Abstract
Purpose
Within the spectrum of the Ehlers-Danlos syndromes (EDS), vascular complications are usually associated with the vascular subtype of EDS. Vascular complications are also observed in other EDS subtypes, but the reports are anecdotal and the information is dispersed. To better document the nature of vascular complications among “nonvascular” EDS subtypes, we performed a systematic review.
Methods
We queried three databases for English-language studies from inception until May 2017, documenting both phenotypes and genotypes of patients with nonvascular EDS subtypes. The outcome included the number and nature of vascular complications.
Results
A total of 112 papers were included and data were collected from 467 patients, of whom 77 presented with a vascular phenotype. Severe complications included mainly hematomas (53%), frequently reported in musculocontractural and classical-like EDS; intracranial hemorrhages (18%), with a high risk in dermatosparaxis EDS; and arterial dissections (16%), frequently reported in kyphoscoliotic and classical EDS. Other, more minor, vascular complications were reported in cardiac-valvular, arthrochalasia, spondylodysplastic, and periodontal EDS.
Conclusion
Potentially life-threatening vascular complications are a rare but important finding in several nonvascular EDS subtypes, highlighting a need for more systematic documentation. This review will help familiarize clinicians with the spectrum of vascular complications in EDS and guide follow-up and management.
Similar content being viewed by others
Introduction
The Ehlers-Danlos syndrome (EDS) is an umbrella term for a group of clinically and genetically heterogeneous connective tissue disorders. Over the past two decades the Villefranche Nosology has been the standard for classifying EDS. It recognized six subtypes, most of which were caused by defects in the primary structure of collagen or collagen-modifying enzymes.1 Recent discoveries have, however, expanded the pathogenic spectrum to include EDS variants that are caused by defects in both noncollagenous extracellular matrix proteins and intracellular processes.2, 3, 4, 5, 6, 7, 8, 9, 10 This has led to an EDS reclassification: a task that was recently accomplished by an international EDS consortium.11
Skin hyperextensibility and joint hypermobility are the clinical hallmarks of EDS, but more variable signs of soft connective tissue fragility are helpful in discriminating between the different types. Historically, arterial aneurysm and dissection have been synonymous with the vascular type of EDS (vEDS). This type of EDS is characterized by the presence of a thin, translucent skin, which bruises very easily, and joint hypermobility, which is often confined to the small joints. The clinical picture is, however, dominated by a remarkable vascular fragility that leads to spontaneous rupture of blood vessel walls, often without preceding vascular dilatation or aneurysm formation. Other life-threatening complications include rupture of the gastrointestinal (GI) tract, gravid uterus, or other internal organs, such as liver or spleen.12 The calculated median survival for vEDS patients is 48 years, with most deaths resulting from arterial rupture.13 Complications are rare in childhood, but 25% will have a first complication by the age of 20 years, and more than 80% will have had at least one complication by the age of 40 years. Therapeutic interventions are limited to symptomatic measures.14 Hitherto, the only evidence-based treatment strategy has been the administration of celiprolol, a cardioselective β-blocker with β2 agonist vasodilatory properties, which has been reported to reduce heart rate and pulsatile pressures in essential hypertension and could therefore decrease the continuous and pulsatile mechanical stress on collagen fibers within the arterial wall.15, 16 vEDS is caused by heterozygous mutations in the type III procollagen-encoding gene COL3A1.17 Genotype–phenotype correlations have been extensively investigated. Substitution of triple helical glycine residues and splice donor site mutations, leading to exon skipping, are generally associated with a shorter life expectancy, whereas mutations leading to COL3A1 haploinsufficiency are usually associated with a milder phenotype, a delay in the onset of complications, and a longer life expectancy.18, 19
Vascular complications, including arterial aneurysms and ruptures, subcutaneous hematomas, gum bleeding, and prolonged perioperative and menstrual bleeding, have also been described in other, “nonvascular” subtypes of EDS.20, 21 Most of these reports are anecdotal, and the occurrence of such complications in the different EDS subtypes is not well documented. In view of the vast clinical and genetic heterogeneity of EDS, it may therefore be difficult for clinicians to predict, for a specific EDS patient, whether vascular complications should be taken into account, and how patients should be followed.
We reviewed the medical literature on EDS in a systematic manner to better document the nature of vascular complications in patients with a nonvascular EDS diagnosis confirmed via molecular testing. This review will help familiarize clinicians with the spectrum of vascular complications in nonvascular EDS subtypes as well as guide follow-up and management.
Materials and methods
Study design
This systematic review was designed and carried out in accordance with the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement for reporting systematic reviews.22
Search strategy
The aim of this study was to collect all available data on vascular complications in the nonvascular EDS subtypes described in the updated EDS classification (Table 1). We queried the PubMed and Web of Science databases, taking into account the different notations for the EDS subtypes (e.g., classic versus classical). For classical EDS (COL5A1/2), for example, the following keywords were used: Ehlers-Danlos syndrome, classic*, COL5A1, COL5A2, Ehlers-Danlos syndrome type I, Ehlers-Danlos syndrome type II. All keywords for each EDS subtype are outlined in Table 1. Based on these keywords, search strings to query the respected databases for, for example, classical EDS (COL5A1/2) were constructed as follows: (“Ehlers-Danlos syndrome”[Title/Abstract] AND (classic*[Title/Abstract] OR COL5A1[Title/Abstract] OR COL5A2[Title/Abstract])) OR “Ehlers-Danlos syndrome type I”[Title/Abstract] OR “Ehlers-Danlos syndrome type II”[Title/Abstract]. To exclude nonrelevant references, queries were restricted to “title” and “abstract” in PubMed and “topic” in Web of Science. All search strings are outlined in Supplementary Table S1 online. The Leiden Open Variation Database (http://www.lovd.nl/3.0/home) was queried for additional references. All references published from inception until 31 May, 2017 were eligible for inclusion in this review. A bibliography was created using EndNote X7 (Thomson Reuters, New York, NY).
Screening process and eligibility criteria
Primary literature screening was performed by two investigators (S.D. and T.V.D.), independently, according to the following inclusion criteria: (i) population: nonvascular subtypes of EDS, (ii) language: English, (iii) papers: no short conference proceedings or meeting abstracts, and (iv) availability of the full text. Next, eligibility for inclusion in the review after full-text screening of the remaining papers was further assessed using the following criteria: descriptions of both (i) the patient’s phenotype and (ii) the pathogenic genetic defect. Patients for whom only linkage to a gene or a biochemical diagnosis was demonstrated were excluded. Hypermobile EDS was also not included, since its genetic etiology remains unknown and because—prior to the updated 2017 classification on EDS11—its definition covered a broad clinical spectrum, with variable signs of connective tissue fragility that closely overlapped with joint hypermobility syndrome. The following types of studies were considered: clinical trials, case-control studies, cross-sectional studies, cohort studies, case series, and case reports published in peer-reviewed scientific journals. Excluded were cell culture laboratory studies, animal studies, and reviews. Titles and abstracts were checked with regard to the predefined eligibility criteria. Abstracts with unclear methodology were included in full-text assessment to avoid exclusion of potentially relevant papers.
Quality assessment
Quality assessment tools for case series, case-control, cross-sectional, and cohort studies are available from the National Heart, Blood, and Lung Institute (Bethesda, MD) (https://www.nhlbi.nih.gov/). Quality assessment tools for case reports are available from the Joanna Briggs Institute (Adelaide, Australia) (http://joannabriggs.org/). Each study was classified into one of the following groups: (i) good if all quality criteria were judged as “present,” (ii) fair if one or more key domains were “unclear,” or (iii) poor if one or more key domains were “absent” (Supplementary Table S2).
Data extraction and aggregation
Data extraction and aggregation were performed by a single investigator (S.D.). Uncertainties were resolved through discussion with the principal investigator (F.M.). The following data, if available, were extracted from the included references: (i) study characteristics (authors and year of publication), (ii) patient attributes (patient and family identifiers, age at time of referral or vascular complication, and relevant comorbidities), (iii) vascular features (type and location of vascular complication, recurrence, management, and cause of death), and (iv) mutation. If a patient or family was described more than once, the most informative reference was used for data collection. The spectrum of vascular complications was further categorized into (i) hematomas, (ii) intracranial hemorrhages, (iii) arterial dissections, (iv) arterial aneurysms, (v) GI bleedings, (vi) perioperative hemorrhages, and (vii) sporadic vascular complications. The data collection on hematomas included only those that occurred spontaneously or from minor trauma or were described as severe in nature (e.g., “large,” “massive,” “repeated,” “profuse”). Easy bruising was not included in this study for many reasons. It is often described in a nondescriptive manner, and as a symptom it is often overreported by both patients and caregivers. We therefore felt that including easy bruising would lead to an overrepresentation of vascular complications in nonvascular EDS. The primary outcome of these data-processing procedures was the derivation of the total number of nonvascular EDS patients, along with the number of patients reported with, respectively, none, one, or more than one vascular complication. The secondary outcome included the number, type, and location of reported vascular complications.
Results
Search results and study characteristics
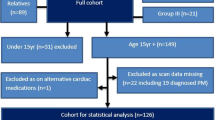
A flow diagram of the search selection process is depicted in Figure 1. Our search strategy identified 809 unique papers, 547 of which were excluded after primary screening based on title and abstract. From the remaining 262 papers selected for full-text screening, 112 papers were found to meet the predefined inclusion criteria (Figure 1). All included papers are listed in Supplementary Table S2 and a brief overview of all included studies and patients is presented in Table 2. The types of studies reported by these papers included mainly case reports (n = 51) and series (n = 59), a cross-sectional study (n = 1), and a cohort study (n = 1). There was a quality assessment of the included papers, of which 38 were classified as good, 43 as fair, and 31 as poor. None of them were rejected based on assessed quality alone, so that a large population could be obtained, based on which it is possible to draw firm conclusions. These 112 papers report on 467 patients (197 males, 238 females, and 32 not defined) from 342 unrelated families, and include 29 papers on classical EDS (cEDS; COL5A1/2: n = 25; COL1A1 p.(Arg312Cys): n=4),23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51 6 on classical-like EDS (clEDS; TNXB),52, 53, 54, 55, 56, 57 three on cardiac-valvular EDS (cvEDS; COL1A2),58, 59, 60 ten on arthrochalasia EDS (aEDS; COL1A1/2),61, 62, 63, 64, 65, 66, 67, 68, 69, 70 six on dermatosparaxis EDS (dEDS; ADAMTS2),71, 72, 73, 74, 75, 76 22 on kyphoscoliotic EDS (kEDS; PLOD1: n = 17, FKBP14: n = 5),4, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97 ten on brittle cornea syndrome (BCS; ZNF469: n = 5; PRDM5: n=5),2, 98, 99, 100, 101, 102, 103, 104, 105, 106 11 on spondylodysplastic EDS (spEDS; B4GALT7: n = 5; B3GALT6: n = 4; SLC39A13: n = 2),5, 8, 9, 107, 108, 109, 110, 111, 112, 113, 114 12 on musculocontractural EDS (mcEDS; CHST14: n = 11; DSE: n = 1),6, 7, 115, 116, 117, 118, 119, 120, 121, 122, 123, 124 two on myopathic EDS (mEDS; COL12A1),125, 126 and one on periodontal EDS (pEDS; C1R/S).127 One paper that fulfilled the inclusion criteria was withheld because it contained data for a patient described more thoroughly in another paper.128
Nonvascular EDS with vascular complications
Seventy-seven of 467 (17%) individuals with nonvascular EDS (age: ranging from birth to 62 years) were reported with a total number of 100 vascular complications. Vascular complications were, in terms of percentage, most frequently reported in mcEDS-DSE (2/3, 67%), mcEDS-CHST14 (27/43, 63%), clEDS (10/19, 53%), cvEDS (2/5, 40%), dEDS (5/15, 33%), cEDS-COL1A1 (3/12, 25%), kEDS-FKBP14 (2/10, 20%), kEDS-PLOD1 (8/54, 15%), spEDS-SLC39A13 (1/8, 13%), cEDS-COL5A1/2 (12/110, 11%), aEDS (1/17, 6%), pEDS (3/55, 5%), and spEDS-B3GALT6 (1/25, 4%) (Figure 2a). Of these 77 individuals, 14 (18%) suffered more than one complication, with an average of 1.3, ranging from one to six complications per case. This, however, corresponds to only 3% (14/467) of all individuals included in this systematic review. The occurrence of multiple complications was reported most frequently in mcEDS-CHST14 (8/43, 19%) (Table 3). No vascular complications were recorded in spEDS-B4GALT7, BCS and mEDS.
Vascular complications in nonvascular EDS. (a) The number of patients with vascular complications is presented for each nonvascular EDS subtype in terms of percentage. The ratios relate to the total number of patients with vascular complication(s), to the total number per subtype. (b) The number of each type of complication is presented in terms of percentage. EDS, Ehlers-Danlos syndrome (for the definitions of the various subtypes of EDS, see Table 1); GI, gastrointestinal.
Mortality
Overall, death due to vascular complications was reported in eight individuals (8/467, 2%). In cEDS-COL5A1, three adult patients died from rupture of a large or medium-sized artery (mean age: 35 years, range 28–43 years)25 and one 9-year old patient died from multiorgan failure secondary to the rupture of an aneurysm of the superior mesenteric artery.31 One patient with cvEDS died from bleeding complications during aortic valve replacement surgery at the age of 45 years.60 In dEDS, one patient died shortly after birth from severe hemorrhage and shock.72 One patient with mcEDS-CHST14 died from a large intracerebral hemorrhage at the age of 59 years, and, finally, one with kEDS-PLOD1 died from an arterial rupture at an unspecified site at an unknown age.89, 116
Vascular phenotypes and management
To provide an overview of the type of vascular complications in nonvascular EDS patients, each reported complication (n = 100) was categorized as either (i) hematoma (53/100, 53%), (ii) intracranial hemorrhage (18/100, 18%), (iii) spontaneous arterial dissection (16/100, 16%), (iv) arterial aneurysm (5/100, 5%), (v) GI bleeding (1/100, 1%), (vi) perioperative hemorrhage (5/100 5%), or (vii) sporadic vascular complication (2/100, 2%). An overview of the vascular phenotypes is presented in Figure 2b, and Table 3 summarizes the reported type of complications per nonvascular EDS subtype.
Hematoma
The most frequent of all vascular complications was the formation of hematomas (53/100, 53%), either spontaneously (10/53, 19%) or after minor trauma (32/53, 60%), such as a minor fall. They were reported primarily in mcEDS patients (25/46, 54%) (DSE: 2/3, 67%; CHST14: 23/43, 53%)6, 115, 116, 117, 118, 119, 120, 121, 122 and clEDS patients (10/19, 53%),53, 55, 56, 57 and, to a lesser extent, in patients with dEDS (2/15, 13%)72, 74 and cEDS-COL5A1 (3/110, 3%).34, 38 Most hematomas were subcutaneous (41/53, 77%), but epidural, spinal, scalp, and stomach wall hematomas were reported as well. Management was reported only in mcEDS, where some hematomas from minor trauma required transfusion (n = 7), surgical drainage (n = 6), and/or admittance to the intensive care unit (n = 3). One mcEDS-CHST14 patient presented with a spontaneous hematoma and was treated with emergency surgical drainage and transfusion.115
Intracranial hemorrhage
The second most frequently reported vascular complication was intracranial hemorrhage (18/100, 18%), including 10 intracerebral, three subdural, one subarachnoid, one epidural, and three unspecified hemorrhages. dEDS denoted a high risk of intracerebral hemorrhage, affecting 20% of the patients (3/15), mostly at birth.73, 76 Intracranial hemorrhages were also reported in mcEDS-CHST14 (4/43, 9%),116, 118, 120 kEDS-PLOD1 (4/54, 7%),78, 80, 86, 87 pEDS (2/55, 4%),127 spEDS (2/61, 3%) (SLC39A13: 1/8, 13%; B3GALT6: 1/25, 4%),5, 9 and cEDS-COL5A1 (1/110, 1%).25
Arterial dissection
Overall, 13 patients suffered a total of 16 arterial dissections (16/100, 16%): eight patients with cEDS (8/122, 7%) (COL1A1: 2/12, 17%; COL5A1: 6/110, 5%),25, 27, 33, 49, 50 seven patients with kEDS (7/64, 11%) (PLOD1: 5/54, 9%; FKBP14: 2/10, 20%),79, 80, 89, 95, 97 and one with cvEDS (1/5, 20%).60 Dissections occurred most frequently in medium-sized or large arteries, including the iliac, femoral, renal, celiac, hypogastric, subclavian, superior mesenteric, brachial, and coronary arteries. Aortic dissection was reported in one patient with cEDS-COL5A1.25 Data about management is limited and mostly anecdotal, but seven of these dissections were treated surgically and two with endovascular techniques.25, 27, 49, 79, 95, 97
Arterial aneurysm
Arterial aneurysms were reported in five patients (5/100, 5%), including cEDS (4/122, 3%; COL5A1: 3/110, 3%; COL1A1: 1/12, 8%) and kEDS-PLOD1 (1/54, 2%).25, 28, 30, 31, 82 Large (n = 1) and medium-sized (n = 3) arteries were affected and aortic root dilatation was described in one cEDS patient.28 Three aneurysms were reported as having ruptured, two in connection with cEDS-COL5A1 and one with kEDS-PLOD1; they were managed using endovascular techniques.30, 31, 82 One patient underwent surgery twice and was followed up using regular computed tomography angiography.25
Gastrointestinal bleeding
Severe GI bleeding was reported in one cEDS-COL5A2 (1/110, 1%) patient, who suffered a perforation of the terminal ileum at birth and was treated with an ileostomy.23 Several more common GI problems are described below.
Perioperative hemorrhage
Perioperative hemorrhage (5/100, 5%) was reported in two patients with mcEDS-CHST14 (2/43, 5%) during a laparoscopic procedure and surgery for dislocations,63, 118, 123 in one patient with aEDS (1/18, 6%) who bled excessively at surgery (unspecified),63 in one patient with pEDS (1/55, 2%) who had a profuse bleeding after hysterectomy,127 and in one cvEDS patient (1/5, 20%), who underwent aortic valve replacement surgery and died from it at the age of 45 years.60
Sporadic vascular complications
One cEDS-COL5A1 patient (1/110, 1%) was reported with a pulmonary artery hypoplasia,28 and one dEDS patient (1/15, 7%) presented with a pleural serohemorrhagic effusion of the left lung.74
Common and aspecific vascular features
Several vascular and bleeding complications, which are presumed to be relatively frequent in the general population, have been reported in various EDS subtypes. Since these vascular features are potentially aspecific, we listed them as complementary but did not add them to the total number of complications.
Menometrorrhagia was reported in two patients with cEDS-COL5A1 (2/65 females, 3%)28 and postpartum hemorrhaging was reported in one patient with aEDS (1/12 females, 8%) after the birth of each of her children and in one clEDS (1/12 females, 8%).53, 65 The severity of the latter was not described.
Minor GI bleeding was reported in two clEDS (2/18, 11%),53, 57 two dEDS (2/15, 13%),71 two mcEDS-CHST14 (2/43, 5%),115 and one pEDS (1/55, 2%) patients.127 Two dEDS patients suffered rectal prolapse with anal bleeding,71 and one clEDS patient had a gastric ulcer.53 The origin of the bleeding was not specified in the other clEDS and the pEDS patient.
Venous complications such as varicose veins and deep venous thrombosis (DVT) were reported in a number of patients. In cEDS, seven patients presented with varicose veins (7/122, 6%; COL5A1: 4/110, 4%; COL1A1: 3/12, 25%) and three with a DVT (3/110, 3%).28, 33, 42, 44, 48 Two patients with spEDS-SLC39A13 (2/8, 25%) presented with varicose veins,5 and one patient with kEDS-PLOD1 (1/54, 2%) presented with a DVT from compression stasis, which was treated by fasciotomy.93 All of these patients were over the age of 40 years, except for the kEDS-PLOD1 patient, who presented with a DVT at the age of 15 years.93
Five patients were reported with small bleedings: three of these suffered from gum bleeding (mcEDS-CHST14: 2/43, 5%; cEDS-COL5A1: 1/110, 1%),28, 118, 123 and two dEDS patients suffered from epistaxis (2/15, 13%).71, 129
Discussion
Vascular complications are an important finding in nonvascular subtypes of EDS. Overall, 77/467 (17%) of the patients included in this study presented with relatively severe vascular complications. In line with the clinical and genetic heterogeneity of this group of disorders, there are important differences in the number, severity, and type of complications associated with the different EDS subtypes (Table 3). Vascular complications, for example, are most frequently reported in mcEDS (CHST14/DSE) and in clEDS (TNXB), being present in about two thirds and half of the patients, respectively. In contrast, to date no vascular complications have been reported for spEDS-B4GALT7, BCS, and mEDS.
In mcEDS and clEDS, the vascular phenotype shows a large predominance of hematomas. In clEDS, hematomas are mostly spontaneous and subcutaneous, whereas in mcEDS they mostly occur after minor trauma, and also affect other locations besides subcutaneous tissues (e.g., scalp, spine, and buttock). Furthermore, these hematomas are different from the commonly reported easy bruising in EDS and the large hematomas in clEDS, because they can be severe in nature and sometimes necessitate surgery and blood transfusion. Of note, four mcEDS-CHST14 patients were reported to have intracranial bleeding, of which one person died at the age of 59 years. Therefore, this appears to be a rare but important complication of mcEDS.
cEDS, cvEDS, dEDS, and kEDS are associated with a lower, but nonetheless important risk of vascular complications. Moreover, in these subtypes, the majority of vascular complications are severe and potentially life-threatening, and include intracranial hemorrhages, arterial aneurysms, and arterial dissections. The latter complications each account for approximately 5–16% of all reported vascular complications in nonvascular EDS. Arterial aneurysms have been reported in a few patients with cEDS (COL5A1/COL1A1) and kEDS-PLOD1. Arterial dissections are most frequently reported in kEDS-FKBP14 (20% of 10 kEDS-FKBP14 patients) and cEDS due to COL1A1 p.(Arg312Cys) (17% of 12 cEDS-COL1A1 patients), and to a lesser extent in kEDS-PLOD1 (9% of 54 kEDS-PLOD1 patients) and cEDS-COL5A1 (5% of 110 cEDS-COL5A1 patients). One patient with cvEDS was also reported to have an arterial dissection. As in vEDS, these aneurysms and dissections mostly affect large and medium-sized arteries, such as the iliac, femoral, renal, celiac, hypogastric, subclavian, superior mesenteric, brachial, and coronary arteries. Aortic dissection was reported in only one patient with cEDS-COL5A1. Arterial aneurysms and dissections have not been reported for any of the other EDS subtypes. Overall, arterial aneurysms and dissections are significantly less frequently reported than in vEDS, where such complications are reported in approximately 50% of patients.18, 130
In dEDS, 25% of the patients suffered from intracranial hemorrhage, especially in the perinatal period. Intracranial hemorrhage was also reported in a range of other subtypes, including kEDS-PLOD1 (also in the perinatal period), mcEDS-CHST14, pEDS, spEDS (SLC39A13 and B3GALT6), and cEDS-COL5A1 (at ages ranging from 2 to 62 years). Despite a relatively high occurrence of these severe vascular complications, reported mortality was only 2% (8/467).
In addition to these severe and potentially life-threatening problems, several other, and often minor, bleeding manifestations, such as gynecological and GI bleedings, perioperative hemorrhage, varicose veins, DVT, gum bleeding, and epistaxis have been reported in a variety of EDS subtypes, including cEDS, clEDS, aEDS, dEDS, kEDS-PLOD1, spEDS-B3GALT6, mcEDS-CHST14, and pEDS. However, it should be noted that these bleeding manifestations are also relatively common in the general population: e.g., the estimated incidence for menometrorrhagia is 10–20%,131 3–10% for perioperative hemorrhage,132 10–15% and 20–25% for varicose veins in men and women respectively.133 As such, these manifestations were not counted toward the total number of vascular complications, but were merely listed as complementary in this review.
Data on treatment and treatment outcome of arterial complications in nonvascular EDS are very limited, being reported in only 11 of 77 (14%) patients with complications. In the case of arterial aneurysms and dissections, these patients underwent endovascular stenting (n = 5), classical surgery (n = 4), or conservative treatment (n = 2). It is difficult, based on the available data, to formulate guidelines for the surveillance and follow-up of arterial aneurysms in nonvascular EDS. Especially for subtypes associated with increased risk for arterial dissections and aneurysms, surveillance strategies based on noninvasive imaging, such as ultrasound, magnetic resonance angiography, and computed tomography angiography with or without venous contrast, could be recommended, as in the care guidelines for vEDS.134 Guidelines for cardiovascular care and surgery for Marfan and Loeys-Dietz syndromes, including annual cardiovascular imaging, blood pressure–lowering medication (e.g., angiotensin receptor blockers, β-blockers, or angiotensin-converting enzyme inhibitors), and surgery when approaching surgical thresholds for aortic root dimensions or acute dissection, could also be used until EDS subtype-specific recommendations are available.135, 136 There are no reports about the use of celiprolol, the β-blocker that has proven to delay vascular complications in vEDS.15, 16 One could argue, however, for its use in EDS subtypes with an increased risk of arterial rupture and/or aneurysm (e.g., cEDS, kEDS). Another drug that has anecdotally been reported to be useful in treating or preventing bleeding episodes and/or hematomas in several EDS subtypes, such as mcEDS, kEDS, and vEDS, is the procoagulant desmopressin (DDAVP).116, 137, 138
The study’s inherent limitations should be taken into account when interpreting the results. First, the systematic and predefined search strategy aimed to identify all potentially relevant studies, but the risk of selection bias is not negligible. Second, extensive natural history studies on EDS are rare: the data summarized here are derived mostly from either case reports (n = 51) or case series (n = 59), which are prone to publication and selection bias, because the cases are mainly self-selected. Third, asymptomatic features (e.g., silent aneurysms) are often not actively screened for such retrospective studies. Finally, for some subtypes, such as dEDS, kEDS-FKBP14, spEDS-B4GALT7, mcEDS-DSE, and mEDS, very few patients have been reported to date. As such, it is likely that the current review is only a mere approximation of the occurrence and spectrum of vascular complications in nonvascular EDS.
In conclusion, vascular complications are important and sometimes severe in some nonvascular EDS subtypes. They are found mainly in mcEDS, clEDS, dEDS, kEDS, and cEDS. Hematomas are most frequently reported (53%) and predominate mcEDS and clEDS. Serious, potentially life-threatening complications such as intracranial hemorrhage, arterial aneurysms, and dissections of large and medium-sized arteries, account for approximately 18, 5 and 16% of vascular complications, respectively, and are most frequently seen in dEDS (intracranial hemorrhage), cEDS (arterial dissections and aneurysms), and kEDS (arterial dissections). No complications were reported yet in spEDS-B4GALT7, BCS, and mEDS. Given the relatively high occurrence of vascular complications in EDS patients, referral for cardiovascular assessment and regular follow-up imaging may be required upon diagnosis, especially for cEDS, dEDS, and kEDS. If vascular complications are detected, the type and location of a complication will guide treatment options. However, to optimize management and care guidelines, future research should be focused on more systematic documentation of vascular and bleeding complications and their management in natural history studies and registries.
References
Beighton P, De Paepe A, Steinmann B, Tsipouras P, Wenstrup RJ . Ehlers-Danlos syndromes: revised nosology, Villefranche, 1997. Ehlers-Danlos National Foundation (USA) and Ehlers-Danlos Support Group (UK). Am J Med Genet 1998;77:31–37.
Al-Owain M, Al-Dosari MS, Sunker A, Shuaib T, Alkuraya FS . Identification of a novel ZNF469 mutation in a large family with Ehlers-Danlos phenotype. Gene 2012;511:447–450.
Micheal S, Khan MI, Islam F et al. Identification of mutations in the PRDM5 gene in brittle cornea syndrome. Cornea 2016;35:853–859.
Baumann M, Giunta C, Krabichler B et al. Mutations in FKBP14 cause a variant of Ehlers-Danlos syndrome with progressive kyphoscoliosis, myopathy, and hearing loss. Am J Hum Genet 2012;90:201–216.
Fukada T, Civic N, Furuichi T et al. The zinc transporter SLC39A13/ZIP13 is required for connective tissue development; its involvement in BMP/TGF-beta signaling pathways. PLoS One 2008;3:e3642.
Miyake N, Kosho T, Mizumoto S et al. Loss-of-function mutations of CHST14 in a new type of Ehlers-Danlos syndrome. Hum Mutat 2010;31:966–974.
Muller T, Mizumoto S, Suresh I et al. Loss of dermatan sulfate epimerase (DSE) function results in musculocontractural Ehlers-Danlos syndrome. Hum Mol Genet 2013;22:3761–3772.
Faiyaz-Ul-Haque M, Zaidi SH, Al-Ali M et al. A novel missense mutation in the galactosyltransferase-I (B4GALT7) gene in a family exhibiting facioskeletal anomalies and Ehlers-Danlos syndrome resembling the progeroid type. Am J Med Genet A 2004;128A:39–45.
Malfait F, Kariminejad A, Van Damme T et al. Defective initiation of glycosaminoglycan synthesis due to B3GALT6 mutations causes a pleiotropic Ehlers-Danlos-syndrome-like connective tissue disorder. Am J Hum Genet 2013;92:935–945.
Burch GH, Gong Y, Liu W et al. Tenascin-X deficiency is associated with Ehlers-Danlos syndrome. Nat Genet 1997;17:104–108.
Malfait F, Francomano C, Byers P et al. The 2017 international classification of the Ehlers-Danlos syndromes. Am J Med Genet C Semin Med Genet 2017;175:8–26.
Germain DP . Clinical and genetic features of vascular Ehlers-Danlos syndrome. Ann Vasc Surg 2002;16:391–397.
Pepin M, Schwarze U, Superti-Furga A, Byers PH . Clinical and genetic features of Ehlers-Danlos syndrome type IV, the vascular type. N Engl J Med 2000;342:673–680.
Beridze N, Frishman WH . Vascular Ehlers-Danlos syndrome: pathophysiology, diagnosis, and prevention and treatment of its complications. Cardiol Rev 2012;20:4–7.
Ong KT, Perdu J, De Backer J et al. Effect of celiprolol on prevention of cardiovascular events in vascular Ehlers-Danlos syndrome: a prospective randomised, open, blinded-endpoints trial. Lancet 2010;376:1476–1484.
Boutouyrie P, Bussy C, Hayoz D et al. Local pulse pressure and regression of arterial wall hypertrophy during long-term antihypertensive treatment. Circulation 2000;101:2601–2606.
Pope FM, Martin GR, McKusick VA . Inheritance of Ehlers-Danlos type IV syndrome. J Med Genet 1977;14:200–204.
Frank M, Albuisson J, Ranque B et al. The type of variants at the COL3A1 gene associates with the phenotype and severity of vascular Ehlers-Danlos syndrome. Eur J Hum Genet 2015;23:1657–1664.
Leistritz DF, Pepin MG, Schwarze U, Byers PH . COL3A1 haploinsufficiency results in a variety of Ehlers-Danlos syndrome type IV with delayed onset of complications and longer life expectancy. Genet Med 2011;13:717–722.
De Paepe A, Malfait F . Bleeding and bruising in patients with Ehlers-Danlos syndrome and other collagen vascular disorders. Br J Haematol 2004;127:491–500.
Malfait F, De Paepe A . Bleeding in the heritable connective tissue disorders: mechanisms, diagnosis and treatment. Blood Rev 2009;23:191–197.
PRISMA Group Moher D PRISMA Group Liberati A PRISMA Group Tetzlaff J PRISMA Group Altman DG PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 2009;339:b2535.
Watanabe M, Nakagawa R, Naruto T et al. A novel missense mutation of COL5A2 in a patient with Ehlers-Danlos syndrome. Hum Genome Var 2016;3:16030.
Cazzato D, Castori M, Lombardi R et al. Small fiber neuropathy is a common feature of Ehlers-Danlos syndromes. Neurology 2016;87:155–159.
Monroe GR, Harakalova M, van der Crabben SN et al. Familial Ehlers-Danlos syndrome with lethal arterial events caused by a mutation in COL5A1. Am J Med Genet A 2015;167:1196–1203.
Casey MC, Robertson I, Waters PS, Hanaghan J, Khan W, Barry K . Non-operative management of diverticular perforation in a patient with suspected Ehlers-Danlos syndrome. Int J Surg Case Rep 2014;5:135–137.
Yasuda S, Imoto K, Uchida K et al. Successful endovascular treatment of a ruptured superior mesenteric artery in a patient with EhlersDanlos syndrome. Ann Vasc Surg 2013;27:975 e971–975.
Ritelli M, Dordoni C, Venturini M et al. Clinical and molecular characterization of 40 patients with classic Ehlers-Danlos syndrome: identification of 18 COL5A1 and 2 COL5A2 novel mutations. Orphanet J Rare Dis 2013;8:58.
Morais P, Ferreira O, Magina S et al. Classic Ehlers-Danlos syndrome: case report and brief review of literature. Acta Dermatovenerol Croat 2013;21:118–122.
Mehta S, Dhar SU, Birnbaum Y . Common iliac artery aneurysm and spontaneous dissection with contralateral iatrogenic common iliac artery dissection in classic Ehlers-Danlos syndrome. Int J Angiol 2012;21:167–170.
de Leeuw K, Goorhuis JF, Tielliu IF et al. Superior mesenteric artery aneurysm in a 9-year-old boy with classical Ehlers-Danlos syndrome. Am J Med Genet A 2012;158A:626–629.
Symoens S, Malfait F, Vlummens P, Hermanns-Le T, Syx D, De Paepe A . A novel splice variant in the N-propeptide of COL5A1 causes an EDS phenotype with severe kyphoscoliosis and eye involvement. PloS one. 2011;6:e20121.
Borck G, Beighton P, Wilhelm C, Kohlhase J, Kubisch C . Arterial rupture in classic Ehlers-Danlos syndrome with COL5A1 mutation. Am J Med Genet A 2010;152A:2090–2093.
Symoens S, Malfait F, Renard M et al. COL5A1 signal peptide mutations interfere with protein secretion and cause classic Ehlers-Danlos syndrome. Hum Mutat 2009;30:E395–E403.
Malfait F, Coucke P, Symoens S, Loeys B, Nuytinck L, De Paepe A . The molecular basis of classic Ehlers-Danlos syndrome: a comprehensive study of biochemical and molecular findings in 48 unrelated patients. Hum Mutat 2005;25:28–37.
Pallotta R, Ehresmann T, Fusilli P, De Paepe A, Nuytinck L . Discordance between phenotypic appearance and genotypic findings in a familial case of classical Ehlers-Danlos syndrome. Am J Med Genet A 2004;128A:436–438.
Takahara K, Schwarze U, Imamura Y et al. Order of intron removal influences multiple splice outcomes, including a two-exon skip, in a COL5A1 acceptor-site mutation that results in abnormal pro-alpha1(V) N-propeptides and Ehlers-Danlos syndrome type I. Am J Hum Genet 2002;71:451–465.
Giunta C, Nuytinck L, Raghunath M, Hausser I, De Paepe A, Steinmann B . Homozygous Gly530Ser substitution in COL5A1 causes mild classical Ehlers-Danlos syndrome. Am J Med Genet 2002;109:284–290.
Stoler JM, Bromley B, Castro MA, Cole WG, Florer J, Wenstrup RJ . Separation of amniotic membranes after amniocentesis in an individual with the classic form of EDS and haploinsufficiency for COL5A1 expression. Am J Med Genet 2001;101:174–177.
Bouma P, Cabral WA, Cole WG, Marini JC . COL5A1 exon 14 splice acceptor mutation causes a functional null allele, haploinsufficiency of alpha 1(V) and abnormal heterotypic interstitial fibrils in Ehlers-Danlos syndrome II. J Biol Chem 2001;276:13356–13364.
Schwarze U, Atkinson M, Hoffman GG, Greenspan DS, Byers PH . Null alleles of the COL5A1 gene of type V collagen are a cause of the classical forms of Ehlers-Danlos syndrome (types I and II). Am J Hum Genet 2000;66:1757–1765.
Giunta C, Steinmann B . Compound heterozygosity for a disease-causing G1489E [corrected] and disease-modifying G530S substitution in COL5A1 of a patient with the classical type of Ehlers-Danlos syndrome: an explanation of intrafamilial variability? Am J Med Genet 2000;90:72–79.
Michalickova K, Susic M, Willing MC, Wenstrup RJ, Cole WG . Mutations of the alpha2(V) chain of type V collagen impair matrix assembly and produce Ehlers-Danlos syndrome type I. Hum Mol Genet 1998;7:249–255.
De Paepe A, Nuytinck L, Hausser I, Anton-Lamprecht I, Naeyaert JM . Mutations in the COL5A1 gene are causal in the Ehlers-Danlos syndromes I and II. Am J Hum Genet 1997;60:547–554.
Wenstrup RJ, Langland GT, Willing MC, D’Souza VN, Cole WG . A splice-junction mutation in the region of COL5A1 that codes for the carboxyl propeptide of pro alpha 1(V) chains results in the gravis form of the Ehlers-Danlos syndrome (type I). Hum Mol Genet 1996;5:1733–1736.
Nicholls AC, Oliver JE, McCarron S, Harrison JB, Greenspan DS, Pope FM . An exon skipping mutation of a type V collagen gene (COL5A1) in Ehlers-Danlos syndrome. J Med Genet 1996;33:940–946.
Toriello HV, Glover TW, Takahara K et al. A translocation interrupts the COL5A1 gene in a patient with Ehlers-Danlos syndrome and hypomelanosis of Ito. Nat Genet. 1996;13:361–365.
Colombi M, Dordoni C, Venturini M, Zanca A, Calzavara-Pinton P, Ritelli M . Delineation of Ehlers-Danlos syndrome phenotype due to the c.934C>T, p.(Arg312Cys) mutation in COL1A1: report on a three-generation family without cardiovascular events, and literature review. Am J Med Genet A 2017;173:524–530.
Gaines R, Tinkle BT, Halandras PM, Al-Nouri O, Crisostomo P, Cho JS . Spontaneous ruptured dissection of the right common iliac artery in a patient with classic Ehlers-Danlos syndrome phenotype. Ann Vasc Surg 2015;29:595 e511–594.
Malfait F, Symoens S, De Backer J et al. Three arginine to cysteine substitutions in the pro-alpha (I)-collagen chain cause Ehlers-Danlos syndrome with a propensity to arterial rupture in early adulthood. Hum Mutat. 2007;28:387–395.
Nuytinck L, Freund M, Lagae L, Pierard GE, Hermanns-Le T, De Paepe A . Classical Ehlers-Danlos syndrome caused by a mutation in type I collagen. Am J Hum Genet 2000;66:1398–1402.
Kaufman CS, Butler MG . Mutation in TNXB gene causes moderate to severe Ehlers-Danlos syndrome. World J Med Genet 2016;6:17–21.
Demirdas S, Dulfer E, Robert L et al. Recognizing the tenascin-X deficient type of Ehlers-Danlos syndrome: a cross-sectional study in 17 patients. Clin Genet 2016;91:411–425.
Chen W, Perritt AF, Morissette R et al. Ehlers-Danlos syndrome caused by biallelic TNXB variants in patients with congenital adrenal hyperplasia. Human Mutat 2016;37:893–897.
Sakiyama T, Kubo A, Sasaki T et al. Recurrent gastrointestinal perforation in a patient with Ehlers-Danlos syndrome due to tenascin-X deficiency. J Dermatol 2015;42:511–514.
Penisson-Besnier I, Allamand V, Beurrier P et al. Compound heterozygous mutations of the TNXB gene cause primary myopathy. Neuromuscul Disord 2013;23:664–669.
Schalkwijk J, Zweers MC, Steijlen PM et al. A recessive form of the Ehlers-Danlos syndrome caused by tenascin-X deficiency. N Engl J Med 2001;345:1167–1175.
Melis D, Cappuccio G, Ginocchio VM et al. Cardiac valve disease: an unreported feature in Ehlers Danlos syndrome arthrocalasia type? Ital J Pediatr 2012;38:65.
Malfait F, Symoens S, Coucke P, Nunes L, De Almeida S, De Paepe A . Total absence of the alpha2(I) chain of collagen type I causes a rare form of Ehlers-Danlos syndrome with hypermobility and propensity to cardiac valvular problems. J Med Genet 2006;43:e36.
Schwarze U, Hata R, McKusick VA et al. Rare autosomal recessive cardiac valvular form of Ehlers-Danlos syndrome results from mutations in the COL1A2 gene that activate the nonsense-mediated RNA decay pathway. Am J Hum Genet 2004;74:917–930.
Hatamochi A, Hamada T, Yoshino M, Hashimoto T . The first Japanese case of the arthrochalasia type of Ehlers-Danlos syndrome with COL1A2 gene mutation. Gene 2014;538:199–203.
Giunta C, Superti-Furga A, Spranger S, Cole WG, Steinmann B . Ehlers-Danlos syndrome type VII: clinical features and molecular defects. J Bone Joint Surg Am 1999;81:225–238.
Byers PH, Duvic M, Atkinson M et al. Ehlers-Danlos syndrome type VIIA and VIIB result from splice-junction mutations or genomic deletions that involve exon 6 in the COL1A1 and COL1A2 genes of type I collagen. Am J Med Genet 1997;72:94–105.
Ho KK, Kong RY, Kuffner T, Hsu LH, Ma L, Cheah KS . Further evidence that the failure to cleave the aminopropeptide of type I procollagen is the cause of Ehlers-Danlos syndrome type VII. Hum Mut 1994;3:358–364.
Carr AJ, Chiodo AA, Hilton JM, Chow CW, Hockey A, Cole WG . The clinical features of Ehlers-Danlos syndrome type VIIB resulting from a base substitution at the splice acceptor site of intron 5 of the COL1A2 gene. J Med Genet 1994;31:306–311.
Chiodo AA, Hockey A, Cole WG . A base substitution at the splice acceptor site of intron 5 of the COL1A2 gene activates a cryptic splice site within exon 6 and generates abnormal type I procollagen in a patient with Ehlers-Danlos syndrome type VII. J Biol Chem 1992;267:6361–6369.
Watson RB, Wallis GA, Holmes DF, Viljoen D, Byers PH, Kadler KE . Ehlers Danlos syndrome type VIIB. Incomplete cleavage of abnormal type I procollagen by N-proteinase in vitro results in the formation of copolymers of collagen and partially cleaved pNcollagen that are near circular in cross-section. J Biol Chem 1992;267:9093–9100.
Vasan NS, Kuivaniemi H, Vogel BE et al. A mutation in the pro alpha 2(I) gene (COL1A2) for type I procollagen in Ehlers-Danlos syndrome type VII: evidence suggesting that skipping of exon 6 in RNA splicing may be a common cause of the phenotype. Am J Hum Genet 1991;48:305–317.
D’Alessio M, Ramirez F, Blumberg BD et al. Characterization of a COL1A1 splicing defect in a case of Ehlers-Danlos syndrome type VII: further evidence of molecular homogeneity. Am J Hum Genet 1991;49:400–406.
Wirtz MK, Glanville RW, Steinmann B, Rao VH, Hollister DW . Ehlers-Danlos syndrome type VIIB. Deletion of 18 amino acids comprising the N-telopeptide region of a pro-alpha 2(I) chain. J Biol Chem 1987;262:16376–16385.
Van Damme T, Colige A, Syx D et al. Expanding the clinical and mutational spectrum of the Ehlers-Danlos syndrome, dermatosparaxis type. Genet Med 2016;18:882–891.
Solomons J, Coucke P, Symoens S et al. Dermatosparaxis (Ehlers-Danlos type VIIC): prenatal diagnosis following a previous pregnancy with unexpected skull fractures at delivery. Am J Med Genet A 2013;161A:1122–1125.
Bar-Yosef O, Polak-Charcon S, Hoffman C, Feldman ZP, Frydman M, Kuint J . Multiple congenital skull fractures as a presentation of Ehlers-Danlos syndrome type VIIC. Am J Med Genet A 2008;146A:3054–3057.
Malfait F, De Coster P, Hausser I et al. The natural history, including orofacial features of three patients with Ehlers-Danlos syndrome, dermatosparaxis type (EDS type VIIC). Am J Med Genet A 2004;131:18–28.
Colige A, Nuytinck L, Hausser I et al. Novel types of mutation responsible for the dermatosparactic type of Ehlers-Danlos syndrome (Type VIIC) and common polymorphisms in the ADAMTS2 gene. J Invest Dermatol 2004;123:656–663.
Colige A, Sieron AL, Li SW et al. Human Ehlers-Danlos syndrome type VII C and bovine dermatosparaxis are caused by mutations in the procollagen I N-proteinase gene. Am J Hum Genet 1999;65:308–317.
Abdalla EM, Rohrbach M, Burer C et al. Kyphoscoliotic type of Ehlers-Danlos Syndrome (EDS VIA) in six Egyptian patients presenting with a homogeneous clinical phenotype. Eur J Pediatr 2015;174:105–112.
Tosun A, Kurtgoz S, Dursun S, Bozkurt G . A case of Ehlers-Danlos syndrome type VIA with a novel PLOD1 gene mutation. Pediatr Neurol 2014;51:566–569.
Gok E, Goksel OS, Alpagut U, Dayioglu E . Spontaneous brachial pseudo-aneurysm in a 12-year-old with kyphoscoliosis-type Ehlers-Danlos syndrome. Eur J Vasc Endovasc Surg 2012;44:482–484.
Rohrbach M, Vandersteen A, Yis U et al. Phenotypic variability of the kyphoscoliotic type of Ehlers-Danlos syndrome (EDS VIA): clinical, molecular and biochemical delineation. Orphanet J Rare Dis 2011;6:46.
Kariminejad A, Bozorgmehr B, Khatami A, Kariminejad MH, Giunta C, Steinmann B . Ehlers-Danlos syndrome type VI in a 17-year-old Iranian boy with severe muscular weakness—a diagnostic challenge? Iran J Pediatr 2010;20:358–362.
Voermans NC, Bonnemann CG, Lammens M, van Engelen BG, Hamel BC . Myopathy and polyneuropathy in an adolescent with the kyphoscoliotic type of Ehlers-Danlos syndrome. Am J Med Genet A 2009;149A:2311–2316.
Yis U, Dirik E, Chambaz C, Steinmann B, Giunta C . Differential diagnosis of muscular hypotonia in infants: the kyphoscoliotic type of Ehlers-Danlos syndrome (EDS VI). Neuromuscul Disord 2008;18:210–214.
Salavoura K, Valari M, Kolialexi A, Mavrou A, Kitsiou S . A case of Ehlers Danlos syndrome type VI. Genet Couns 2006;17:291–294.
Giunta C, Randolph A, Al-Gazali LI, Brunner HG, Kraenzlin ME, Steinmann B . Nevo syndrome is allelic to the kyphoscoliotic type of the Ehlers-Danlos syndrome (EDS VIA). Am J Med Genet A 2005;133A:158–164.
Giunta C, Randolph A, Steinmann B . Mutation analysis of the PLOD1 gene: an efficient multistep approach to the molecular diagnosis of the kyphoscoliotic type of Ehlers-Danlos syndrome (EDS VIA). Mol Genet Metab 2005;86:269–276.
Brunk I, Stover B, Ikonomidou C, Brinckmann J, Neumann LM . Ehlers-Danlos syndrome type VI with cystic malformations of the meninges in a 7-year-old girl. Eur J Pediatr 2004;163:214–217.
Eyre D, Shao P, Weis MA, Steinmann B . The kyphoscoliotic type of Ehlers-Danlos syndrome (type VI): differential effects on the hydroxylation of lysine in collagens I and II revealed by analysis of cross-linked telopeptides from urine. Mol Genet Metab 2002;76:211–216.
Yeowell HN, Allen JD, Walker LC, Overstreet MA, Murad S, Thai SF . Deletion of cysteine 369 in lysyl hydroxylase 1 eliminates enzyme activity and causes Ehlers-Danlos syndrome type VI. Matrix Biol 2000;19:37–46.
Yeowell HN, Walker LC, Farmer B, Heikkinen J, Myllyla R . Mutational analysis of the lysyl hydroxylase 1 gene (PLOD) in six unrelated patients with Ehlers-Danlos syndrome type VI: prenatal exclusion of this disorder in one family. Hum Mutat. 2000;16:90.
Walker LC, Marini JC, Grange DK, Filie J, Yeowell HN . A patient with Ehlers-Danlos syndrome type VI is homozygous for a premature termination codon in exon 14 of the lysyl hydroxylase 1 gene. Mol Genet Metab 1999;67:74–82.
Pousi B, Hautala T, Heikkinen J, Pajunen L, Kivirikko KI, Myllyla R . Alu-Alu recombination results in a duplication of seven exons in the lysyl hydroxylase gene in a patient with the type VI variant of Ehlers-Danlos syndrome. Am J Hum Genet 1994;55:899–906.
Dembure PP, Janko AR, Priest JH, Elsas LJ . Ascorbate regulation of collagen biosynthesis in Ehlers-Danlos syndrome, type VI. Metabolism 1987;36:687–691.
Bursztejn AC, Baumann M, Lipsker D . Ehlers-Danlos syndrome related to FKBP14 mutations: detailed cutaneous phenotype. Clin Exp Dermatol 2017;42:64–67.
Dordoni C, Ciaccio C, Venturini M, Calzavara-Pinton P, Ritelli M, Colombi M . Further delineation of FKBP14-related Ehlers-Danlos syndrome: A patient with early vascular complications and non-progressive kyphoscoliosis, and literature review. Am J Med Genet A 2016;170:2031–2038.
Aldeeri AA, Alazami AM, Hijazi H, Alzahrani F, Alkuraya FS . Excessively redundant umbilical skin as a potential early clinical feature of Morquio syndrome and FKBP14-related Ehlers-Danlos syndrome. Clin Genet 2014;86:469–472.
Murray ML, Yang M, Fauth C, Byers PH . FKBP14-related Ehlers-Danlos syndrome: expansion of the phenotype to include vascular complications. Am J Med Genet A 2014;164A:1750–1755.
Rohrbach M, Spencer HL, Porter LF et al. ZNF469 frequently mutated in the brittle cornea syndrome (BCS) is a single exon gene possibly regulating the expression of several extracellular matrix components. Mol Genet Metab 2013;109:289–295.
Khan AO, Aldahmesh MA, Mohamed JN, Alkuraya FS . Blue sclera with and without corneal fragility (brittle cornea syndrome) in a consanguineous family harboring ZNF469 mutation (p.E1392X). Arch Ophthalmol. 2010;128:1376–1379.
Christensen AE, Knappskog PM, Midtbo M et al. Brittle cornea syndrome associated with a missense mutation in the zinc-finger 469 gene. Invest Ophthalmol Vis Sci 2010;51:47–52.
Abu A, Frydman M, Marek D et al. Deleterious mutations in the Zinc-Finger 469 gene cause brittle cornea syndrome. Am J Hum Genet 2008;82:1217–1222.
Porter LF, Galli GG, Williamson S et al. A role for repressive complexes and H3K9 di-methylation in PRDM5-associated brittle cornea syndrome. Hum Mol Genet 2015;24:6565–6579.
Porter LF, Gallego-Pinazo R, Keeling CL et al. Bruch’s membrane abnormalities in PRDM5-related brittle cornea syndrome. Orphanet J Rare Dis 2015;10:145.
Avgitidou G, Siebelmann S, Bachmann B, Kohlhase J, Heindl LM, Cursiefen C . Brittle cornea syndrome: case report with novel mutation in the PRDM5 gene and review of the literature. Case Rep Ophthalmol Med 2015;2015:637084.
Aldahmesh MA, Mohamed JY, Alkuraya FS . A novel mutation in PRDM5 in brittle cornea syndrome. Clin Genet 2012;81:198–199.
Burkitt Wright EM, Spencer HL, Daly SB et al. Mutations in PRDM5 in brittle cornea syndrome identify a pathway regulating extracellular matrix development and maintenance. Am J Hum Genet 2011;88:767–777.
Salter CG, Davies JH, Moon RJ et al. Further defining the phenotypic spectrum of B4GALT7 mutations. Am J Med Genet A 2016;170:1556–1563.
Cartault F, Munier P, Jacquemont ML et al. Expanding the clinical spectrum of B4GALT7 deficiency: homozygous p.R270C mutation with founder effect causes Larsen of Reunion Island syndrome. Eur J Hum Genet 2015;23:49–53.
Guo MH, Stoler J, Lui J et al. Redefining the progeroid form of Ehlers-Danlos syndrome: report of the fourth patient with B4GALT7 deficiency and review of the literature. Am J Med Genet A 2013;161A:2519–2527.
Okajima T, Fukumoto S, Furukawa K, Urano T . Molecular basis for the progeroid variant of Ehlers-Danlos syndrome. Identification and characterization of two mutations in galactosyltransferase I gene. J Biol Chem 1999;274:28841–28844.
Vorster AA, Beighton P, Ramesar RS . Spondyloepimetaphyseal dysplasia with joint laxity (Beighton type); mutation analysis in eight affected South African families. Clin Genet 2015;87:492–495.
Sellars EA, Bosanko KA, Lepard T, Garnica A, Schaefer GB . A newborn with complex skeletal abnormalities, joint contractures, and bilateral corneal clouding with sclerocornea. Semin Pediatr Neurol 2014;21:84–87.
Nakajima M, Mizumoto S, Miyake N et al. Mutations in B3GALT6, which encodes a glycosaminoglycan linker region enzyme, cause a spectrum of skeletal and connective tissue disorders. Am J Hum Genet 2013;92:927–934.
Giunta C, Elcioglu NH, Albrecht B et al. Spondylocheiro dysplastic form of the Ehlers-Danlos syndrome—an autosomal-recessive entity caused by mutations in the zinc transporter gene SLC39A13. Am J Hum Genet 2008;82:1290–1305.
Mizumoto S, Kosho T, Hatamochi A et al. Defect in dermatan sulfate in urine of patients with Ehlers-Danlos syndrome caused by a CHST14/D4ST1 deficiency. Clin Biochem 2017;50:670–677.
Janecke AR, Li B, Boehm M et al. The phenotype of the musculocontractural type of Ehlers-Danlos syndrome due to CHST14 mutations. Am J Med Genet A 2016;170A:103–115.
Mochida K, Amano M, Miyake N, Matsumoto N, Hatamochi A, Kosho T . Dermatan 4-O-sulfotransferase 1-deficient Ehlers-Danlos syndrome complicated by a large subcutaneous hematoma on the back. J Dermatol 2016;43:832–833.
Syx D, Van Damme T, Symoens S et al. Genetic heterogeneity and clinical variability in musculocontractural Ehlers-Danlos syndrome caused by impaired dermatan sulfate biosynthesis. Hum Mutat 2015;36:535–547.
Winters KA, Jiang Z, Xu W et al. Re-assigned diagnosis of D4ST1-deficient Ehlers-Danlos syndrome (adducted thumb-clubfoot syndrome) after initial diagnosis of Marden-Walker syndrome. Am J Med Genet A 2012;158A:2935–2940.
Mendoza-Londono R, Chitayat D, Kahr WH et al. Extracellular matrix and platelet function in patients with musculocontractural Ehlers-Danlos syndrome caused by mutations in the CHST14 gene. Am J Med Genet A 2012;158A:1344–1354.
Voermans NC, Kempers M, Lammens M et al. Myopathy in a 20-year-old female patient with D4ST-1 deficient Ehlers-Danlos syndrome due to a homozygous CHST14 mutation. Am J Med Genet A 2012;158A:850–855.
Shimizu K, Okamoto N, Miyake N et al. Delineation of dermatan 4-O-sulfotransferase 1 deficient Ehlers-Danlos syndrome: observation of two additional patients and comprehensive review of 20 reported patients. Am J Med Genet A 2011;155A:1949–1958.
Malfait F, Syx D, Vlummens P et al. Musculocontractural Ehlers-Danlos Syndrome (former EDS type VIB) and adducted thumb clubfoot syndrome (ATCS) represent a single clinical entity caused by mutations in the dermatan-4-sulfotransferase 1 encoding CHST14 gene. Hum Mutat 2010;31:1233–1239.
Dundar M, Muller T, Zhang Q et al. Loss of dermatan-4-sulfotransferase 1 function results in adducted thumb-clubfoot syndrome. Am J Hum Genet 2009;85:873–882.
Zou Y, Zwolanek D, Izu Y et al. Recessive and dominant mutations in COL12A1 cause a novel EDS/myopathy overlap syndrome in humans and mice. Hum Mol Genet 2014;23:2339–2352.
Hicks D, Farsani GT, Laval S et al. Mutations in the collagen XII gene define a new form of extracellular matrix-related myopathy. Hum Mol Genet 2014;23:2353–2363.
Kapferer-Seebacher I, Pepin M, Werner R et al. Periodontal Ehlers-Danlos syndrome is caused by mutations in C1R and C1S, which encode subcomponents C1r and C1s of complement. Am J Hum Genet 2016;99:1005–1014.
Gotte M, Kresse H . Defective glycosaminoglycan substitution of decorin in a patient with progeroid syndrome is a direct consequence of two point mutations in the galactosyltransferase I (beta4GalT-7) gene. Biochem Genet 2005;43:65–77.
Fergelot P, Coupry I, Rooryck C et al. Atypical male and female presentations of FLNA-related periventricular nodular heterotopia. Eur J Med Genet 2012;55:313–318.
Pepin MG, Schwarze U, Rice KM, Liu M, Leistritz D, Byers PH . Survival is affected by mutation type and molecular mechanism in vascular Ehlers-Danlos syndrome (EDS type IV). Genet Med 2014;16:881–888.
El-Hemaidi I, Gharaibeh A, Shehata H . Menorrhagia and bleeding disorders. Curr Opin Obstet Gynecol 2007;19:513–520.
Knight M, Callaghan WM, Berg C et al. Trends in postpartum hemorrhage in high resource countries: a review and recommendations from the International Postpartum Hemorrhage Collaborative Group. BMC Pregnancy Childbirth 2009;9:55.
Callam MJ . Epidemiology of varicose veins. Br J Surg 1994;81:167–173.
Pepin MG, Murray ML, Byers PH. Vascular Ehlers-Danlos syndrome. In: Pagon RA, Adam MP, Ardinger HH, et al. (eds.). GeneReviews. University of Washington: Seattle, WA, 1993.
Loeys BL, Dietz HC, Braverman AC et al. The revised Ghent nosology for the Marfan syndrome. J Med Genet 2010;47:476–485.
MacCarrick G, Black JH 3rd, Bowdin S et al. Loeys-Dietz syndrome: a primer for diagnosis and management. Genet Med 2014;16:576–587.
Mast KJ, Nunes ME, Ruymann FB, Kerlin BA . Desmopressin responsiveness in children with Ehlers-Danlos syndrome associated bleeding symptoms. Br J Haematol 2009;144:230–233.
Stine KC, Becton DL . DDAVP therapy controls bleeding in Ehlers-Danlos syndrome. J Pediatr Hematol Oncol 1997;19:156–158.
Acknowledgements
This work was supported by Methusalem grant 08/01M01108 from Ghent University. F.M. is a fellow of the Fund for Scientific Research (FWO), Belgium.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary material is linked to the online version of the paper at
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
D'hondt, S., Van Damme, T. & Malfait, F. Vascular phenotypes in nonvascular subtypes of the Ehlers-Danlos syndrome: a systematic review. Genet Med 20, 562–573 (2018). https://doi.org/10.1038/gim.2017.138
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gim.2017.138
Keywords
This article is cited by
-
People with a connective tissue disorder may be especially vulnerable to the endothelial damage that characterizes long COVID due to the fragility of their vasculature and slow wound healing
Angiogenesis (2024)
-
Unveiling cellular and molecular aspects of ascending thoracic aortic aneurysms and dissections
Basic Research in Cardiology (2024)
-
Retinal arterial tortuosity in Ehlers–Danlos syndromes
Eye (2023)
-
Abusive head trauma: neuroimaging mimics and diagnostic complexities
Pediatric Radiology (2021)
-
Extracellular matrix, regional heterogeneity of the aorta, and aortic aneurysm
Experimental & Molecular Medicine (2019)