Abstract
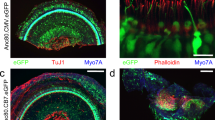
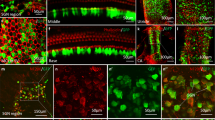
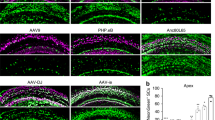
Loss of balance is often due to loss of vestibular hair cells. In mammals, regeneration of functional hair cells in the mature sensory epithelium is limited; therefore, loss of sensory cells can lead to debilitating balance problems. Delivery of the transcription factor atonal (atoh1) after aminoglycoside ototoxicity has previously been shown to induce the transdifferentiation of supporting cells into new hair cells and restore function. A problem with mouse aminoglycoside models is that the partial loss of hair cells seen in human disease is difficult to establish consistently. To more closely mirror human clinical balance dysfunction, we have used systemic application of 3,3′-iminodipropionitrile (IDPN), a vestibulotoxic nitrile compound known to cause vestibular hair cell loss, to induce a consistent partial loss of vestibular hair cells. To determine if balance function could be restored, we delivered atoh1 using a new adenovirus vector, based on Ad28. The Ad28 adenovector is based on a human serotype with a low seroprevalence that appears to target gene delivery to vestibular supporting cells. To further provide cell type selectivity of gene delivery, we expressed atoh1 using the supporting cell-specific glial fibrillary acid protein promoter. Delivery of this vector to IDPN-damaged vestibular organs resulted in a significant recovery of vestibular hair cells and restoration of balance, as measured by time on rotarod compared with untreated controls.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Tsuji K, Velazquez-Villasenor L, Rauch SD, Glynn RJ, Wall III C, Merchant SN . Temporal bone studies of the human peripheral vestibular system. Meniere's disease. Ann Otol Rhinol Laryngol Suppl 2000; 181: 26–31.
Kawamoto K, Izumikawa M, Beyer LA, Atkin GM, Raphael Y . Spontaneous hair cell regeneration in the mouse utricle following gentamicin ototoxicity. Hear Res 2009; 247: 17–26.
Warchol ME, Lambert PR, Goldstein BJ, Forge A, Corwin JT . Regenerative proliferation in inner ear sensory epithelia from adult guinea pigs and humans. Science 1993; 259: 1619–1622.
Zheng JL, Keller G, Gao WQ . Immunocytochemical and morphological evidence for intracellular self-repair as an important contributor to mammalian hair cell recovery. J Neurosci 1999; 19: 2161–2170.
Zajonc TP, Roland PS . Vertigo and motion sickness. Part II: pharmacologic treatment. Ear Nose Throat J 2006; 85: 25–35.
Tsuji K, Velazquez-Villasenor L, Rauch SD, Glynn RJ, Wall III C, Merchant SN . Temporal bone studies of the human peripheral vestibular system. Aminoglycoside ototoxicity. Annal Otol Rhinol Laryngol Suppl 2000; 181: 20–25.
Adler HJ, Raphael Y . New hair cells arise from supporting cell conversion in the acoustically damaged chick inner ear. Neurosci Lett 1996; 205: 17–20.
Akazawa C, Ishibashi M, Shimizu C, Nakanishi S, Kageyama R . A mammalian helix-loop-helix factor structurally related to the product of Drosophila proneural gene atonal is a positive transcriptional regulator expressed in the developing nervous system. J Biol Chem 1995; 270: 8730–8738.
Bermingham NA, Hassan BA, Price SD, Vollrath MA, Ben-Arie N, Eatock RA et al. Math1: an essential gene for the generation of inner ear hair cells. Science 1999; 284: 1837–1841.
Fritzsch B, Matei VA, Nichols DH, Bermingham N, Jones K, Beisel KW et al. Atoh1 null mice show directed afferent fiber growth to undifferentiated ear sensory epithelia followed by incomplete fiber retention. Dev Dyn 2005; 233: 570–583.
Izumikawa M, Minoda R, Kawamoto K, Abrashkin K, Swiderski DL, Dolan DF et al. Auditory hair cell replacement and hearing improvement by Atoh1 gene therapy in deaf mammals. Nat Med 2005; 11: 271–276.
Staecker H, Praetorius M, Baker K, Brough DE . Vestibular hair cell regeneration and restoration of balance function induced by math1 gene transfer. Otol Neurotol 2007; 28: 223–231.
Zheng JL, Gao WQ . Overexpression of Math1 induces robust production of extra hair cells in postnatal rat inner ears. Nat Neurosci 2000; 3: 580–586.
Oesterle EC, Campbell S, Taylor RR, Forge A, Hume CR . Sox2 and JAGGED1 expression in normal and drug-damaged adult mouse inner ear. J Assoc Res Otolaryngol 2008; 9: 65–89.
Izumikawa M, Batts SA, Miyazawa T, Swiderski DL, Raphael Y . Response of the flat cochlear epithelium to forced expression of Atoh1. Hear Res 2008; 240: 52–56.
Delay J, Pichot P, Thuillier J, Marquiset JP . Effect of aminodipropionitrile on motor behavior of the white mouse. C R Seances Soc Biol Fil 1952; 146: 533–534.
Llorens J, Dememes D, Sans A . The behavioral syndrome caused by 3,3′-iminodipropionitrile and related nitriles in the rat is associated with degeneration of the vestibular sensory hair cells. Toxicol Appl Pharmacol 1993; 123: 199–210.
Soler-Martin C, Diez-Padrisa N, Boadas-Vaello P, Llorens J . Behavioral disturbances and hair cell loss in the inner ear following nitrile exposure in mice, guinea pigs, and frogs. Toxicol Sci 2007; 96: 123–132.
Seoane A, Dememes D, Llorens J . Relationship between insult intensity and mode of hair cell loss in the vestibular system of rats exposed to 3,3′-iminodipropionitrile. J Comp Neurol 2001; 439: 385–399.
Anniko M, Thornell LE, Gustavsson H, Virtanen I . Intermediate filaments in the newborn inner ear of the mouse. ORL J Otorhinolaryngol Relat Spec 1986; 48: 98–106.
Dechesne CJ, Scarfone E, Atger P, Desmadryl G . Neurofilament proteins form an annular superstructure in guinea-pig type I vestibular hair cells. J Neurocytol 1994; 23: 631–640.
Seoane A, Dememes D, Llorens J . Distal effects in a model of proximal axonopathy: 3,3′-iminodipropionitrile causes specific loss of neurofilaments in rat vestibular afferent endings. Acta Neuropathol 2003; 106: 458–470.
Hasson T, Heintzelman MB, Santos-Sacchi J, Corey DP, Mooseker MS . Expression in cochlea and retina of myosin VIIa, the gene product defective in Usher syndrome type 1B. Proc Natl Acad Sci USA 1995; 92: 9815–9819.
Carter RJ, Morton J, Dunnett SB . Motor coordination and balance in rodents. Curr Protoc Neurosci 2001, Chapter 8, unit 8 12.
Llorens J, Dememes D . Hair cell degeneration resulting from 3,3′-iminodipropionitrile toxicity in the rat vestibular epithelia. Hear Res 1994; 76: 78–86.
Baker K, Brough DE, Staecker H . Repair of the vestibular system via adenovector delivery of Atoh1: a potential treatment for balance disorders. Adv Otorhinolaryngol 2009; 66: 52–63.
Praetorius M, Hsu C, Baker K, Brough DE, Plinkert P, Staecker H . Adenovector-mediated hair cell regeneration is affected by promoter type. Acta Otolaryngol 2009: 1–8.
Vogels R, Zuijdgeest D, van Rijnsoever R, Hartkoorn E, Damen I, de Bethune MP et al. Replication-deficient human adenovirus type 35 vectors for gene transfer and vaccination: efficient human cell infection and bypass of preexisting adenovirus immunity. J Virol 2003; 77: 8263–8271.
Kahl CA, Bonnell J, Hiriyanna S, Fultz M, Nyberg-Hoffman C, Chen P et al. Potent immune responses induced by vaccine regimens with 1 a novel adenovirus vector based on rare human serotype 28. Vaccine 2010; 28: 5691–5702.
Brough DE, Lizonova A, Hsu C, Kulesa VA, Kovesdi I . A gene transfer vector-cell line system for complete functional complementation of adenovirus early regions E1 and E4. J Virol 1996; 70: 6497–6501.
Gall JG, Lizonova A, Ettyreddy D, McVey D, Zuber M, Kovesdi I et al. Rescue and production of vaccine and therapeutic adenovirus vectors expressing inhibitory transgenes. Mol Biotechnol 2007; 35: 263–273.
Abrahamsen K, Kong HL, Mastrangeli A, Brough DE, Lizonova A, Crystal RG et al. Characterization of a replication-defective non-subgroup C adenovirus vector. J Virol 1997; 71: 8946–8951.
Praetorius M, Baker K, Brough DE, Plinkert P, Staecker H . Pharmacodynamics of adenovector distribution within the inner ear tissues of the mouse. Hear Res 2007; 227: 53–58.
Acknowledgements
This study was supported by NIDCD R01DC008424 and NICHD HD02528.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Schlecker, C., Praetorius, M., Brough, D. et al. Selective atonal gene delivery improves balance function in a mouse model of vestibular disease. Gene Ther 18, 884–890 (2011). https://doi.org/10.1038/gt.2011.33
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2011.33
Keywords
This article is cited by
-
cVEMP correlated with imbalance in a mouse model of vestibular disorder
Environmental Health and Preventive Medicine (2019)
-
Quantitative Assessment of Anti-Gravity Reflexes to Evaluate Vestibular Dysfunction in Rats
Journal of the Association for Research in Otolaryngology (2019)
-
Effects of 3,3′-Iminodipropionitrile on Hair Cell Numbers in Cristae of CBA/CaJ and C57BL/6J Mice
Journal of the Association for Research in Otolaryngology (2018)
-
Hair cell stereociliary bundle regeneration by espin gene transduction after aminoglycoside damage and hair cell induction by Notch inhibition
Gene Therapy (2016)
-
Spatial and Age-Dependent Hair Cell Generation in the Postnatal Mammalian Utricle
Molecular Neurobiology (2016)