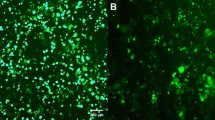
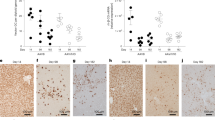
Abstract
Partial resistance of primary mouse hepatocytes to lentiviral (LV) vector transduction poses a challenge for ex vivo gene therapy protocols in models of monogenetic liver disease. We thus sought to optimize ex vivo LV gene transfer while preserving the hepatocyte integrity for subsequent transplantation into recipient animals. We found that culture media supplemented with epidermal growth factor (EGF) and, to a lesser extent, hepatocyte growth factor (HGF) markedly improved transduction efficacy at various multiplicities of infection. Up to 87% of primary hepatocytes were transduced in the presence of 10 ng EGF, compared with ∼30% in standard culture medium (SCMs). The increased number of transgene-expressing cells correlated with increased nuclear import and more integrated pro-viral copies per cell. Higher LV transduction efficacy was not associated with proliferation, as transduction capacity of gammaretroviral vectors remained low (<1%). Finally, we developed an LV transduction protocol for short-term (maximum 24 h) adherent hepatocyte cultures. LV-transduced hepatocytes showed liver repopulation capacities similar to freshly isolated hepatocytes in alb-uPA mouse recipients. Our findings highlight the importance of EGF for efficient LV transduction of primary hepatocytes in culture and should facilitate studies of LV gene transfer in mouse models of monogenetic liver disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Perera MT, Mirza DF, Elias E . Liver transplantation: Issues for the next 20 years. J Gastroenterol Hepatol 2009; 24 (Suppl 3): S124–S131.
Grossman M, Rader DJ, Muller DW, Kolansky DM, Kozarsky K, Clark 3rd BJ et al. A pilot study of ex vivo gene therapy for homozygous familial hypercholesterolaemia. Nat Med 1995; 1: 1148–1154.
Amado RG, Chen IS . Lentiviral vectors—the promise of gene therapy within reach? Science 1999; 285: 674–676.
Zychlinski D, Schambach A, Modlich U, Maetzig T, Meyer J, Grassman E et al. Physiological promoters reduce the genotoxic risk of integrating gene vectors. Mol Ther 2008; 16: 718–725.
Montini E, Cesana D, Schmidt M, Sanvito F, Bartholomae CC, Ranzani M et al. The genotoxic potential of retroviral vectors is strongly modulated by vector design and integration site selection in a mouse model of HSC gene therapy. J Clin Invest 2009; 119: 964–975.
Modlich U, Navarro S, Zychlinski D, Maetzig T, Knoess S, Brugman MH et al. Insertional transformation of hematopoietic cells by self-inactivating lentiviral and gammaretroviral vectors. Mol Ther 2009; 17: 1919–1928.
Weber A, Groyer-Picard MT, Franco D, Dagher I . Hepatocyte transplantation in animal models. Liver Transpl 2009; 15: 7–14.
Nguyen TH, Mainot S, Lainas P, Groyer-Picard MT, Franco D, Dagher I et al. Ex vivo liver-directed gene therapy for the treatment of metabolic diseases: advances in hepatocyte transplantation and retroviral vectors. Curr Gene Ther 2009; 9: 136–149.
Kerr TA, Davidson NO . Therapeutic RNA manipulation in liver disease. Hepatology 2010; 51: 1055–1061.
Nguyen TH, Oberholzer J, Birraux J, Majno P, Morel P, Trono D . Highly efficient lentiviral vector-mediated transduction of nondividing, fully reimplantable primary hepatocytes. Mol Ther 2002; 6: 199–209.
Burns JC, Friedmann T, Driever W, Burrascano M, Yee JK . Vesicular stomatitis virus G glycoprotein pseudotyped retroviral vectors: concentration to very high titer and efficient gene transfer into mammalian and nonmammalian cells. Proc Natl Acad Sci USA 1993; 90: 8033–8037.
Ohnishi N, Itoh K, Itoh Y, Baum C, Higashitsuji H, Yamaguchi K et al. High expression of transgenes mediated by hybrid retroviral vectors in hepatocytes: comparison of promoters from murine retroviruses in vitro and in vivo. Gene Therapy 2002; 9: 303–306.
Castell JV, Gomez-Lechon MJ . Liver cell culture techniques. Methods Mol Biol 2009; 481: 35–46.
Selden C, Mellor N, Rees M, Laurson J, Kirwan M, Escors D et al. Growth factors improve gene expression after lentiviral transduction in human adult and fetal hepatocytes. J Gene Med 2007; 9: 67–76.
Schaudies RP, Savage Jr CR . Isolation of rat epidermal growth factor (r-EGF): chemical, biological immunological comparisons with mouse and human EGF. Comp Biochem Physiol B 1986; 84: 497–505.
Schambach A, Bohne J, Baum C, Hermann FG, Egerer L, von Laer D et al. Woodchuck hepatitis virus post-transcriptional regulatory element deleted from X protein and promoter sequences enhances retroviral vector titer and expression. Gene Therapy 2006; 13: 641–645.
Richman RA, Claus TH, Pilkis SJ, Friedman DL . Hormonal stimulation of DNA synthesis in primary cultures of adult rat hepatocytes. Proc Natl Acad Sci USA 1976; 73: 3589–3593.
Gohda E, Tsubouchi H, Nakayama H, Hirono S, Sakiyama O, Takahashi K et al. Purification and partial characterization of hepatocyte growth factor from plasma of a patient with fulminant hepatic failure. J Clin Invest 1988; 81: 414–419.
Suzuki Y, Craigie R . The road to chromatin—nuclear entry of retroviruses. Nat Rev Microbiol 2007; 5: 187–196.
Miller DG, Adam MA, Miller AD . Gene transfer by retrovirus vectors occurs only in cells that are actively replicating at the time of infection. Mol Cell Biol 1990; 10: 4239–4242.
Mandal D, Prasad VR . Analysis of 2-LTR circle junctions of viral DNA in infected cells. Methods Mol Biol 2009; 485: 73–85.
Butler SL, Hansen MS, Bushman FD . A quantitative assay for HIV DNA integration in vivo. Nat Med 2001; 7: 631–634.
Tervo HM, Goffinet C, Keppler OT . Mouse T-cells restrict replication of human immunodeficiency virus at the level of integration. Retrovirology 2008; 5: 58.
Haridass D, Yuan Q, Becker PD, Cantz T, Iken M, Rothe M et al. Repopulation efficiencies of adult hepatocytes, fetal liver progenitor cells, and embryonic stem cell-derived hepatic cells in albumin-promoter–enhancer urokinase-type plasminogen activator mice. Am J Pathol 2009; 175: 1483–1492.
Okabe M, Ikawa M, Kominami K, Nakanishi T, Nishimune Y . ‘Green mice’ as a source of ubiquitous green cells. FEBS Lett 1997; 407: 313–319.
Korin YD, Zack JA . Progression to the G1b phase of the cell cycle is required for completion of human immunodeficiency virus type 1 reverse transcription in T cells. J Virol 1998; 72: 3161–3168.
Funke S, Maisner A, Muhlebach MD, Koehl U, Grez M, Cattaneo R et al. Targeted cell entry of lentiviral vectors. Mol Ther 2008; 16: 1427–1436.
Desfarges S, Salin B, Calmels C, Andreola ML, Parissi V, Fournier M . HIV-1 integrase trafficking in S. cerevisiae: a useful model to dissect the microtubule network involvement of viral protein nuclear import. Yeast 2009; 26: 39–54.
Buchholz CJ, Muhlebach MD, Cichutek K . Lentiviral vectors with measles virus glycoproteins—dream team for gene transfer? Trends Biotechnol 2009; 27: 259–265.
Lo HW, Ali-Seyed M, Wu Y, Bartholomeusz G, Hsu SC, Hung MC . Nuclear–cytoplasmic transport of EGFR involves receptor endocytosis, importin beta1 and CRM1. J Cell Biochem 2006; 98: 1570–1583.
Wang YN, Yamaguchi H, Hsu JM, Hung MC . Nuclear trafficking of the epidermal growth factor receptor family membrane proteins. Oncogene 2010; 29: 3997–4006.
O’Neill LS, Skinner AM, Woodward JA, Kurre P . Entry kinetics and cell–cell transmission of surface-bound retroviral vector particles. J Gene Med 2010; 12: 463–476.
Schambach A, Bohne J, Chandra S, Will E, Margison GP, Williams DA et al. Equal potency of gammaretroviral and lentiviral SIN vectors for expression of O6-methylguanine-DNA methyltransferase in hematopoietic cells. Mol Ther 2006; 13: 391–400.
Dull T, Zufferey R, Kelly M, Mandel RJ, Nguyen M, Trono D et al. A third-generation lentivirus vector with a conditional packaging system. J Virol 1998; 72: 8463–8471.
Schambach A, Mueller D, Galla M, Verstegen MM, Wagemaker G, Loew R et al. Overcoming promoter competition in packaging cells improves production of self-inactivating retroviral vectors. Gene Therapy 2006; 13: 1524–1533.
Kutner RH, Zhang XY, Reiser J . Production, concentration and titration of pseudotyped HIV-1-based lentiviral vectors. Nat Protoc 2009; 4: 495–505.
Seglen PO . Hepatocyte suspensions and cultures as tools in experimental carcinogenesis. J Toxicol Environ Health 1979; 5: 551–560.
Sandgren EP, Palmiter RD, Heckel JL, Daugherty CC, Brinster RL, Degen JL . Complete hepatic regeneration after somatic deletion of an albumin-plasminogen activator transgene. Cell 1991; 66: 245–256.
Morosan S, Hez-Deroubaix S, Lunel F, Renia L, Giannini C, Van Rooijen N et al. Liver-stage development of Plasmodium falciparum, in a humanized mouse model. J Infect Dis 2006; 193: 996–1004.
Silva-Barbosa SD, Butler-Browne GS, Di Santo JP, Mouly V . Comparative analysis of genetically engineered immunodeficient mouse strains as recipients for human myoblast transplantation. Cell Transplant 2005; 14: 457–467.
Acknowledgements
We thank Sabine Knöß and Johanna Krause for excellent technical help, and Qinggong Yuan for help with transplantation of cells (all from Hannover Medical School, Hannover, Germany). The LV ID gag/pol construct (pcDNA3.GP.4 × CTE with D64V mutation) was provided by M Milsom (Boston, MA, USA, and DKFZ, Heidelberg, Germany). The study was supported by grants from the Deutsche Forschungsgemeinschaft (SFB 738, SPP 1230 and the Excellence Cluster REBIRTH). The work was supported by the SFB 738 ‘Improvement of conventional and innovative transplants’ (MO, UM), the Cluster of Excellence ‘Rebirth’ (MO) and the Grand Challenge Program of the Bill and Melinda Gates Foundation (MO).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Gene Therapy website
Supplementary information
Rights and permissions
About this article
Cite this article
Rothe, M., Rittelmeyer, I., Iken, M. et al. Epidermal growth factor improves lentivirus vector gene transfer into primary mouse hepatocytes. Gene Ther 19, 425–434 (2012). https://doi.org/10.1038/gt.2011.117
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2011.117
Keywords
This article is cited by
-
Lentiviral gene therapy and vitamin B3 treatment enable granulocytic differentiation of G6PC3-deficient induced pluripotent stem cells
Gene Therapy (2020)
-
Hepatitis B virus X protein identifies the Smc5/6 complex as a host restriction factor
Nature (2016)
-
Generation of lentivirus-induced dendritic cells under GMP-compliant conditions for adaptive immune reconstitution against cytomegalovirus after stem cell transplantation
Journal of Translational Medicine (2015)
-
Lentivirus-induced ‘Smart’ dendritic cells: Pharmacodynamics and GMP-compliant production for immunotherapy against TRP2-positive melanoma
Gene Therapy (2015)
-
RGB marking with lentiviral vectors for multicolor clonal cell tracking
Nature Protocols (2012)