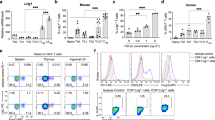
Abstract
Recent insight into the balance of self-tolerance and auto-aggression has raised interest in using human regulatory T (Treg) cells for adoptive immunotherapy of unlimited autoimmune diseases including type-1 diabetes, rhematoid arthritis and multiple sclerosis. The therapeutic use of Treg cells, however, is so far hampered by the inefficiency of current protocols in making them accessible for genetic manipulations. We report here that TCR/CD3 stimulation that is accompanied by extensive CD28 costimulation makes human Treg cells susceptible to retroviral gene transfer ex vivo while preserving their properties in vitro and in vivo. To show the power of genetic manipulation of human Treg cells, we engineered ‘designer Treg cells’ by retroviral expression of a chimeric immunoreceptor with defined specificity, which activates Treg cells in a ligand-dependent manner to proliferate, to secrete high amounts of interleukin-10 and to repress an ongoing cytolytic T-cell response in vivo. The procedure in genetically modifying human Treg cells ex vivo will open a panel of applications for their use in the adoptive therapy of deregulated immune responses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Thornton AM, Shevach EM . CD4+CD25+ immunoregulatory T cells suppress polyclonal T cell activation in vitro by inhibiting interleukin 2 production. J Exp Med 1998; 188: 287–296.
Suri-Payer E, Amar AZ, Thornton AM, Shevach EM . CD4+CD25+ T cells inhibit both the induction and effector function of autoreactive T cells and represent a unique lineage of immunoregulatory cells. J Immunol 1998; 160: 1212–1218.
Kukreja A, Cost G, Marker J, Zhang C, Sun Z, Lin-Su K et al. Multiple immuno-regulatory defects in type-1 diabetes. J Clin Invest 2002; 109: 131–140.
Viglietta V, Baecher-Allan C, Weiner HL, Hafler DA . Loss of functional suppression by CD4+CD25+ regulatory T cells in patients with multiple sclerosis. J Exp Med 2004; 199: 971–979.
Ehrenstein MR, Evans JG, Singh A, Moore S, Warnes G, Isenberg DA et al. Compromised function of regulatory T cells in rheumatoid arthritis and reversal by anti-TNFalpha therapy. J Exp Med 2004; 200: 277–285.
Wood KJ, Sakaguchi S . Regulatory T cells in transplantation tolerance. Nat Rev Immunol 2003; 3: 199–210.
Yamazaki S, Iyoda T, Tarbell K, Olson K, Velinzon K, Inaba K et al. Direct expansion of functional CD25+ CD4+ regulatory T cells by antigen-processing dendritic cells. J Exp Med 2003; 198: 235–247.
Setoguchi R, Hori S, Takahashi T, Sakaguchi S . Homeostatic maintenance of natural Foxp3(+) CD25(+) CD4(+) regulatory T cells by interleukin (IL)-2 and induction of autoimmune disease by IL-2 neutralization. J Exp Med 2005; 201: 723–735.
Hoffmann P, Eder R, Kunz-Schughart LA, Andreesen R, Edinger M . Large-scale in vitro expansion of polyclonal human CD4(+)CD25high regulatory T cells. Blood 2004; 104: 895–903.
Levings MK, Sangregorio R, Roncarolo MG . Human cd25(+)cd4(+) t regulatory cells suppress naive and memory T cell proliferation and can be expanded in vitro without loss of function. J Exp Med 2001; 193: 1295–1302.
Hoffmann P, Eder R, Boeld TJ, Doser K, Piseshka B, Andreesen R et al. Only the CD45RA+ subpopulation of CD4+CD25high T cells gives rise to homogeneous regulatory T-cell lines upon in vitro expansion. Blood 2006; 108: 4260–4267.
Godfrey WR, Ge YG, Spoden DJ, Levine BL, June CH, Blazar BR et al. In vitro-expanded human CD4(+)CD25(+) T-regulatory cells can markedly inhibit allogeneic dendritic cell-stimulated MLR cultures. Blood 2004; 104: 453–461.
Taams LS, Smith J, Rustin MH, Salmon M, Poulter LW, Akbar AN . Human anergic/suppressive CD4(+)CD25(+) T cells: a highly differentiated and apoptosis-prone population. Eur J Immunol 2001; 31: 1122–1131.
Hsieh CS, Liang Y, Tyznik AJ, Self SG, Liggitt D, Rudensky AY . Recognition of the peripheral self by naturally arising CD25+ CD4+ T cell receptors. Immunity 2004; 21: 267–277.
Hombach AA, Kofler D, Hombach A, Rappl G, Abken H . Effective proliferation of human regulatory T cells requires a strong costimulatory CD28 signal that cannot be substituted by IL-2. J Immunol 2007; 179: 7924–7931.
Greenstein AJ, Panvelliwalla DK, Katz LB, Heimann TM, Donnelly J, Pertsimlidis D et al. Tissue carcinoembryonic antigen, dysplasia, and disease duration in colonic inflammatory bowel disease. Am J Gastroenterol 1982; 77: 212–215.
Fischbach W, Mossner J, Seyschab H, Hohn H . Tissue carcinoembryonic antigen and DNA aneuploidy in precancerous and cancerous colorectal lesions. Cancer 1990; 65: 1820–1824.
Maul J, Loddenkemper C, Mundt P, Berg E, Giese T, Stallmach A et al. Peripheral and intestinal regulatory CD4+CD25(high) T cells in inflammatory bowel disease. Gastroenterology 2005; 128: 1868–1878.
Elinav E, Waks T, Eshhar Z . Redirection of regulatory T cells with predetermined specificity for the treatment of experimental colitis in mice. Gastroenterology 2008; 134: 2014–2024.
Weijtens ME, Willemsen RA, Hart EH, Bolhuis RL . A retroviral vector system ‘STITCH’ in combination with an optimized single chain antibody chimeric receptor gene structure allows efficient gene transduction and expression in human T lymphocytes. Gene Therapy 1998; 5: 1195–1203.
Schwegler C, Dorn-Beineke A, Nittka S, Stocking C, Neumaier M . Monoclonal anti-idiotype antibody 6G6.C4 fused to GM-CSF is capable of breaking tolerance to carcinoembryonic antigen (CEA) in CEA-transgenic mice. Cancer Res 2005; 65: 1925–1933.
Pohl C, Denfeld R, Renner C, Jung W, Bohlen H, Sahin U et al. CD30-antigen-specific targeting and activation of T cells via murine bispecific monoclonal antibodies against CD3 and CD28: potential use for the treatment of Hodgkin's lymphoma. Int J Cancer 1993; 54: 820–827.
Hombach A, Wieczarkowiecz A, Marquardt T, Heuser C, Usai L, Pohl C et al. Tumor-specific T cell activation by recombinant immunoreceptors: CD3 zeta signaling and CD28 costimulation are simultaneously required for efficient IL-2 secretion and can be integrated into one combined CD28/CD3 zeta signaling receptor molecule. J Immunol 2001; 167: 6123–6131.
Willemsen RA, Weijtens ME, Ronteltap C, Eshhar Z, Gratama JW, Chames P et al. Grafting primary human T lymphocytes with cancer-specific chimeric single chain and two chain TCR. Gene Therapy 2000; 7: 1369–1377.
Hombach A, Schneider C, Sent D, Koch D, Willemsen RA, Diehl V et al. An entirely humanized CD3 zeta-chain signaling receptor that directs peripheral blood t cells to specific lysis of carcinoembryonic antigen-positive tumor cells. Int J Cancer 2000; 88: 115–120.
Hombach A, Sent D, Schneider C, Heuser C, Koch D, Pohl C et al. T-cell activation by recombinant receptors: CD28 costimulation is required for interleukin 2 secretion and receptor-mediated T-cell proliferation but does not affect receptor-mediated target cell lysis. Cancer Res 2001; 61: 1976–1982.
Weijtens ME, Hart EH, Bolhuis RL . Functional balance between T cell chimeric receptor density and tumor associated antigen density: CTL mediated cytolysis and lymphokine production. Gene Therapy 2000; 7: 35–42.
Angulo R, Fulcher DA . Measurement of Candida-specific blastogenesis: comparison of carboxyfluorescein succinimidyl ester labelling of T cells, thymidine incorporation, and CD69 expression. Cytometry 1998; 34: 143–151.
Acknowledgements
We thank Birgit Hops, Frank Steiger and Petra Hofmann for technical assistance. This study was supported by the European Commission through the ATTACK project and the Fortune program of the Medical Faculty of the University of Cologne.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hombach, A., Kofler, D., Rappl, G. et al. Redirecting human CD4+CD25+ regulatory T cells from the peripheral blood with pre-defined target specificity. Gene Ther 16, 1088–1096 (2009). https://doi.org/10.1038/gt.2009.75
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2009.75
Keywords
This article is cited by
-
Treg cell-based therapies: challenges and perspectives
Nature Reviews Immunology (2020)
-
Stem cell-derived tissue-associated regulatory T cells ameliorate the development of autoimmunity
Scientific Reports (2016)
-
Cell Mediators of Autoimmune Hepatitis and Their Therapeutic Implications
Digestive Diseases and Sciences (2015)
-
Cytotoxic response of human regulatory T cells upon T-cell receptor-mediated activation: a matter of purity
Blood Cancer Journal (2014)
-
CAR/FoxP3-engineered T regulatory cells target the CNS and suppress EAE upon intranasal delivery
Journal of Neuroinflammation (2012)