought to be the most common male sexual dysfunction; however, the prevalence of lifelong (LL)-PE is relatively low. The aim of this study was to investigate the effects of on-demand vardenafil (10 mg) to modify the intravaginal ejaculatory latency time (IELT) in men with LL-PE without erectile dysfunction. Forty-two men (18–35 years) were enrolled in a 16-week, double-blind, placebo-controlled, cross-over study. Primary end point was the modification from baseline of IELT assessed by stopwatch technique; secondary end points were post-ejaculatory refractory time (PERT) and variations of scores at the Index of Premature Ejaculation questionnaire. The changes in geometric mean IELT were superior after taking vardenafil (0.6±0.3 vs 4.5±1.1 min, P<0.01), compared with placebo (0.7±0.3 vs 0.9±1.0 min, ns). PERT dropped significantly after vardenafil (16.7±2.0 vs 4.3±0.9 min, P<0.001), compared with placebo (15.3±2.2 vs 15.8±2.3 min). Patients who took vardenafil (vs placebo) reported significantly (P<0.01) increased ejaculatory control (6±2 vs 16±2), improved overall sexual satisfaction (7±2 vs 15±1) and distress (4±1 vs 8±1) scores, respectively. Multiple regression analysis (r2=0.86) for IELT by the number of attempts at sexual intercourse showed significant differences between the slopes of lines for placebo and vardenafil (P<0.0001). The most common adverse events for vardenafil (vs placebo) were headache (10 vs 3%), flushing (12 vs 0%) and dyspepsia (10 vs 0%), which tended to disappear over the time. In conclusion, in our study, vardenafil increased IELT and reduced PERT in men with LL-PE. Besides, improvements in confidence, perception of ejaculatory control and overall sexual satisfaction were reported.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Porst H, Montorsi F, Rosen RC, Gaynor L, Grupe S, Alexander J . The Premature Ejaculation Prevalence and Attitudes (PEPA) survey: prevalence, comorbidities, and professional help-seeking. Eur Urol 2007; 51: 816–823.
Waldinger MD, Zwinderman AH, Olivier B, Schweitzer DH . Proposal for a definition of lifelong premature ejaculation based on epidemiological stopwatch data. J Sex Med 2005; 2: 498–507.
McMahon CG, Althof S, Waldinger MD, Porst H, Dean J, Sharlip I et al., International Society for Sexual Medicine Ad Hoc Committee for Definition of Premature Ejaculation. An evidence-based definition of lifelong premature ejaculation: report of the International Society for Sexual Medicine Ad Hoc Committee for the Definition of Premature Ejaculation. BJU Int 2008; 102: 338–350.
Waldinger MD, Schweitzer DH . The use of old and recent DSM definitions of premature ejaculation in observational studies: a contribution to the present debate for a new classification of PE in the DSM-V. J Sex Med 2008; 5: 1079–1087.
Waldinger MD . Lifelong premature ejaculation: from authority-based to evidence-based medicine. BJU Int 2004; 93: 201–207.
Waldinger MD, Zwinderman AH, Schweitzer DH, Olivier B . Relevance of methodological design for the interpretation of efficacy of drug treatment of premature ejaculation: a systematic review and meta-analysis. Int J Impot Res 2004; 16: 369–381.
Mathers MJ, Klotz T, Roth S, Lümmen G, Sommer F . Safety and efficacy of vardenafil versus sertraline in the treatment of premature ejaculation: a randomised, prospective and crossover study. Andrologia 2009; 41: 169–175.
Aversa A, Mazzilli F, Rossi T, Delfino M, Isidori AM, Fabbri A . Effects of sildenafil (Viagra) administration on seminal parameters and post-ejaculatory refractory time in normal males. Hum Reprod 2000; 15: 131–134.
Mondaini N, Ponchietti R, Muir GH, Montorsi F, Di Loro F, Lombardi G et al. Sildenafil does not improve sexual function in men without erectile dysfunction but does reduce the post-orgasmic refractory time. Int J Impot Res 2003; 15: 225–228.
Althof S, Rosen R, Symonds T, Mundayat R, May K, Abraham L . Development and validation of new questionnaire to assess sexual satisfaction, control, and distress associated with premature ejaculation. J Sex Med 2006; 3: 465–475.
Symonds T, Perelman MA, Althof S, Giuliano F, Martin M, May K et al. Development and validation of a premature ejaculation diagnostic tool. Eur Urol 2007; 52: 565–573.
Cappelleri JC, Rosen RC, Smith MD, Mishra A, Osterloh IH . Diagnostic evaluation of the erectile function domain of the International Index of Erectile Function. Urology 1999; 54: 346–351.
Waldinger MD, Zwinderman AH, Olivier B, Schweitzer DH . Geometric mean IELT and premature ejaculation: appropriate statistics to avoid overestimation of treatment efficacy. J Sex Med 2008; 5: 492–499.
Chen J, Keren-Paz G, Bar-Yosef Y, Matzkin H . The role of phosphodiesterase type 5 inhibitors in the management of premature ejaculation: a critical analysis of basic science and clinical data. Eur Urol 2007; 52: 1331–1339.
Chen J, Mabjeesh NJ, Matzkin H, Greenstein A . Efficacy of sildenafil as adjuvant therapy to selective serotonin reuptake inhibitor in alleviating premature ejaculation. Urology 2003; 61: 197–200.
McMahon CG, Stuckey BGA, Andersen M, Purvis K, Koppiker N, Haughie S et al. Efficacy of Sildenafil Citrate (Viagra) in men with premature ejaculation. J Sex Med 2005; 2: 368–375.
Salonia A, Maga T, Colombo R, Scattoni V, Briganti A, Cestari A et al. A prospective study comparing paroxetine alone versus paroxetine plus sildenafil in patients with premature ejaculation. J Urol 2002; 168: 2486–2489.
Wang WF, Wang Y, Minhas S, Ralph DJ . Can sildenafil treat primary premature ejaculation? A prospective clinical study. Int J Urol 2007; 14: 331–335.
Rowland DL, Motofei IG . The aetiology of premature ejaculation and the mind-body problem: implications for practice. Int J Clin Pract 2007; 61: 77–82.
Pfaus JG . Neurobiology of sexual behavior. Curr Opin Neurobiol 1999; 9: 751–758.
Prickaerts J, van Staveren WC, Sik A, Markerink-van Ittersum M, Niewöhner U, van der Staay FJ et al. Effects of two selective phosphodiesterase type 5 inhibitors, sildenafil and vardenafil, on object recognition memory and hippocampal cyclic GMP levels in the rat. Neuroscience 2002; 113: 351–361.
Castellano M, Rizzoni D, Beschi M, Muiesan ML, Porteri E, Bettoni G et al. Relationship between sympathetic nervous system activity, baroreflex and cardiovascular effects after acute nitric oxide synthesis inhibition in humans. J Hypertens 1995; 13: 1153–1161.
Krukoff TL . Central regulation of autonomic function: no brakes? Clin Exp Pharmacol Physiol 1998; 25: 474–478.
Uckert S, Bazrafshan S, Scheller F, Mayer ME, Jonas U, Stief CG . Functional responses of isolated human seminal vesicle tissue to selective phosphodiesterase inhibitors. Urology 2007; 70: 185–189.
Uckert S, Bazrafshan S, Sonnenberg JE, Kuczyk ME . Effects of phosphodiesterase inhibitors on the contractile responses of isolated human seminal vesicle tissue to adrenergic stimulation. J Sex Med 2009; 6: 408–414.
Uckert S, Sormes M, Kedia G, Scheller F, Knapp WH, Jonas U et al. Effects of phosphodiesterase inhibitors on tension induced by norepinephrine and accumulation of cyclic nucleotides in isolated human prostatic tissue. Urology 2008; 71: 526–530.
Kriegsfeld LJ, Demas GE, Huang PL, Burnett AL, Nelson RJ . Ejaculatory abnormalities in mice lacking the gene for endothelial nitric oxide synthase (eNOS−/−). Physiol Behav 1999; 67: 561–566.
Teixeira CE, Priviero FB, Webb RC . Differential effects of the phosphodiesterase type 5 inhibitors sildenafil, vardenafil, and tadalafil in rat aorta. J Pharmacol Exp Ther 2006; 316: 654–661.
Toque HA, Teixeira CE, Priviero FB, Morganti RP, Antunes E, De Nucci G . Vardenafil, but not sildenafil or tadalafil, has calcium-channel blocking activity in rabbit isolated pulmonary artery and human washed platelets. Br J Pharmacol 2008; 154: 787–796.
Waldinger MD, Zwinderman AH, Olivier B, Schweitzer DH . The majority of men with lifelong premature ejaculation prefer daily drug treatment: an observation study in a consecutive group of Dutch men. J Sex Med 2007; 4: 1028–1037.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aversa, A., Pili, M., Francomano, D. et al. Effects of vardenafil administration on intravaginal ejaculatory latency time in men with lifelong premature ejaculation. Int J Impot Res 21, 221–227 (2009). https://doi.org/10.1038/ijir.2009.21
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijir.2009.21
Keywords
This article is cited by
-
Current and emerging treatment options for premature ejaculation
Nature Reviews Urology (2022)
-
Redefining a sexual medicine paradigm: subclinical premature ejaculation as a new taxonomic entity
Nature Reviews Urology (2021)
-
Management of premature ejaculation: a clinical guideline from the Italian Society of Andrology and Sexual Medicine (SIAMS)
Journal of Endocrinological Investigation (2021)
-
Safety and efficacy characteristics of oral drugs in patients with premature ejaculation: a Bayesian network meta-analysis of randomized controlled trials
International Journal of Impotence Research (2019)
-
Pharmacological interventions for premature ejaculation: a mixed-treatment comparison network meta-analysis of randomized clinical trials
International Journal of Impotence Research (2018)