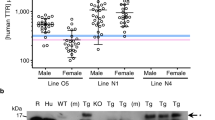
Abstract
Familial amyloidotic polyneuropathy (FAP) is a neurodegenerative disorder characterized by extracellular deposition of amyloid fibrils composed by mutated transthyretin (TTR) mainly in the peripheral nervous system. At present, liver transplantation is still the standard treatment to halt the progression of clinical symptoms in FAP, but new therapeutic strategies are emerging, including the use of TTR stabilizers. Here we propose to establish a new gene therapy approach using adeno-associated virus (AAV) vectors to deliver the trans-suppressor TTR T119M variant to the liver of transgenic TTR V30M mice at different ages. This TTR variant is known for its ability to stabilize the tetrameric protein. Analysis of the gastrointestinal tract of AAV-treated animals revealed a significant reduction in deposition of TTR non-fibrillar aggregates in as much as 34% in stomach and 30% in colon, as well as decreased levels of biomarkers associated with TTR deposition, namely the endoplasmic reticulum stress marker BiP and the extracellular matrix protein MMP-9. Moreover, we showed with different studies that our approach leads to an increase in tetrameric and more stable forms of TTR, in favor of destabilized monomers. Altogether our data suggest the possibility to use this gene therapy approach in a prophylactic manner to prevent FAP pathology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Andrade C . A peculiar form of peripheral neuropathy; familiar atypical generalized amyloidosis with special involvement of the peripheral nerves. Brain 1952; 75: 408–427.
Raz A, Goodman DS . The interaction of thyroxine with human plasma prealbumin and with the prealbumin-retinol-binding protein complex. J Biol Chem 1969; 244: 3230–3237.
Sekijima Y, Wiseman RL, Matteson J, Hammarström P, Miller SR, Sawkar AR et al. The biological and chemical basis for tissue-selective amyloid disease. Cell 2005; 121: 73–85.
Quintas A, Vaz DC, Cardoso I, Saraiva MJ, Brito RM . Tetramer dissociation and monomer partial unfolding precedes protofibril formation in amyloidogenic transthyretin variants. J Biol Chem 2001; 276: 27207–27213.
Adams D, Theaudin M, Cauquil C, Algalarrondo V, Slama M . FAP neuropathy and emerging treatments. Curr Neurol Neurosci Rep 2014; 14: 435.
Ando Y . Liver transplantation and new therapeutic approaches for familial amyloidotic polyneuropathy (FAP). Med Mol Morphol 2005; 38: 142–154.
Ando Y, Ueda M . Diagnosis and therapeutic approaches to transthyretin amyloidosis. Curr Med Chem 2012; 19: 2312–2323.
Alves IL, Altland K, Almeida MR, Winter P, Saraiva MJ . Screening and biochemical characterization of transthyretin variants in the Portuguese population. Hum Mutat 1997; 9: 226–233.
Coelho T, Maia LF, Martins da Silva A, Waddington Cruz M, Plante-Bordeneuve V, Lozeron P et al. Tafamidis for transthyretin familial amyloid polyneuropathy: a randomized, controlled trial. Neurology 2012; 79: 785–792.
Berk JL, Suhr OB, Obici L, Sekijima Y, Zeldenrust SR, Yamashita T et al. Repurposing diflunisal for familial amyloid polyneuropathy: a randomized clinical trial. JAMA 2013; 310: 2658–2667.
Coelho T, Chorão R, Sousa A, Alves I, Torres MF, Saraiva MJM . Compound heterozygotes of transthyretin Met30 and transthyretin Met119 are protected from the devastating effects of familial amyloid polyneuropathy. Neuromuscul Disord 1996; 6: S20.
Alves IL, Hays MT, Saraiva MJ . Comparative stability and clearance of [Met30]transthyretin and [Met119]transthyretin. Eur J Biochem 1997; 249: 662–668.
Sebastiao MP, Lamzin V, Saraiva MJ, Damas AM . Transthyretin stability as a key factor in amyloidogenesis: X-ray analysis at atomic resolution. J Mol Biol 2001; 306: 733–744.
Hammarström P, Schneider F, Kelly JW . Trans-suppression of misfolding in an amyloid disease. Science 2001; 293: 2459–2462.
Hammarström P, Wiseman RL, Powers ET, Kelly JW . Prevention of transthyretin amyloid disease by changing protein misfolding energetics. Science 2003; 299: 713–716.
Daya S, Berns KI . Gene therapy using adeno-associated virus vectors. Clin Microbiol Rev 2008; 21: 583–593.
Nakai H, Fuess S, Storm TA, Muramatsu S, Nara Y, Kay MA . Unrestricted hepatocyte transduction with adeno-associated virus serotype 8 vectors in mice. J Virol 2005; 79: 214–224.
Mingozzi F, Liu Y-L, Dobrzynski E, Kaufhold A, Liu JH, Wang Y et al. Induction of immune tolerance to coagulation factor IX antigen by in vivo hepatic gene transfer. J Clin Invest 2003; 111: 1347–1356.
Reed GH, Wittwer CT . Sensitivity and specificity of single-nucleotide polymorphism scanning by high-resolution melting analysis. Clin Chem 2004; 50: 1748–1754.
Reed GH, Kent JO, Wittwer CT . High-resolution DNA melting analysis for simple and efficient molecular diagnostics. Pharmacogenomics 2007; 8: 597–608.
Malentacchi F, Forni G, Vinci S, Orlando C . Quantitative evaluation of DNA methylation by optimization of a differential-high resolution melt analysis protocol. Nucleic Acids Res 2009; 37: e86.
Alves IL, Divino CM, Schussler GC, Altland K, Almeida MR, Palha JA et al. Thyroxine binding in a TTR Met 119 kindred. J Clin Endocrinol Metab 1993; 77: 484–488.
Almeida MR, Damas AM, Lans MC, Brouwer A, Saraiva MJ . Thyroxine binding to transthyretin Met 119. Comparative studies of different heterozygotic carriers and structural analysis. Endocrine 1997; 6: 309–315.
Altland K, Rauh S, Hackler R . Demonstation of human prealbumin by double one-dimensional slab gel electrophoresis. Electrophoresis 1981; 2: 148–155.
Monteiro FA, Sousa MM, Cardoso I, do Amaral JB, Guimarães A, Saraiva MJ . Activation of ERK1/2 MAP kinases in familial amyloidotic polyneuropathy. J Neurochem 2006; 97: 151–161.
Kohno K, Palha JA, Miyakawa K, Saraiva MJ, Ito S, Mabuchi T et al. Analysis of amyloid deposition in a transgenic mouse model of homozygous familial amyloidotic polyneuropathy. Am J Pathol 1997; 150: 1497–1508.
Sousa MM, Cardoso I, Fernandes R, Guimarães A, Saraiva MJ . Deposition of transthyretin in early stages of familial amyloidotic polyneuropathy: evidence for toxicity of nonfibrillar aggregates. Am J Pathol 2001; 159: 1993–2000.
Sousa MM, do Amaral JB, Guimarães A, Saraiva MJ . Up-regulation of the extracellular matrix remodeling genes, biglycan, neutrophil gelatinase-associated lipocalin, and matrix metalloproteinase-9 in familial amyloid polyneuropathy. FASEB J 2005; 19: 124–126.
Teixeira PF, Cerca F, Santos SD, Saraiva MJ . Endoplasmic reticulum stress associated with extracellular aggregates. Evidence from transthyretin deposition in familial amyloid polyneuropathy. J Biol Chem 2006; 281: 21998–22003.
Macedo B, Batista AR, do Amaral JB, Saraiva MJ . Biomarkers in the assessment of therapies for familial amyloidotic polyneuropathy. Mol Med 2007; 13: 584–591.
Cardoso I, Brito M, Saraiva MJ . Extracellular matrix markers for disease progression and follow-up of therapies in familial amyloid polyneuropathy V30M TTR-related. Dis Markers 2008; 25: 37–47.
Nakamura M, Ando Y, Nagahara S, Sano A, Ochiya T, Maeda S et al. Targeted conversion of the transthyretin gene in vitro and in vivo. Gene Therapy 2004; 11: 838–846.
Benson MD, Kluve-Beckerman B, Zeldenrust SR, Siesky AM, Bodenmiller DM, Showalter AD et al. Targeted suppression of an amyloidogenic transthyretin with antisense oligonucleotides. Muscle Nerve 2006; 33: 609–618.
Benson MD, Smith RA, Hung G, Kluve-Beckerman B, Showalter AD, Sloop KW et al. Suppression of choroid plexus transthyretin levels by antisense oligonucleotide treatment. Amyloid 2010; 17: 43–49.
Coelho T, Adams D, Silva A, Lozeron P, Hawkins PN, Mant T et al. Safety and efficacy of RNAi therapy for transthyretin amyloidosis. N Engl J Med 2013; 369: 819–829.
Kamata M, Susanto MT, Chen ISY . Enhanced transthyretin tetramer stability following expression of an amyloid disease transsuppressor variant in mammalian cells. J Gene Med 2009; 11: 103–111.
Xiao W, Berta SC, Lu MM, Moscioni AD, Tazelaar J, Wilson JM . Adeno-associated virus as a vector for liver-directed gene therapy. J Virol 1998; 72: 10222–10226.
Warrington KH, Herzog RW . Treatment of human disease by adeno-associated viral gene transfer. Hum Genet 2006; 119: 571–603.
Mccarty DM, Fu H, Monahan PE, Toulson CE, Naik P, Samulski RJ . Adeno-associated virus terminal repeat (TR) mutant generates self-complementary vectors to overcome the rate-limiting step to transduction in vivo. Gene Therapy 2003; 10: 2112–2118.
Davidoff AM, Gray JT, Ng CY, Zhang Y, Zhou J, Spence Y et al. Comparison of the ability of adeno-associated viral vectors pseudotyped with serotype 2, 5, and 8 capsid proteins to mediate efficient transduction of the liver in murine and nonhuman primate models. Mol Ther 2005; 11: 875–888.
Nathwani AC, Cochrane M, McIntosh J, Ng CY, Zhou J, Gray JT et al. Enhancing transduction of the liver by adeno-associated viral vectors. Gene Therapy 2009; 16: 60–69.
Wang Z, Zhu T, Qiao C, Zhou L, Wang B, Zhang J et al. Adeno-associated virus serotype 8 efficiently delivers genes to muscle and heart. Nat Biotechnol 2005; 23: 321–328.
Broekman ML, Baek RC, Comer LA, Fernandez JL, Seyfried TN, Sena-Esteves M . Complete correction of enzymatic deficiency and neurochemistry in the GM1-gangliosidosis mouse brain by neonatal adeno-associated virus-mediated gene delivery. Mol Ther 2007; 15: 30–37.
Nathwani AC, Gray JT, Ng CY, Zhou J, Spence Y, Waddington SN et al. Self-complementary adeno-associated virus vectors containing a novel liver-specific human factor IX expression cassette enable highly efficient transduction of murine and nonhuman primate liver. Blood 2006; 107: 2653–2661.
Broekman ML, Comer LA, Hyman BT, Sena-Esteves M . Adeno-associated virus vectors serotyped with AAV8 capsid are more efficient than AAV-1 or -2 serotypes for widespread gene delivery to the neonatal mouse brain. Neuroscience 2006; 138: 501–510.
Cardoso I, Almeida MR, Ferreira N, Arsequell G, Valencia G, Saraiva MJ . Comparative in vitro and ex vivo activities of selected inhibitors of transthyretin aggregation: relevance in drug design. Biochem J 2007; 408: 131–138.
Bustin SA, Benes V, Garson JA, Hellemans J, Huggett J, Kubista et al. The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem 2009; 55: 611–622.
Almeida MR, Macedo B, Cardoso I, Alves I, Valencia G, Arsequell G et al. Selective binding to transthyretin and tetramer stabilization in serum from patients with familial amyloidotic polyneuropathy by an iodinated diflunisal derivative. Biochem J 2004; 381: 351–356.
Acknowledgements
We would like to thank Paula Gonçalves, Susete Costelha and Anabela Teixeira (NBM Lab, IBMC) for tissue processing and helpful discussions, respectively. We are indebted to Eduardo Lopes from Bio-Rad Portugal for providing the equipment and software used in HRM, as well as for his helpful discussions on the subject. The authors also thank Fundação para a Ciência e Tecnologia (FCT), Portugal, for fellowship SFRH/BD/40511/2007 to ARB. This work was supported by FEDER funds through the Operational Competitiveness Program—COMPETE and by national funding from FCT under the projects FCOMP-01-0124-FEDER-022718 (PEST-c/SAU/LA0002/2011 and PTDC/SAU-ORG/111313/2009).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Batista, A., Gianni, D., Ventosa, M. et al. Gene therapy approach to FAP: in vivo influence of T119M in TTR deposition in a transgenic V30M mouse model. Gene Ther 21, 1041–1050 (2014). https://doi.org/10.1038/gt.2014.86
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2014.86