Abstract
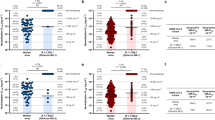
Adeno-associated viruses (AAV) have attracted attention as potential vectors for gene therapy and vaccines against several diseases, including HIV-1 infection. However, the presence of neutralizing antibodies (NAbs) after natural AAV infections inhibits their transfection in re-exposed subjects. To identify candidate AAV vectors for therapeutic or prophylactic HIV vaccines, NAbs against AAV2, AAV5 and AAV8 were screened in the sera of healthy individuals in China and 10 developed countries and an HIV-1-infected Chinese population. Seroprevalence was higher for AAV2 (96.6%) and AAV8 (82.0%) than for AAV5 (40.2%) in normal Chinese subjects. Among individuals seropositive for AAV5, >80% had low NAb titers (<1:90). The prevalence and titers of NAbs against the three AAVs were significantly higher in China than in developed countries (P<0.01). The prevalence of NAbs against AAV5 did not differ significantly between healthy and HIV-1-infected Chinese subjects (P=0.39). Co-occurrence of NAbs against AAV2, AAV5, and AAV8 was observed in the healthy population, and 15, 41, and 41% of individuals were AAV2+, AAV2+/AAV8+, and AAV2+/AAV5+/AAV8+, respectively. Therefore, AAV5 exposure is low in healthy and HIV-1-infected populations Chinese individuals, and vectors based on AAV5 may be appropriate for human gene therapy or vaccines.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Daya S, Berns KI . Gene therapy using adeno-associated virus vectors. Clin Microbiol Rev 2008; 21: 583–593.
Tenenbaum L, Lehtonen E, Monahan PE . Evaluation of risks related to the use of adeno-associated virus-based vectors. Curr Gene Ther 2003; 3: 545–565.
Li H, Malani N, Hamilton SR, Schlachterman A, Bussadori G, Edmonson SE et al. Assessing the potential for AAV vector genotoxicity in a murine model. Blood 2012; 117: 3311–3319.
Mingozzi F, High KA . Immune responses to AAV in clinical trials. Curr Gene Ther 2011; 11: 321–330.
Mueller C, Flotte TR . Clinical gene therapy using recombinant adeno-associated virus vectors. Gene Therapy 2008; 15: 858–863.
Xin KQ, Mizukami H, Urabe M, Toda Y, Shinoda K, Yoshida A et al. Induction of robust immune responses against human immunodeficiency virus is supported by the inherent tropism of adeno-associated virus type 5 for dendritic cells. J Virol 2006; 80: 11899–11910.
Manno CS, Pierce GF, Arruda VR, Glader B, Ragni M, Rasko JJ et al. Successful transduction of liver in hemophilia by AAV-Factor IX and limitations imposed by the host immune response. Nat Med 2006; 12: 342–347.
Lin J, Calcedo R, Vandenberghe LH, Figueredo JM, Wilson JM . Impact of preexisting vector immunity on the efficacy of adeno-associated virus-based HIV-1 Gag vaccines. Hum Gene Ther 2008; 19: 663–669.
Jiang H, Couto LB, Patarroyo-White S, Liu T, Nagy D, Vargas JA et al. Effects of transient immunosuppression on adenoassociated, virus-mediated, liver-directed gene transfer in rhesus macaques and implications for human gene therapy. Blood 2006; 108: 3321–3328.
Scallan CD, Jiang H, Liu T, Patarroyo-White S, Sommer JM, Zhou S et al. Human immunoglobulin inhibits liver transduction by AAV vectors at low AAV2 neutralizing titers in SCID mice. Blood 2006; 107: 1810–1817.
Wang L, Calcedo R, Bell P, Lin J, Grant RL, Siegel DL et al. Impact of pre-existing immunity on gene transfer to nonhuman primate liver with adeno-associated virus 8 vectors. Hum Gene Ther 2011; 22: 1389–1401.
Boutin S, Monteilhet V, Veron P, Leborgne C, Benveniste O, Montus MF et al. Prevalence of serum IgG and neutralizing factors against adeno-associated virus (AAV) types 1, 2, 5, 6, 8, and 9 in the healthy population: implications for gene therapy using AAV vectors. Hum Gene Ther 2010; 21: 704–712.
Petry H, Brooks A, Orme A, Wang P, Liu P, Xie J et al. Effect of viral dose on neutralizing antibody response and transgene expression after AAV1 vector re-administration in mice. Gene Therapy 2008; 15: 54–60.
Calcedo R, Vandenberghe LH, Gao G, Lin J, Wilson JM . Worldwide epidemiology of neutralizing antibodies to adeno-associated viruses. J Infect Dis 2009; 199: 381–390.
van der Marel S, Comijn EM, Verspaget HW, van Deventer S, van den Brink GR, Petry H et al. Neutralizing antibodies against adeno-associated viruses in inflammatory bowel disease patients: implications for gene therapy. Inflamm Bowel Dis 2011; 17: 2436–2442.
Kwon I, Schaffer DV . Designer gene delivery vectors: molecular engineering and evolution of adeno-associated viral vectors for enhanced gene transfer. Pharm Res 2008; 25: 489–499.
Peden CS, Burger C, Muzyczka N, Mandel RJ . Circulating anti-wild-type adeno-associated virus type 2 (AAV2) antibodies inhibit recombinant AAV2 (rAAV2)-mediated, but not rAAV5-mediated, gene transfer in the brain. J Virol 2004; 78: 6344–6359.
Gao GP, Alvira MR, Wang L, Calcedo R, Johnston J, Wilson JM . Novel adeno-associated viruses from rhesus monkeys as vectors for human gene therapy. Proc Natl Acad Sci USA 2002; 99: 11854–11859.
Pei H, Wan X, Hu W, Dong X, Li B . Construction and detection of a novel type of recombinant human rAAV2/2-ND4. Eye Sci 2013; 28: 55–59.
Dong X, Tian W, Yuan Z, Tan S, Wu X . A novel packaging system of recombinant AAV5/5 vector. Chin J Biotechnol 2010; 26: 679–686.
Wang Z, Yang S, Zhou L, Du H, Mo W, Zeng Y . Specific cellular immune responses in mice immunized with DNA, adeno-associated virus and adenoviral vaccines of Epstein-Barr virus-LMP2 alone or in combination. Sci China Life Sci 2011; 54: 263–266.
Dong X, Tian W, Wang G, Dong Z, Shen W, Zheng G et al. Establishment of an AAV reverse infection-based array. PLoS One 2010; 5: e13479.
Liu Q, Huang W, Zhao C, Zhang L, Meng S, Gao D et al. The prevalence of neutralizing antibodies against AAV serotype 1 in healthy subjects in China: implications for gene therapy and vaccines using AAV1 vector. J Med Virol 2013; 85: 1550–1556.
Grimm D, Kay MA . From virus evolution to vector revolution: use of naturally occurring serotypes of adeno-associated virus (AAV) as novel vectors for human gene therapy. Curr Gene Ther 2003; 3: 281–304.
Vincent T, Harvey BG, Hogan SM, Bailey CJ, Crystal RG, Leopold PL . Rapid assessment of adenovirus serum neutralizing antibody titer based on quantitative, morphometric evaluation of capsid binding and intracellular trafficking: population analysis of adenovirus capsid association with cells is predictive of adenovirus infectivity. J Virol 2001; 75: 1516–1521.
Halbert CL, Miller AD, McNamara S, Emerson J, Gibson RL, Ramsey B et al. Prevalence of neutralizing antibodies against adeno-associated virus (AAV) types 2, 5, and 6 in cystic fibrosis and normal populations: Implications for gene therapy using AAV vectors. Hum Gene Ther 2006; 17: 440–447.
Chirmule N, Propert K, Magosin S, Qian Y, Qian R, Wilson J . Immune responses to adenovirus and adeno-associated virus in humans. Gene Therapy 1999; 6: 1574–1583.
Gao G, Vandenberghe LH, Alvira MR, Lu Y, Calcedo R, Zhou X et al. Clades of adeno-associated viruses are widely disseminated in human tissues. J Virol 2004; 78: 6381–6388.
Maheshri N, Koerber JT, Kaspar BK, Schaffer DV . Directed evolution of adeno-associated virus yields enhanced gene delivery vectors. Nat Biotechnol 2006; 24: 198–204.
Li C, Diprimio N, Bowles DE, Hirsch ML, Monahan PE, Asokan A et al. Single amino acid modification of adeno-associated virus capsid changes transduction and humoral immune profiles. J Virol 2012; 86: 7752–7759.
Riviere C, Danos O, Douar AM . Long-term expression and repeated administration of AAV type 1, 2 and 5 vectors in skeletal muscle of immunocompetent adult mice. Gene Therapy 2006; 13: 1300–1308.
Zaiss AK, Muruve DA . Immune responses to adeno-associated virus vectors. Curr Gene Ther 2005; 5: 323–331.
Lee GK, Maheshri N, Kaspar B, Schaffer DV . PEG conjugation moderately protects adeno-associated viral vectors against antibody neutralization. Biotechnol Bioeng 2005; 92: 24–34.
Acknowledgements
We thank Dr Xiaoyan Dong for providing the rAAV–Fluc and AAV vectors. We also thank Dr Xuerong Jia of the Beijing Wantai Biological Pharmacy, Dr Furong Ren of the Beijing Red Cross Blood Center, Professor Jingyun Li of the Beijing Institute of Microbiology and Epidemiology and Dr Lisi Yao of the Chinese Academy of Inspection and Quarantine for providing the human sera. The cooperation of the human donors involved in the epidemiological study is greatly appreciated. This study was supported by grants from the National Science and Technology Major Project (2012ZX10004701-001). The funder had no role in the study design, data collection or analysis, decision to publish or the preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Liu, Q., Huang, W., Zhang, H. et al. Neutralizing antibodies against AAV2, AAV5 and AAV8 in healthy and HIV-1-infected subjects in China: implications for gene therapy using AAV vectors. Gene Ther 21, 732–738 (2014). https://doi.org/10.1038/gt.2014.47
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2014.47
This article is cited by
-
Multicenter assessment and longitudinal study of the prevalence of antibodies and related adaptive immune responses to AAV in adult males with hemophilia
Gene Therapy (2024)
-
Immune profiling of adeno-associated virus response identifies B cell-specific targets that enable vector re-administration in mice
Gene Therapy (2023)
-
Application of Gene Therapy in Hemophilia
Current Medical Science (2022)
-
Single-dose rAAV5-based vaccine provides long-term protective immunity against SARS-CoV-2 and its variants
Virology Journal (2022)
-
Advances in gene therapy for hemophilia: basis, current status, and future perspectives
International Journal of Hematology (2020)