Abstract
Purpose:
This review assessed the psychological impact that acquiring personal and familial genetic information has on children. We also examined the concordance between the available empirical data and clinical guidance/perspectives articles.
Methods:
We screened 591 abstracts and identified 13 studies, representing 966 children. Ten studies assessed 386 children tested for familial adenomatous polyposis (n = 171), hereditary cardiac disease (n = 134), and other conditions (n = 81). Three studies addressed the impact of BRCA1/2 testing of a family member on 580 children.
Results:
Serious adverse psychological outcomes were uncommon. Most studies reported no significant increase in mean anxiety, depression, and distress scores (n = 8, 61.5%); however, some children experienced intrafamilial distress, discrimination, and guilt/regret. Some children were more concerned about their own health or their family members’ health. There was limited consistency between anticipated adverse impact and empirical data.
Conclusions:
The review identified little conclusive evidence of deleterious psychological consequences for children acquiring genetic information. However, there is a lack of data regarding genetic testing for conditions that may not be treatable/modifiable, as well as a dearth of longitudinal studies. Therefore, clinical caution remains essential for the ethical integration of genetic testing into pediatrics. Further research assessing the potential positive and negative effects of genetic testing in childhood is warranted.
Genet Med 18 8, 755–762.
Similar content being viewed by others
Main
Genetic testing for disease predisposition has enhanced diagnostic, therapeutic, and risk-reducing protocols for hereditary illness.1,2 With the growth in genetics-related technologies following the success of the Human Genome Project,3 interest in the psychological implications of these technologies has increased.4,5,6 Research has highlighted both positive and negative implications for those who undergo genetic testing.7
A key medical benefit of testing for a potentially harmful genetic alteration is that it may identify or eliminate the need for targeted screening.3,8 Other potential benefits include the opportunity for targeted therapy or risk-reducing interventions, which may improve survival, and provide a sense of empowerment for patients and their families.9 For example, individuals with a known hereditary cancer syndrome, such as female BRCA1/2 carriers, are encouraged to undergo breast cancer screening at a younger age and at a greater frequency than women in the general population.8 Identified noncarriers may feel less anxious and experience relief from uncertainty.10 Screening resources can be preserved and focused on those at highest risk for the disease, eliminating unnecessary surveillance in those who are mutation-negative.11
Noted disadvantages of genetic testing include possible anxiety, worry, guilt, and depression (at least in the short term) following disclosure of a positive test result.12,13 Patients may also receive an uncertain result, whereby the significance of the genetic alteration is unclear, which can adversely impact psychological functioning.14 Concerns about access to and affordability of life insurance may also arise, especially in those with no current disease, as well as worries about possible employment discrimination when a consumer’s privacy regarding genetic information is not guaranteed.13,15
Although numerous studies have investigated the psychological impact of genetic testing among adults, far less work has explored the potential positive and negative implications for children. There are clear ethical concerns regarding children’s potential loss of autonomy if their parents consent to genetic testing on their behalf.16,17 This is on the basis that children tested at their parents’ request lose their right to make the decision for themselves upon reaching maturity. Yet, in some cases, genetic testing in childhood is important. In familial adenomatous polyposis (FAP), for example, genetic testing enables clinicians to offer treatment and potentially life-saving interventions for children who test positive while sparing those identified as mutation-negative from unnecessary surveillance and anxiety.11
The possibility of a medical benefit of genetic testing is one of the most important justifications for performing genetic testing in a minor.18 Genetic testing in childhood may also allow the opportunity for early psychological adjustment and the ability to alert other family members (i.e., siblings, parents, cousins) to hereditary risk.10,19 Conversely, an important justification for postponing testing until the minor is of an age more appropriate for making a fully informed personal decision is the absence of any potential medical benefit.18
The implications of pediatric genetic testing differ by condition. Some conditions can affect children during early childhood (necessitating early testing), whereas others affect older children and young adults (allowing testing to be postponed until an older age).20,21 Different conditions also have different penetrance rates, ranging from complete penetrance, whereby all or a high percentage of carrier children will develop the disease, to incomplete penetrance, whereby carrier children’s risks differ by condition.20
Further complicating pediatric testing is the mode of inheritance of different conditions.21,22 Children with an autosomal dominant condition have a 50% chance of inheriting a parent’s deleterious mutation. These conditions include those such as FAP, hypertrophic cardiomyopathy, and acute intermittent porphyria. Children from families with a condition with autosomal recessive inheritance (e.g., aspartylglucosaminuria) are at risk for developing the condition themselves (25%) or for being a carrier for the condition, whereas children tested for X-linked conditions have differing risks depending on their gender.21,22
There are two types of genetic information that a child may acquire. The first type relates to when genetic testing is conducted directly on the child, possibly alerting the child to his or her own personal, potentially harmful, genetic alteration (referred to hereafter as “personal genetic testing”). The second relates to the testing of a child’s family member, when the child becomes aware of his or her own potentially increased hereditary risk by learning another family member’s results (referred to hereafter as “genetic testing of family members”).
Children may be affected by either type of genetic information in different or more complex ways when compared with adults. Children’s developmental level or cognitive ability may impact how a child understands information about the potential implications of genetic testing. This understanding is essential to informed assent/consent for genetic testing.23,24 More mature children, for example, are likely to better comprehend the implications of predictive genetic testing than are younger children.17
Acquiring genetic information through genetic testing of family members raises additional unique considerations in children. Knowing, for example, that a mother has increased hereditary risk for cancer may increase the child’s fears about the mother’s future health and may increase the child’s concerns about whether the parent may die at an early age.25,26 Children’s responses may also be affected by their observations of the reactions their family members have to receiving their results,27 They may also struggle to understand what risks they personally face based on their family member’s results, potentially increasing distress.17 However, despite these concerns being highlighted in several works,28,29 the literature regarding the psychological impact of genetic information on children is diverse and has yet to be rigorously reviewed.
Objectives
This review therefore aimed to compile the available evidence documenting the psychological impact of genetic information on children and to assess the degree to which this evidence is supported in clinical guidance/perspectives articles published by lead authors in the field. Specifically, the review aimed to answer these questions:
-
1. What is the psychological impact of personal genetic testing on children?
-
2. What is the psychological impact on children who acquire genetic information from genetic testing conducted for their family members?
-
3. What is the concordance between the existing evidence on the impact of genetic information on children and guidance and perspectives published by the opinion leaders in the field?
Materials and Methods
We used the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement to guide the review process.30
Database search procedure
We searched Medline, EMBASE, PsychInfo, and Science Direct using search terms outlining the relevant topic (e.g., “genetic testing,” “hereditary disease”), pertinent outcomes (e.g., “adverse effects,” “psychosocial impact”), and the target age group (e.g., “child,” “adolescent”). Searches were run and combined using EndNote X7 (Thomson Reuters, New York City, NY). Supplementary Table S1 online summarizes the search terms used.
Article selection: inclusion criteria
Articles were eligible if they:
-
1. Addressed the psychological well-being of children exposed to personal genetic testing or to genetic testing of a family member to clarify their risk of any medical condition;
-
2. Included a pediatric sample (i.e., <21 years of age at the time of receiving genetic information, per the American Academy of Pediatrics definition of pediatric age limits)31 (both parent report and child report of impact on child were eligible);
-
3. Were published in a peer-reviewed journal in English between 1994 and 2015;
-
4. Were original publications in English (systematic reviews, case studies, and comparative studies were excluded).
Data extraction
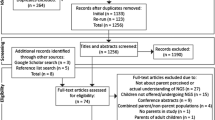
We identified 951 abstracts and exported them to EndNote X7 (Thomson Reuters). After de-duplicating, we screened the remaining 694 articles for publication type (original study) before two reviewers (L.H. and B.C.) screened the remaining 591 abstracts. If an abstract fulfilled all inclusion criteria, then we extracted the full-text article. Twenty (2.9% of 694 eligible abstracts) studies met all the inclusion criteria. Figure 1 summarizes the data extraction procedure. When there was disagreement, the two reviewers consulted a third reviewer (C.W.) to determine article suitability. To supplement the database search we also searched the reference lists of eligible articles, yielding three additional eligible articles.
The PRISMA 30 flow diagram displaying articles included and excluded in this review.
Critical appraisal
We critically appraised each study using the criteria described previously.32 Each study received a categorical assessment, noting whether each study met or failed to meet 14 equally weighted criteria ( Table 1 ).
Data collection procedure
We used three standardized tables to extract information from the captured articles. In summarizing empirical study data, we recorded the study outcomes, study design, and the main findings. We categorized the data according to whether the study pertained to personal genetic information (Supplementary Table S2 online) or familial genetic information (Supplementary Table S3 online). In summarizing guidance/perspectives articles, we considered the target population and the main argued themes (Supplementary Table S4 online). We then compiled an additional table to document the proposed psychological benefits and burdens of pediatric genetic testing in guidance/perspectives articles compared with the results of empirical studies ( Table 2 ). Authorship, journal, and institutional blinding were not used. We summarized the results according to the research question they addressed rather than their methodological approach.
Results
The review captured 10 studies addressing the psychological impact of personal genetic testing on 386 children (171 tested for FAP, 134 children tested for hereditary cardiac disease risk, 81 children tested for other conditions). Three studies addressed the psychological impact of BRCA1/2 testing of family members, representing the perspectives of 580 children. Empirical studies were conducted in the United States (n = 4, 30.7%), the Netherlands (n = 2, 15.4%), Norway (n = 2, 15.4%), Finland (n = 2, 15.4%), Denmark (n = 1, 7.7%), Canada (n = 1, 7.7%), and the United Kingdom (n = 1, 7.7%). Sample sizes ranged from 1033 to 50534 children. Child participant ages ranged from 5 to <25 years33,34 at the time of testing. The review captured 10 clinical guidance/perspectives articles discussing the psychological impact of genetic testing in children, six (60%) of which were published in the United States.
Critical appraisal
All studies included a description of their aims, setting, sampling procedures, study objectives, data collection, and data analysis. In total, 92.3% of studies included a critical reflection of their results, whereas 61.5% of articles reported on result reliability and/or validity, and 84.6% described study limitations or strengths. No study reported the involvement of a consumer representative in the development of the project.
What is the Psychological Impact of Personal Genetic Testing on Children?
Psychological distress
The majority of studies (n = 9, 90%) concluded that adverse psychological implications of personal genetic testing on children were infrequent in the short term. Five studies reported no change in children’s mean anxiety scores after testing, between children testing positive or negative for a genetic mutation, or between children who were found to be mutation carriers or to have an unknown mutation status compared with a control (noncarrier children).28,34,35,36,37 Two studies assessing psychological distress (anxiety and depression combined) reported scores that matched the child’s baseline scores and remained in the normal range after testing for FAP28 and hypertrophic cardiomyopathy risk.36
Five studies, however, found nonsignificant increases in children’s depressive symptoms after genetic testing for FAP28,34,35 or hereditary cardiac disease risk.36,38 When exploring outcomes for particular subgroups, there was some evidence to suggest that children with affected parents and those with positive results may be more at risk. One study reported that children with a mother with breast cancer were at increased risk for anxiety and depression.35 Regarding FAP, both Codori and colleagues35 and Gjone and colleagues39 reported that children had less favorable outcomes if a parent was affected by FAP, whereas Michie and colleagues reported that 10% of children testing positive reported regretting being tested.28 Gjone and colleagues also reported that 36% of children (8/22 children) tested for FAP fulfilled the criteria for a psychiatric diagnosis following testing.39
Quality of life and behavior
Two studies reported that 78% of children tested for two X-linked diseases40 and 84% of children tested for Aspartylglucosaminuria risk37 perceived that genetic testing had no impact on their quality of life. These studies reported that a further 13%40 and 4%37 of children described positive influences on their lives. Meulenkamp et al.41 reported that 15% of children in their study reported worrying about their hereditary predisposition to cardiovascular disease, whereas 90% of children at risk for hereditary risk of acute intermittent porphyria reported that knowledge of their personal risk status had no effect on their educational, occupational, or social lives.42 An FAP study found that some children worried less and showed a decrease in situational distress after testing negative due to lower perceived risk of getting polyposis and increased confidence in their risk estimate.28 Three studies assessing behavioral problems in children tested for FAP risk also reported scores matching that of baseline data or controls.28,34,35
Familial interactions
Two studies described the impact of the result of a child’s genetic status on relationships between the child and other family members. In one, 56.3% of child carriers of the autosomal recessive condition Aspartylglucosaminuria did not report being treated differently by their parents, and 18.75% could not tell if there was a difference in the way they were treated.37 In another study of X-linked conditions, all children reported not being treated differently by their siblings because of their Duchenne muscular dystrophy or Hemophilia A carrier status.40
What is the Psychological Impact on Children Acquiring Genetic Information from Genetic Testing of their Family Members?
Each of the three captured studies assessing the impact of genetic information acquired by children when a family member was tested for a BRCA1/2 mutation concluded that serious adverse psychological implications were uncommon. One study reported that 11% of parents perceived that their children had experienced distress in response to learning about the genetic status of their parents.33 Another study reported that children’s self-report scores for anxiety, depression, and behavioral problems were within the normal range and that most children worried about becoming sick a ‘little bit of the time or less’ (75%).25 However, 55% acknowledged worrying ‘a fair amount or more’ about their risk of developing cancer and 50% reported worrying about a family member’s risk.25 Adolescent daughters with mothers at risk for carrying a BRCA1/2 mutation also reported concerns about developing breast cancer and about their own susceptibility to genetic mutations when compared with the general population, with 85% of daughters believing that they were susceptible to developing breast cancer (compared with 10% of controls).26
What is the Concordance Between Empirical Data on the Impact of Genetic Information on Children and Available Clinical Guidance/Perspectives?
The reported beneficial and negative psychological implications of genetic information in children highlighted in clinical guidance and perspective articles were relatively consistent across articles, as summarized in Supplementary Table S3 online. However, there was little consistency between the proposed adverse psychological implications of genetic information and the available empirical data. Table 2 compares and contrasts each proposed benefit and disadvantage of genetic testing with the available empirical data.
The primary benefit proposed in seven (70%) articles was that genetic testing may lead to early risk-reducing screening or treatment, which might positively impact the child’s psychological well-being (for example, by reducing anxiety or disease-related worry).3,16,29,43,44,45,46 Themes of relief from uncertainty after acquiring genetic information were noted in three articles.3,16,44 Eight (80%) articles presented the view that a reduction in concern about the future may result from children acquiring genetic information. In particular, authors highlighted the benefits of being able to plan employment, social, educational, and economic endeavors according to the genetic status of the patients.3,16,17,29,43,44,46,47
The primary proposed negative outcome of genetic testing was increased anxiety about the child’s future health, noted as a possible adverse implication in six (60%) articles.3,16,27,43,45,47 Potential feelings of guilt and self-blame as a result of genetic testing were noted in five (50%) articles.16,17,29,43,47 Additionally, the potential for survivor guilt in children receiving a negative genetic result was discussed in five (50%) articles.3,16,29,44,47 The potential loss of autonomy in the event that a child’s parent requested the genetic testing was discussed in two (20%) articles.16,29 Eight (80%) articles3,16,17,29,43,44,46,47 highlighted the possibility of harm to the child’s identity as a result of genetic testing, particularly the harm to self-concept and self-image. Four articles highlighted the potential harm to the child’s self-worth due to perceived failure to meet parental expectations.16,17,46,47
Discussion
This review of the psychological impact that acquiring genetic information has on children identified 13 articles from 9 countries addressing only a few genetic conditions, indicating that this area of research is in its infancy. Ten reviewed studies using differing methodologies reported no significant effects on children’s mean anxiety, depression, and quality of life scores when 386 children underwent genetic testing for different conditions. Three studies of the impact on children when their family members were tested also reported few significant effects, although they highlighted some unique concerns of children from families affected by genetic conditions. There was little consistency between the concerns raised in the 10 reviewed clinical guidance/perspectives articles and the available empirical data, highlighting the urgent need for further research to clarify the impact of acquiring genetic information in childhood.
With regard to acquiring personal genetic information in children, some parallels may be drawn between current concerns about testing children and early concerns about testing adults (e.g., for BRCA1/2 mutations).48 With time, many concerns about BRCA1/2 testing have eased, with data suggesting that the risk of adverse psychological outcomes after testing is outweighed by its potential benefits.48 The majority of patients identified as BRCA1/2 carriers do not experience sustained increases in anxiety or depression.48 Noncarriers typically experience relief.48 There is, however, an important subset of adults who experience significant distress after genetic testing.7 There may be a similar pattern in children, such that subgroups of children may experience significant distress. This pattern may have been masked by focusing on mean distress scores in the reviewed studies. Future studies focusing on higher-risk groups (such as children with affected parents) and elucidating the impact of receiving different genetic test results (i.e., positive, negative, and inconclusive results) are important. Also, little is known about the more nuanced aspects of children of differing developmental stages learning about their genetic status. It may be that short-term assessments of childhood anxiety and depression, for example, do not adequately capture the true effect of acquiring genetic information in childhood.
Although early data suggest that a serious impact on traditional psychological parameters such as anxiety and depression appears relatively uncommon, it is critical for any genetic testing in childhood to promote the child’s best interests above all else.49 It remains important for clinicians to monitor for even subclinical levels of distress in their patients, because this distress may contribute to children’s cancer-related worries and could be amenable to psycho-educational and/or psychosocial support. Taking into consideration the family’s pre-existing risks of psychological distress, genetic counseling is essential in any case that involves the disclosure of genetic information to children.50 Because parents are usually best qualified to make decisions for their family, communication and collaboration with the child’s parents is essential to ensure the best outcomes when considering genetic testing in a child.
When considering testing children, the ‘earliest onset’ rule may be applied to protect their autonomy and their right to seek or decline genetic testing, defaulting to test the child at the earliest recorded age of condition onset.11 In some cases, it might be appropriate to consider timing testing to avoid overlapping with the often difficult transition into adolescence. In fact, some of the developmental processes that occur during adolescence (for example, desire for independence and wanting to “fit in” with peers) may make genetic testing at this time particularly difficult.17 For some children, undergoing genetic testing during adolescence may result in psychosocial challenges that could have been avoided if the child had learned his or her genetic status at a younger age.
Limited evidence is available with respect to the psychological implications for children acquiring genetic information when a family member undergoes a genetic test. The most significant adverse findings were an increase in anxiety in one study35 and concerns about cancer risk in another.26 Nonetheless, we know from studies of children with cancer that concealing difficult information builds mistrust. Thus, children need honest information about the genetic test results and clinical status of close family members, but both providers and parents may need help in initiating these conversations and checking to assess the level and nature of the worry of the child over time. Given the lack of evidence base, however, careful clinical support and genetic counseling to support family disclosure are essential.48
Limitations
The broad search terms used for this review led to a lack of specificity in the search, resulting in the identification of few eligible studies from the large pool of captured abstracts (20 eligible articles captured out of 694 abstracts, resulting in a specificity of 2.9% (20/694)). The broad search terms did, however, allow for good sensitivity achieved in the search, with 20/23 eligible articles (87.0%) captured directly by the electronic search. Due to practical limitations, some databases were not searched using all key words (see Supplementary Table S1 online). The inclusion criteria resulted in the omission of works published in different languages and studies published outside peer reviewed journals. Our findings may have been affected by publication bias if studies finding null results were not published and therefore were not captured in this review.51
Many genetic conditions were not investigated or were under-represented in the captured studies, including thalassemia, von Hippel–Lindau syndrome, and Li–Fraumeni syndrome, among others. Also missing was consideration of the potentially different impact of providing genetic testing for conditions whereby the penetrance (and, thus, the disease risk) is unclear or for conditions that have few available treatments or intervention options to manage/reduce the child’s risk. In the era of genomic testing it is reassuring that although there are limited data, there is as yet no convincing evidence of harm.
Future Directions
The empirical data captured by the review did not fully support the perspectives published by leaders in the field; however, strong conclusions are premature given the scarcity of studies, the lack of consistent methodology used across studies, and the rarity of longitudinal data. Many important concerns are yet to be adequately assessed, and it will be critical to examine these in future studies. For example, few (if any) studies have addressed potential disruptions to children’s developing identities, family dynamics, parent-child or sibling-sibling relationships, or survivor guilt. Studies also did not fully address children’s concerns about (or experience of) genetic discrimination. Additional research focused on children’s self-reported experiences and the relationship between child reports and parent reports would be useful given the potential for parent-proxy assessments to yield different data than that reported directly by children.52,53
The review also did not capture any studies of the psychological impact of genomic testing in childhood, when potential psychological implications are more complex. With genomic sequencing and testing, clinicians are usually aiming to identify or exclude a particular diagnosis; however, there is a chance that an incidental germline mutation for an inherited disorder of adult onset may be shown serendipitously. This type of testing may, for example, reveal information about a child’s BRCA1 or BRCA2 mutation status (usually not tested for in childhood), revealing potentially important information about a treatable mutation in the parent. In cases such as these, the clinician will need to weigh the potential benefit to the child’s parent against the lack of evidence of benefit or harm to the child.21
In light of concerns about potential harms of genetic testing in children, the overwhelming majority of empirical studies have, to date, focused on the potential negative impact of genetic testing. The responsible integration of genetics-related technologies into the clinical setting requires comprehensive knowledge of psychosocial concerns related to the testing of children. Given that the empirical studies captured for the review reported limited negative impact on psychological functioning in a number of different domains, an additional focus on investigating the potential benefits of testing is also warranted.
Conclusions
New technologies are changing the way we diagnose, treat, and prevent genetic disease. Given the possible range and complexity of the impact of genetic information on children, a variety of perspectives exist among health professionals with respect to offering genetic testing to children and their family members. This variety is reflected in diverse current clinical practices and policies, which are often driven by individual clinician experiences/opinions, which may or may not be fully supported by the available evidence.
The short-term assessment of psychological outcomes largely revealed few adverse outcomes. However, it remains premature to conclude that similar results during long-term follow-up will be equally favorable.14 It is possible that there are potential unique complexities of genetic testing in children that have not yet been captured by studies completed to date. The psychological impact of newly available genetic tests for an ever-broadening number of diseases is yet to be determined. Similarly lacking is evidence regarding the longer-term psychological impact of genetic testing in children, which may not appear until the child has reached adulthood or particular milestones (e.g., considering marriage or starting their own family). Although current, limited evidence suggests clinical levels of anxiety or depression are not likely to be common in the long-term for most children tested, longitudinal follow-up studies that further explore the psychological benefits and ramifications of children acquiring personal and familial genetic information would be judicious before conclusions are drawn about the impact of genetic testing on children.
Disclosure
The authors declare no conflict of interest.
References
Beery TA, Williams JK. Risk reduction and health promotion behaviors following genetic testing for adult-onset disorders. Genet Test 2007;11:111–123.
Kefford RF, Newton Bishop JA, Bergman W, Tucker MA. Counseling and DNA testing for individuals perceived to be genetically predisposed to melanoma: A consensus statement of the Melanoma Genetics Consortium. J Clin Oncol 1999;17:3245–3251.
Bove CM, Fry ST, MacDonald DJ. Presymptomatic and predisposition genetic testing: ethical and social considerations. Semin Oncol Nurs 1997;13:135–140.
Hamilton JG, Lobel M, Moyer A. Emotional distress following genetic testing for hereditary breast and ovarian cancer: a meta-analytic review. Health Psychol 2009;28:510–518.
Meiser B. Psychological impact of genetic testing for cancer susceptibility: an update of the literature. Psychooncology 2005;14:1060–1074.
Schlich-Bakker KJ, ten Kroode HF, Ausems MG. A literature review of the psychological impact of genetic testing on breast cancer patients. Patient Educ Couns 2006;62:13–20.
Heshka JT, Palleschi C, Howley H, Wilson B, Wells PS. A systematic review of perceived risks, psychological and behavioral impacts of genetic testing. Genet Med 2008;10:19–32.
Eijzenga W, Hahn DE, Aaronson NK, Kluijt I, Bleiker EM. Specific psychosocial issues of individuals undergoing genetic counseling for cancer - a literature review. J Genet Couns 2014;23:133–146.
Malkin D. Surveillance for children at genetic risk for cancer: are we ready? Pediatr Blood Cancer 2014;61:1337–1338.
Aatre RD, Day SM. Psychological issues in genetic testing for inherited cardiovascular diseases. Circ Cardiovasc Genet 2011;4:81–90.
Bruwer Z, Algar U, Vorster A, et al. Predictive genetic testing in children: constitutional mismatch repair deficiency cancer predisposing syndrome. J Genet Couns 2014;23:147–155.
Lerman C, Croyle RT, Tercyak KP, Hamann H. Genetic testing: psychological aspects and implications. J Consult Clin Psychol 2002;70:784–797.
Cameron LD, Muller C. Psychosocial aspects of genetic testing. Curr Opin Psychiatry 2009;22:218–223.
van Dijk S, Otten W, Tollenaar RA, van Asperen CJ, Tibben A. Putting it all behind: long-term psychological impact of an inconclusive DNA test result for breast cancer. Genet Med 2008;10:745–750.
Samuel GN, Jordens CF, Kerridge I. Direct-to-consumer personal genome testing: ethical and regulatory issues that arise from wanting to ‘know’ your DNA. Intern Med J 2010;40:220–224.
MacDonald DJ, Lessick M. Hereditary cancers in children and ethical and psychosocial implications. J Pediatr Nurs 2000;15:217–225.
Duncan RE, Delatycki MB. Predictive genetic testing in young people for adult-onset conditions: where is the empirical evidence? Clin Genet 2006;69:8–16; discussion 17.
Borry P, Stultiens L, Nys H, Cassiman JJ, Dierickx K. Presymptomatic and predictive genetic testing in minors: a systematic review of guidelines and position papers. Clin Genet 2006;70:374–381.
Dalby S. GIG response to the UK Clinical Genetics Society report “The genetic testing of children”. J Med Genet 1995;32:490–491.
Strahm B, Malkin D. Hereditary cancer predisposition in children: genetic basis and clinical implications. Int J Cancer 2006;119:2001–2006.
Botkin JR, Belmont JW, Berg JS, et al. Points to Consider: Ethical, Legal, and Psychosocial Implications of Genetic Testing in Children and Adolescents. Am J Hum Genet 2015;97:6–21.
Clayton EW, McCullough LB, Biesecker LG, Joffe S, Ross LF, Wolf SM ; Clinical Sequencing Exploratory Research (CSER) Consortium Pediatrics Working Group. Addressing the ethical challenges in genetic testing and sequencing of children. Am J Bioeth 2014;14:3–9.
Michie S, Marteau TM. Predictive genetic testing in children: the need for psychological research. Br J Health Psychol 1996;1:3–14.
Geller G, Tambor ES, Bernhardt BA, Fraser G, Wissow LS. Informed consent for enrolling minors in genetic susceptibility research: a qualitative study of at-risk children’s and parents’ views about children’s role in decision-making. J Adolesc Health 2003;32:260–271.
Tercyak KP, Peshkin BN, Streisand R, Lerman C. Psychological issues among children of hereditary breast cancer gene (BRCA1/2) testing participants. Psychooncology 2001;10:336–346.
Cappelli M, Verma S, Korneluk Y, et al. Psychological and genetic counseling implications for adolescent daughters of mothers with breast cancer. Clin Genet 2005;67:481–491.
Herman JD, Appelbaum H. Hereditary breast and ovarian cancer syndrome and issues in pediatric and adolescent practice. J Pediatr Adolesc Gynecol 2010;23:253–258.
Michie S, Bobrow M, Marteau TM. Predictive genetic testing in children and adults: a study of emotional impact. J Med Genet 2001;38:519–526.
Wertz DC, Fanos JH, Reilly PR. Genetic testing for children and adolescents. Who decides? JAMA 1994;272:875–881.
Moher D, Liberati A, Tetzlaff J, Altman DG ; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol 2009;62:1006–1012.
Jennison MH, Coleman AB, Feiertag RB, et al. Age limits of pediatrics. Pediatrics. 1972;49:463.
Jackson C, Cheater FM, Reid I. A systematic review of decision support needs of parents making child health decisions. Health Expect 2008;11:232–251.
Bradbury AR, Patrick-Miller L, Egleston BL, et al. When parents disclose BRCA1/2 test results: their communication and perceptions of offspring response. Cancer 2012;118:3417–3425.
Codori AM, Zawacki KL, Petersen GM, et al. Genetic testing for hereditary colorectal cancer in children: long-term psychological effects. Am J Med Genet A 2003;116A:117–128.
Codori AM, Petersen GM, Boyd PA, Brandt J, Giardiello FM. Genetic testing for cancer in children. Short-term psychological effect. Arch Pediatr Adolesc Med 1996;150:1131–1138.
Jensen M, Havndrup O, Christiansen M, et al. Twelve year follow-up of clinical screening and predictive genetic testing for hypertrophic cardiomyopathy in children and adolescents. Eur Heart J 2011;32:27.
Järvinen O, Hietala M, Aalto AM, et al. A retrospective study of long-term psychosocial consequences and satisfaction after carrier testing in childhood in an autosomal recessive disease: aspartylglucosaminuria. Clin Genet 2000;58:447–454.
Smets EMA, Stam MMH, Meulenkamp TM, et al. Health-related quality of life of children with a positive carrier status for inherited cardiovascular diseases. Am J Med Genet, Part A 2008;146:700–707.
Gjone H, Diseth TH, Fausa O, Nøvik TS, Heiberg A. Familial adenomatous polyposis: mental health, psychosocial functioning and reactions to genetic risk in adolescents. Clin Genet 2011;79:35–43.
Järvinen O, Lehesjoki AE, Lindlöf M, Uutela A, Kääriäinen H. Carrier testing of children for two X-linked diseases: A retrospective study of comprehension of the test results and social and psychological significance of the testing. Pediatrics 2000;106:1460–1465.
Meulenkamp TM, Tibben A, Mollema ED, et al. Predictive genetic testing for cardiovascular diseases: impact on carrier children. Am J Med Genet A 2008;146A:3136–3146.
Andersen J, Sandberg S, Raheim M, Gjengedal E. Psychosocial aspects of predictive genetic testing for acute intermittent porphyria in minors. Br J Dermatol 2011;164:1133–1134.
Patenaude AF. The genetic testing of children for cancer susceptibility: ethical, legal, and social issues. Behav Sci Law 1996;14:393–410.
Grosfeld FJ, Lips CJ, Beemer FA, van Spijker HG, Brouwers-Smalbraak GJ, ten Kroode HF. Psychological risks of genetically testing children for a hereditary cancer syndrome. Patient Educ Couns 1997;32:63–67.
Wilfond B, Ross LF. From genetics to genomics: ethics, policy, and parental decision-making. J Pediatr Psychol 2009;34:639–647.
Robertson S, Savulescu J. Is there a case in favour of predictive genetic testing in young children? Bioethics 2001;15:26–49.
Malpas PJ. Predictive genetic testing of children for adult-onset diseases and psychological harm. J Med Ethics 2008;34:275–278.
Butow PN, Lobb EA, Meiser B, Barratt A, Tucker KM. Psychological outcomes and risk perception after genetic testing and counselling in breast cancer: a systematic review. Med J Aust 2003;178:77–81.
American Academy of Pediatrics. Ethical issues with genetic testing in pediatrics. Pediatrics 2001;107:1451–1455.
Hadley DW, Ashida S, Jenkins JF, et al. Generation after generation: exploring the psychological impact of providing genetic services through a cascading approach. Genet Med 2010;12:808–815.
Hopewell S, Loudon K, Clarke MJ, Oxman AD, Dickersin K. Publication bias in clinical trials due to statistical significance or direction of trial results. Cochrane Database Syst. Rev 2009:MR000006.
Hermont AP, Scarpelli AC, Paiva SM, Auad SM, Pordeus IA. Anxiety and worry when coping with cancer treatment: agreement between patient and proxy responses. Qual Life Res 2015;24:1389–1396.
Parsons SK, Fairclough DL, Wang J, Hinds PS. Comparing longitudinal assessments of quality of life by patient and parent in newly diagnosed children with cancer: the value of both raters’ perspectives. Qual Life Res 2012;21:915–923.
Wertz DC, Fanos JH, Reilly PR. Testing healthy children and adolescents: recommendations for avoiding harm. Genetic Resour 1994;8:16–20.
Acknowledgements
C.E.W. is supported by a Career Development Fellowship from the National Health and Medical Research Council of Australia (APP1067501) and an Early Career Development fellowship from the Cancer Institute of NSW (ID: 11/ECF/3–43). The Behavioral Sciences Unit is proudly supported by the Kids with Cancer Foundation. We also acknowledge the contribution of Bridget Cavanagh (B.C.) in the process of article selection and review.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
(ZIP 55 kb)
Rights and permissions
About this article
Cite this article
Wakefield, C., Hanlon, L., Tucker, K. et al. The psychological impact of genetic information on children: a systematic review. Genet Med 18, 755–762 (2016). https://doi.org/10.1038/gim.2015.181
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gim.2015.181
Keywords
This article is cited by
-
The case for screening in early life for ‘non-treatable’ disorders: ethics, evidence and proportionality. A report from the Health Council of the Netherlands
European Journal of Human Genetics (2022)
-
Should we respect parents’ views about which results to return from genomic sequencing?
Human Genetics (2022)
-
Polygenic risk scores in the clinic: new perspectives needed on familiar ethical issues
Genome Medicine (2021)
-
‘We Should View Him as an Individual’: The Role of the Child’s Future Autonomy in Shared Decision-Making About Unsolicited Findings in Pediatric Exome Sequencing
Health Care Analysis (2021)
-
Genetics-related service and information needs of childhood cancer survivors and parents: a mixed-methods study
European Journal of Human Genetics (2020)