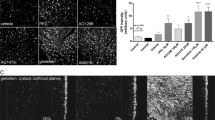
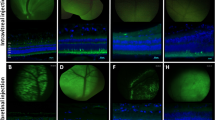
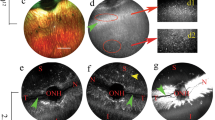
Abstract
Gene therapy using adeno-associated viral (AAV) vectors for the treatment of retinal degenerations has shown safety and efficacy in clinical trials. However, very high levels of vector expression may be necessary for the treatment of conditions such as Stargardt disease where a dual vector approach is potentially needed, or in optogenetic strategies for end-stage degeneration in order to achieve maximal light sensitivity. In this study, we assessed two vectors with single capsid mutations, rAAV2/2(Y444F) and rAAV2/8(Y733F) in their ability to transduce retina in the Abca4−/− and rd1 mouse models of retinal degeneration. We noted significantly increased photoreceptor transduction using rAAV2/8(Y733F) in the Abca4−/− mouse, in contrast to previous work where vectors tested in this model have shown low levels of photoreceptor transduction. Bipolar cell transduction was achieved following subretinal delivery of both vectors in the rd1 mouse, and via intravitreal delivery of rAAV2/2(Y444F). The successful use of rAAV2/8(Y733F) to target bipolar cells was further validated on human tissue using an ex vivo culture system of retinal explants. Capsid mutant AAV vectors transduce human retinal cells and may be particularly suited to treat retinal degenerations in which high levels of transgene expression are required.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bainbridge JW, Smith AJ, Barker SS, Robbie S, Henderson R, Balaggan K et al. Effect of gene therapy on visual function in Leber's congenital amaurosis. N Engl J Med 2008; 358: 2231–2239.
Maguire AM, Simonelli F, Pierce EA, Pugh Jr EN, Mingozzi F, Bennicelli J et al. Safety and efficacy of gene transfer for Leber's congenital amaurosis. N Engl J Med 2008; 358: 2240–2248.
Cideciyan AV, Hauswirth WW, Aleman TS, Kaushal S, Schwartz SB, Boye SL et al. Vision 1 year after gene therapy for Leber's congenital amaurosis. N Engl J Med 2009; 361: 725–727.
Maclaren RE, Groppe M, Barnard AR, Cottriall CL, Tolmachova T, Seymour L et al. Retinal gene therapy in patients with choroideremia: initial findings from a phase 1/2 clinical trial. Lancet 2014; 383: 1129–1137.
Bennett J, Ashtari M, Wellman J, Marshall KA, Cyckowski LL, Chung DC et al. AAV2 gene therapy readministration in three adults with congenital blindness. Sci Transl Med 2012; 4: 120ra15.
Jacobson SG, Cideciyan AV, Ratnakaram R, Heon E, Schwartz SB, Roman AJ et al. Gene therapy for leber congenital amaurosis caused by RPE65 mutations: safety and efficacy in 15 children and adults followed up to 3 years. Arch Ophthalmol 2012; 130: 9–24.
Testa F, Maguire AM, Rossi S, Pierce EA, Melillo P, Marshall K et al. Three-year follow-up after unilateral subretinal delivery of adeno-associated virus in patients with Leber congenital Amaurosis type 2. Ophthalmology 2013; 120: 1283–1291.
Petrs-Silva H, Dinculescu A, Li Q, Deng WT, Pang JJ, Min SH et al. Novel properties of tyrosine-mutant AAV2 vectors in the mouse retina. Mol Ther 2011; 19: 293–301.
Dalkara D, Byrne LC, Klimczak RR, Visel M, Yin L, Merigan WH et al. In vivo-directed evolution of a new adeno-associated virus for therapeutic outer retinal gene delivery from the vitreous. Sci Transl Med 2013; 5: 189ra76.
Kay CN, Ryals RC, Aslanidi GV, Min SH, Ruan Q, Sun J et al. Targeting photoreceptors via intravitreal delivery using novel, capsid-mutated AAV vectors. PLoS One 2013; 8: e62097.
Rabinowitz JE, Rolling F, Li C, Conrath H, Xiao W, Xiao X et al. Cross-packaging of a single adeno-associated virus (AAV) type 2 vector genome into multiple AAV serotypes enables transduction with broad specificity. J Virol 2002; 76: 791–801.
Vandenberghe LH, Bell P, Maguire AM, Cearley CN, Xiao R, Calcedo R et al. Dosage thresholds for AAV2 and AAV8 photoreceptor gene therapy in monkey. Sci Transl Med 2011; 3: 88ra54.
Zhong L, Li B, Mah CS, Govindasamy L, Agbandje-McKenna M, Cooper M et al. Next generation of adeno-associated virus 2 vectors: point mutations in tyrosines lead to high-efficiency transduction at lower doses. Proc Natl Acad Sci USA 2008; 105: 7827–7832.
Petrs-Silva H, Dinculescu A, Li Q, Min SH, Chiodo V, Pang JJ et al. High-efficiency transduction of the mouse retina by tyrosine-mutant AAV serotype vectors. Mol Ther 2009; 17: 463–471.
Lochrie MA, Tatsuno GP, Christie B, McDonnell JW, Zhou S, Surosky R et al. Mutations on the external surfaces of adeno-associated virus type 2 capsids that affect transduction and neutralization. J Virol 2006; 80: 821–834.
Doroudchi MM, Greenberg KP, Liu J, Silka KA, Boyden ES, Lockridge JA et al. Virally delivered channelrhodopsin-2 safely and effectively restores visual function in multiple mouse models of blindness. Mol Ther 2011; 19: 1220–1229.
Ku CA, Chiodo VA, Boye SL, Goldberg AF, Li T, Hauswirth WW et al. Gene therapy using self-complementary Y733F capsid mutant AAV2/8 restores vision in a model of early onset Leber congenital amaurosis. Hum Mol Genet 2011; 20: 4569–4581.
McCarty DM, Fu H, Monahan PE, Toulson CE, Naik P, Samulski RJ . Adeno-associated virus terminal repeat (TR) mutant generates self-complementary vectors to overcome the rate-limiting step to transduction in vivo. Gene Ther 2003; 10: 2112–2118.
Yokoi K, Kachi S, Zhang HS, Gregory PD, Spratt SK, Samulski RJ et al. Ocular gene transfer with self-complementary AAV vectors. Invest Ophthalmol Vis Sci 2007; 48: 3324–3328.
Lipinski DM, Thake M, MacLaren RE . Clinical applications of retinal gene therapy. Prog Retin Eye Res 2013; 32: 22–47.
Allocca M, Doria M, Petrillo M, Colella P, Garcia-Hoyos M, Gibbs D et al. Serotype-dependent packaging of large genes in adeno-associated viral vectors results in effective gene delivery in mice. J Clin Invest 2008; 118: 1955–1964.
Hirsch ML, Agbandje-McKenna M, Samulski RJ . Little vector, big gene transduction: fragmented genome reassembly of adeno-associated virus. Mol Ther 2010; 18: 6–8.
Lopes VS, Boye SE, Louie CM, Boye S, Dyka F, Chiodo V et al. Retinal gene therapy with a large MYO7A cDNA using adeno-associated virus. Gene Ther 2013; 20: 824–833.
Charbel Issa P, De Silva SR, Lipinski DM, Singh MS, Mouravlev A, You Q et al. Assessment of tropism and effectiveness of new primate-derived hybrid recombinant AAV serotypes in the mouse and primate retina. PLoS One 2013; 8: e60361.
Bowes C, Li T, Danciger M, Baxter LC, Applebury ML, Farber DB . Retinal degeneration in the rd mouse is caused by a defect in the beta subunit of rod cGMP-phosphodiesterase. Nature 1990; 347: 677–680.
Lagali PS, Balya D, Awatramani GB, Munch TA, Kim DS, Busskamp V et al. Light-activated channels targeted to ON bipolar cells restore visual function in retinal degeneration. Nat Neurosci 2008; 11: 667–675.
Cronin T, Vandenberghe LH, Hantz P, Juttner J, Reimann A, Kacso AE et al. Efficient transduction and optogenetic stimulation of retinal bipolar cells by a synthetic adeno-associated virus capsid and promoter. EMBO Mol Med 2014; 6: 1175–1190.
Mace E, Caplette R, Marre O, Sengupta A, Chaffiol A, Barbe P et al. Targeting channelrhodopsin-2 to ON-bipolar cells with vitreally administered AAV restores ON and OFF visual responses in blind mice. Mol Ther 2014; 23: 7–16.
Yin L, Greenberg K, Hunter JJ, Dalkara D, Kolstad KD, Masella BD et al. Intravitreal injection of AAV2 transduces macaque inner retina. Invest Ophthalmol Vis Sci 2011; 52: 2775–2783.
Johnson TV, Martin KR . Development and characterization of an adult retinal explant organotypic tissue culture system as an in vitro intraocular stem cell transplantation model. Invest Ophthalmol Vis Sci 2008; 49: 3503–3512.
Lipinski DM, Singh MS, MacLaren RE . Assessment of cone survival in response to CNTF, GDNF, and VEGF165b in a novel ex vivo model of end-stage retinitis pigmentosa. Invest Ophthalmol Vis Sci 2011; 52: 7340–7346.
Tolmachova T, Tolmachov OE, Barnard AR, de Silva SR, Lipinski DM, Walker NJ et al. Functional expression of Rab escort protein 1 following AAV2-mediated gene delivery in the retina of choroideremia mice and human cells ex vivo. J Mol Med 2013; 91: 825–837.
Charbel Issa P, Barnard AR, Singh MS, Carter E, Jiang Z, Radu RA et al. Fundus autofluorescence in the Abca4(-/-) mouse model of Stargardt disease—correlation with accumulation of A2E, retinal function, and histology. Invest Ophthalmol Vis Sci 2013; 54: 5602–5612.
Bowles DE, Rabinowitz JE, Samulski RJ . Marker rescue of adeno-associated virus (AAV) capsid mutants: a novel approach for chimeric AAV production. J Virol 2003; 77: 423–432.
Fradot M, Busskamp V, Forster V, Cronin T, Leveillard T, Bennett J et al. Gene therapy in ophthalmology: validation on cultured retinal cells and explants from postmortem human eyes. Hum Gene Ther 2011; 22: 587–593.
Weng J, Mata NL, Azarian SM, Tzekov RT, Birch DG, Travis GH . Insights into the function of Rim protein in photoreceptors and etiology of Stargardt's disease from the phenotype in abcr knockout mice. Cell 1999; 98: 13–23.
Charbel Issa P, Singh MS, Lipinski DM, Chong NV, Barnard AR, Maclaren RE . Optimization of in vivo confocal autofluorescence imaging of the ocular fundus in mice and its application to models of human retinal degeneration. Invest Ophthalmol Vis Sci 2012; 53: 1066–1075.
Acknowledgements
This study was supported by Wellcome Trust, NIHR Biomedical Research Centres of Oxford and Moorfields, MRC, Fight for Sight, Royal College of Surgeons ProRetina.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
REM is a Founder and Director of Nightstarx Ltd (Gibbs Building, 215 Euston Road, London NW1 2BE), a choroideremia gene therapy company established by the University of Oxford and funded by the Wellcome Trust through Syncona Partners.
Additional information
Supplementary Information accompanies this paper on Gene Therapy website
Rights and permissions
About this article
Cite this article
De Silva, S., Charbel Issa, P., Singh, M. et al. Single residue AAV capsid mutation improves transduction of photoreceptors in the Abca4−/− mouse and bipolar cells in the rd1 mouse and human retina ex vivo. Gene Ther 23, 767–774 (2016). https://doi.org/10.1038/gt.2016.54
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2016.54
This article is cited by
-
X-Linked Retinitis Pigmentosa Gene Therapy: Preclinical Aspects
Ophthalmology and Therapy (2023)
-
A 2020 vision of ocular gene therapy
Gene Therapy (2021)
-
Exosome-associated AAV2 vector mediates robust gene delivery into the murine retina upon intravitreal injection
Scientific Reports (2017)
-
Tropism of engineered and evolved recombinant AAV serotypes in the rd1 mouse and ex vivo primate retina
Gene Therapy (2017)
-
Emerging Gene Therapies for Genetic Hearing Loss
Journal of the Association for Research in Otolaryngology (2017)