Abstract
Purpose:
Autism spectrum disorders are associated with defects in social response and communication that often occur in the context of intellectual disability. Rett syndrome is one example in which epilepsy, motor impairment, and motor disturbance may co-occur. Mutations in histone demethylases are known to occur in several of these syndromes. Herein, we aimed to identify whether mutations in the candidate histone demethylase JMJD1C (jumonji domain containing 1C) are implicated in these disorders.
Methods:
We performed the mutational and functional analysis of JMJD1C in 215 cases of autism spectrum disorders, intellectual disability, and Rett syndrome without a known genetic defect.
Results:
We found seven JMJD1C variants that were not present in any control sample (~ 6,000) and caused an amino acid change involving a different functional group. From these, two de novo JMJD1C germline mutations were identified in a case of Rett syndrome and in a patient with intellectual disability. The functional study of the JMJD1C mutant Rett syndrome patient demonstrated that the altered protein had abnormal subcellular localization, diminished activity to demethylate the DNA damage-response protein MDC1, and reduced binding to MECP2. We confirmed that JMJD1C protein is widely expressed in brain regions and that its depletion compromises dendritic activity.
Conclusions:
Our findings indicate that mutations in JMJD1C contribute to the development of Rett syndrome and intellectual disability.
Genet Med 18 1, 378–385.
Similar content being viewed by others
Introduction
Autism spectrum disorders are a heterogeneous clinical and genetic group of neurodevelopmental defects that are characterized by impaired social communication functions and inappropriate repetitive behavior.1 This family of disorders is characterized by enormous phenotypic variability, from mild primary deficits in language pragmatics2 to major neurological phenotypes, such as that of Rett syndrome (OMIM 312750), where it co-occurs with epilepsy, motor impairment, and sleep disturbance.3 The disabilities associated with autism spectrum disorders are often so severe that affected individuals do not generally reach parenthood, thereby preventing comprehensive familial genetic studies from being undertaken. However, genetic alterations are already recognized as major etiological factors. In this regard, concordance with autism spectrum disorders is higher than with any other cognitive or behavioral disorder.4,5 In addition to the contribution of polymorphic variants that confer low or moderate risk of the appearance of these neurodevelopmental defects, a cause of autism spectrum disorders can be the occurrence of de novo mutations affecting genes in a number of cellular pathways.6,7,8 A similar scenario can be proposed for the genetic contribution to the even more heterogeneous group of disorders classified as intellectual disabilities.9
Among the described genetic defects associated with intellectual disabilities, our attention was caught by a single case report of an autistic patient carrying a de novo balanced paracentric inversion 46, XY in (10)(q11.1;q21.3) in which the distal breakpoint disrupted what was at that time known as the TRIP8 gene,10 which has been characterized as a member of the jmJC domain-containing protein family involved in the methyl group removal and renamed as JMJD1C (jumonji domain containing 1C).11,12,13 Support for a role of JMJD1C in autism has been further fostered by large-scale exome sequencing studies in which three de novo variants have been identified,14,15 at least one of which is a loss-of-function mutation.15 However, two of the three described variants occurred in unaffected siblings. To investigate whether JMJD1C mutations occur in intellectual disability, Rett syndrome, and autism spectrum disorders, we performed a comprehensive screening to identify single nucleotide changes and indels in a large collection of 215 patients in whom a genetic defect had not previously been identified.
Materials and Methods
Patients
The samples used in this study consisted of 215 patients of either gender with autism, Rett syndrome, or intellectual disability from the Hospital Clínic, Sant Joan de Deu and Pompeu Fabra University, Barcelona, Catalonia, Spain. A cohort of 500 healthy volunteers was obtained from the same geographic region (Catalonia, Spain). All were Caucasian individuals (such as in the case cohort) and were matched according to gender distribution (case cohort: female, 53% [114/215]; male, 47% [101/215]; control cohort: female, 58% [290/500]; male, 42% [210/500]). Ethical approval for the molecular genetic studies was obtained from each institutional review board. DNA was extracted from peripheral blood leukocytes using standard techniques. We measured DNA concentration with the Quant-iT Picogreen (Invitrogen, Life Technologies – Grand Island, NY) and then normalized all concentrations to 25–50 ng/μl before proceeding with the Access Array amplification.
Fluidigm access array
Forty-eight pairs of primers were designed using the Access Array Amplicon Tagging Assay design service from Fluidigm to cover all 26 exons of the JMJD1C gene (NM_032776.1), including exon-intron boundaries. These primers also contained the sequencing adaptors necessary for subsequent sequencing in the 454 GS Junior Sequencer.
Multiplex ligation-dependent probe amplification
Large rearrangements in the JMJD1C gene were studied using multiplex ligation-dependent probe amplification. We designed nine such probes specific to the JMJD1C gene and six control probes according to the instructions provided by MRC-Holland.
Sanger sequencing
The variants were validated by Sanger sequencing using a BigDye Terminator v3.1 Cycle Sequencing Kit (Life Technologies, Grand Island, NY) in an Applied Biosystems 3730/DNA Analyzer (Applied Biosystems – Life Technologies – Grand Island, NY). The raw data were analyzed with Codon Code Aligner Software (CodonCode Corporation – Centerville, MA).
Exome sequencing
The patient and healthy parents were analyzed by whole exome sequencing with TruSeq Sample Preparation Kit (Illumina, San Diego, CA). Exomes were captured with TruSeq Exome Enrichment Kit (Illumina) and paired-end 100 × 2 sequenced with the HiScan SQ (Illumina). Format DNA and Protein Sequence Alignment Qual (FASTQ) files were analyzed as described in Supplementary Methods online. The raw data were analyzed in Centre Nacional d’Anàlisi Genòmica (CNAG), Barcelona, Catalonia, Spain.
Cell culture and vectors
JMJD1C coding sequence in the pCMV6-AC-GFP vector was purchased from Origene (Rockville, MD) (RG214878). The mutants were generated with Mutant QuikChange Site-Directed Mutagenesis Kit (Agilent Technologies, Santa Clara, CA). Wild-type (WT) WT and Pro163Leu and His2336Ala mutants were subcloned in the pCMV6-entry vector to introduce Myc-DDK-tag. shRNAs against the coding sequence of the mouse Jmjd1c gene were cloned in the pLVX-shRNA2 vector between the BamHI and EcoRI restriction sites (shRNA24 target: CAGAGACTGCTTGAGGAAT). Hek293 cells were cultivated in Dulbecco’s modified Eagles medium with 10% fetal bovine serum. To generate stable WT or mutant clones, Hek293 cells were transfected with Lipofectamine 2000 (Invitrogen), selected with G418 antibiotic, and individual clones were isolated 2 weeks later. For transient expression, 6 mg of vector were transfected in 35-mm six-well plates with jetPRIME transfection reagent (Polyplus, NY) following the manufacturer’s instructions. Primary cultures of hippocampal neurons were prepared from neonate mice (P0) of either sex. Cultures were infected at 3DIV with lentiviral vectors to express scramble or shRNAs against JMJD1C together with a GFP tracer (pLVX-shRNA2 system). Coverslips were fixed, and protein was extracted at 15DIV.
Immunoprecipitation
Seven hundred fifty micrograms of chromatin fraction were diluted 10-fold in immunoprecipitation (IP) buffer (5 mmol/l Tris–HCl pH 7.6, 15 mmol/l HEPES pH 8.0, 1 mmol/l Ethylene diamine tetraacetic acid, 1 mmol/l ethylene glycol tetraacetic acid, 0.1% sodium dodecyl sulfate (SDS), 1% Triton X-100) incubated with 2 μg of antibodies anti-Me-Lysine (Abcam ab23366, Cambridge, UK) overnight at 4 °C and for 2 hours with PureProteome Protein A/G Magnetic Beads. Beads were washed twice with IP buffer and twice in RBS NP-40 and eluted in laemli buffer in reduction conditions at 70 °C for 10 minutes.
MeCP2 immunoprecipitation
For the MeCP2 immunoprecipitation procedure, anti-JMJD1C and anti-MeCP2 antibodies were coupled to Dynabeads Protein G (Invitrogen). JMJD1C transfected HEK293F cells were transiently transfected with MeCP2-Flag tagged plasmid and the nuclear fraction was obtained by RIPA buffer (10 mmol/l TRIS-Cl pH 8.0, 1 mmol/l EDTA (Ethylene diamine tetraacetic acid), 0.5 mmol/l EGTA (ethylene glycol tetraacetic acid), 1% Triton X-100, 0.1 % sodium deoxycholate, 0.1% SDS, and 140 mmol/l NaCl) supplemented with protease inhibitors (complete; Roche, Rotkreuz, Switzerland) and hybridized with the antibodies at 4 °C for 2 hours. Then, 150 mM NaCl phosphate-buffered saline (PBS) buffer was used for washing. Human IgG was used as negative control. Anti-Flag HRP (M2-SIGMA) antibody was used to visualize binding.
Western blot
Protein extract of Hek293 cells and primary neuronal culture were obtained in radioimmunoprecipitation assay (RIPA) buffer supplemented with a complete protease inhibitor cocktail tablet (Roche) and sonicated. Then, 50 μg of each protein sample were denatured in Laemli buffer 4% β-mercaptoethanol for 10 minutes at 95 °C, separated on a 7.5% or 15% SDS-polyacrylamide gel, and then transferred onto a polyvinylidene fluoride membrane (Immobilon-P; Millipore, Hessen, Germany) by liquid electroblotting. The antibodies and dilutions used were as follows: rabbit anti-JMJD1C 1:2,000 (09-817; Millipore); mouse anti-nucleolin 1:1,000 (SC-8031; Santa Cruz, Dallas, Texas); rabbit anti-H3 1:10,000 (Abcam AB1791); mouse anti-H3 1:4,000 (Abcam AB10799); and rabbit anti-MDC1 1:5,000 (Abcam AB11171). The blots were developed with Luminata Crescendo Western HRP substrate (MERCK MILLIPORE - Hessen, Germany) or with the LiCor Odyssey System (LI-COR – Bad Homburg, Germany).
Immunofluorescence
Cells were fixed in 4% paraformaldehyde–phosphate-buffered serum (PFA-PBS), quenched in 100 mmol/l glycine-PBS, and permeabilized with 0.25% Triton X-100, 1% bovine serum albumin, and PBS. The cells were blocked with 0.2% gelatin and 0.25% Triton X-100. Antibody dilutions were prepared in 0.25% Triton X-100, 1% bovine serum albumin, and PBS. The dilutions used were: rabbit anti-JMJD1C 1:200; chicken anti-Map2 1:5,000; and anti-β-tubulin 1:1,500 (Abcam AB21058). Nuclei were stained with 2 mg/ml Hoechst 33342. Coverslips were mounted in ProLong Gold antifade reagent (Molecular Probes – Life Technologies – Grand Island, NY). Confocal images were captured with a Leica SP5 confocal microscope (Leica, Buffalo Grove, IL). For fluorescence recovery after photobleaching analysis, the cells were maintained at 37 °C in an atmosphere of 5% CO2. We captured images every 70 seconds at 63×, at 128 × 128 resolution, and at 1,400 Hz with bidirectional acquisition. We captured 25 control images at 3% laser transmission before bleaching and then bleached the region of interest (ROI) inside a nucleus 25 times at a nominal level of 100% laser transmission. For this experiment, 150 images were captured after bleaching. The row data were analyzed with FrapAnalyzer software (Igor Pro 6.1 Software – WaveMetrics – Lake Oswego, OR).
RNA extraction and real-time PCR
Total RNA was extracted from cell lysates using TRIzol Reagent (Invitrogen), purified using the RNeasy Kit (Qiagen, Valencia, CA), and 2 µg were retrotranscribed using the ThermoScriptTM RT-PCR System (Invitrogen). Real-time PCR reactions were performed in triplicate on an Applied Biosystems 7,900HT Fast Real-Time PCR system using 20 ng cDNA, 5 µl SYBR Green PCR Master Mix (Applied Biosystem), and 150 nmol/l specific primers (sequences are available on request) in a final volume of 10 µl.
Results
JMJD1C mutational analyses in samples from autism spectrum disorders, intellectual disability, and Rett syndrome
We first collected blood samples in EDTA tubes for DNA extraction from 215 patients with autism spectrum disorders (n = 69; 58 males and 11 females), intellectual disability (IQ < 70) (n = 85; 43 males and 42 females), or Rett syndrome (n = 61; all females) without mutations in the disease-associated genes MECP2, CDKL5, and FOXG1.16 Approval for this was obtained from the corresponding institutional review boards. The presence of single-nucleotide changes and indels in the JMJD1C gene was analyzed by sequencing using a GSJUNIOR system with an amplicon library prepared with the Fluidigm Access Array, and the presence of larger genetic defects was assessed by multiplex ligation-dependent probe amplification. We detected no major genomic defects at the JMJD1C gene locus using the multiplex ligation-dependent probe amplification approach. Identified synonymous variants for JMJD1C are shown in Supplementary Table S1 online and seven previously informed JMJD1C nucleotide variants are described in Supplementary Table S2 online. Most importantly, the sequencing strategy identified seven nucleotide changes in the exonic regions of JMJD1C that were not present in the NHLBI Exome Sequencing Project (ESP) Exome Variant Server (~6,000 control samples) (Supplementary Table S3 online). These seven nucleotide changes were also absent in the 1000 Genomes Project17 and the NCBI database of genetic variation.18 Furthermore, we sequenced 500 healthy volunteers for these seven missense variants and none of the studied samples showed the described changes (Supplementary Table S3 online and Supplementary Figure S1 online). Not finding prior documentation of a variant in a mutation database is not in itself evidence of pathogenicity, and most missense variants are rare due to factors such as rapid population growth and weak purifying selection.19 Interestingly, four of our seven (57%) missense mutations were clustered in the exon 10 of JMJD1C, where the only two previously reported JMJD1C missense mutations were also found14,15 ( Figure 1a ). In addition, the seven single-nucleotide shifts caused an amino acid change involving a different functional group (Supplementary Table S3 online) that could alter important domains of the JMJD1C protein, such as the nuclear localization signal and the JmjC hydroxylase domain ( Figure 1a ). Unfortunately, for the understanding of the functional consequences of the JMJD1C mutations undergoing study, the 3D structure of this protein is only available for the C-terminal end (amino acid positions from 2,157 to 2,497) corresponding to the JmJC domain.20 From the described mutations only c. 6997A>G (T/A 2333) (Supplementary Table S3 online) is included in the available structure, and the identified change affects a critical amino acid in one of the beta strands that confers the core beta barrel for histone substrate interaction ( Figure 1b ). Conventional Sanger sequencing confirmed the GSJUNIOR sequencing results ( Figure 1c ). The seven point mutations described occurred in one case of Rett syndrome that was not associated with MECP2, CDKL5, and FOXG1 defects, in three autistic patients, and in three patients with intellectual disability (Supplementary Table S3 online).
Diagram of JMJD1C and mutations found. (a) Black asterisks indicate the position of mutations identified in the JMJD1C gene. The three previous mutations identified by Iossifov et al. and Neal et al. are indicated by green and orange asterisks, respectively. The most important motif and domains are: NLS_Bp, bipartite nuclear localization signal; C1_2, phorbol esther/diacylglycerol-binding domain; AR, androgen receptor-interacting zone; LxxL, motif involved in transcriptional regulation; JMJC, JmjC hydroxylase domain. (b) JMJD1C 3D structure for the C-terminal end corresponding to the JmjC domain derived from the Protein Data Bank (PDB) code 2YPD, X-Ray diffraction data for 2.1 Angstroms resolution. The T2333 amino acid is indicated by a black arrow. (c) Chromatograms of Sanger sequencing showing c.2830C>T, c.3559A>G, and c.3743A<G mutations.
Our experimental, genetic, and clinical experiences with Rett syndrome enabled us to select the newly identified JMJD1C nucleotide change in this severe form of autism spectrum disorder and further characterize its functional relevance. The c. 488C>T nucleotide change in exon 4 of the JMJD1C gene causes a proline-to-leucine shift in codon 163 of the protein. The potential pathogenic involvement of the JMJD1C-Pro163Leu change is also suggested because the wild-type proline amino acid is highly conserved in all mammalian JMJD1Cs ( Figure 2a ), and because it is included in the balanced JMJD1C inversion that occurs in the aforementioned autistic patient.10
Our patient with the JMJD1C-Pro163Leu mutant is a 29-year-old female who was born at term, weighed 3,800 g, and presented a 9/10 APGAR value. She showed stagnation in head growth and also in normal development at 14 months old and lost social interaction at 18 months old. The patient started to use single words at 20 months old and propositive sentences at 24 months old, but only babbled at 36 month old. The patient also presented gait dyspraxia, hand-washing stereotype, learning impairment, teeth grinding, air swallowing, kyphoscoliosis, and tonic epilepsy. She was diagnosed as having classical Rett syndrome from the clinical standpoint, but without mutations in the known MECP2, CDKL5, and FOXG1 Rett-associated genes. Sanger sequencing of the progenitors showed that neither of the parents was a carrier of the described JMJD1C nucleotide change ( Figure 2b ). Thus, the described mutation can be considered a de novo germline event. In this regard, from the other five identified JMJD1C missense changes that were not present in the NHLBI Exome Variant Server, we identified by conventional Sanger sequencing an additional germline de novo mutation. The c. 3559A>G nucleotide change (T/A amino acid change) occurred in a patient with intellectual disability (case 4 in Supplementary Table S3 online) but it was not present in the parents (Supplementary Figure S2 online). In a recently developed statistical framework to distinguish disease-relevant mutations from background variation due to random expectation,21 JMJD1C (NM_032776) was reported as having a mutation probability of 9.0804e-05.21 Thus, out of 215 samples we would expect to find 0.0195 mutated, so discovering 2 de novo mutations out of 215 samples is higher than expected by chance (102.4-fold, binomial test, P = 1.9e-4). One possible way to explain the higher rate of observed JMJD1C mutations than that in the described study21 could be related to the fact that the previously analyzed 5,000 samples corresponded to autism spectrum disorders,21 whereas our work jointly profiled autism with Rett syndrome and intellectual disability. Most importantly, our two de novo mutations were only identified in these last two entities. Thus, the identified JMJD1C mutation rate could match the one reported21 because we did not find any de novo mutation in our 69 cases of autism spectrum disorders. Interestingly, when we applied the PMUT predictor for these two de novo JMJD1C mutations,22 a neural network trained with neutral and pathogenic mutations extracted from the protein database SwissVar, we found that both of them were considered potentially pathogenic. To discard any other pathogenic mutation in our Rett syndrome patient, the patient and the healthy parents were analyzed by whole exome sequencing as described in Methods. The only de novo mutation identified in the patient was the described c. 488C>T nucleotide change in exon 4 of the JMJD1C. The complete exome sequencing data of these three samples are available at the Sequence Read Archive (http://www.ncbi.nlm.nih.gov/sra) under the codes SRX667201 (patient), SRX667384 (mother), and SRX667386 (father).
Functional analyses of a Rett syndrome–associated JMJD1C-Pro163Leu mutation
To address the effects of the identified JMJD1C-Pro163Leu mutation, we first studied the intracellular localization of the protein. Endogenous JMJD1C was mainly cytoplasmic in Hek293 cells ( Figure 3a ), a pattern that was also observed in the transfected JMJD1C-WT-GFP in stable clones ( Figure 3a ). However, clones expressing the mutant JMJD1C-Pro163Ala-GFP forms show a strong nuclear mark ( Figure 3a ). To characterize the subcellular scenarios in which the mutant JMJD1C protein was located in more detail, we fractionated cytoplasmic, nuclear, and chromatin-bound proteins. We found that if endogenous and transfected JMJD1C proteins were almost absent from the chromatin fraction, then the mutant JMJD1C-Pro163Ala-GFP was markedly enriched in the chromatin ( Figure 3b ). The use of the fluorescence recovery after photobleaching assay in nuclear areas confirmed that mutant JMJD1C-Pro163Ala-GFP had reduced recovery in comparison with the JMJD1C wild-type transfected protein ( Figure 3c ). Most importantly, we also found that the mutant JMJD1C-Pro163Ala protein was less efficient in demethylating a non-histone target of JMJD1C that has been recently identified, MDC1 (mediator of DNA-damage checkpoint 1), a regulator of the RAP80-BRCA1 branch of the DNA damage-response pathway.13 Using immunoprecipitation with an antibody against methylated lysines followed by western blot with the MDC1 antibody, we observed that Hek293 cells overexpressing the wild-type JMJD1C protein efficiently demethylated the MDC1 protein but that the mutant JMJD1C-Pro163Ala-GFP form showed diminished activity ( Figure 3d ). Thus, the studied JMJD1C mutation might also impair the repair of DNA damage in the studied patient. However, JMJD1C could also exert additional functions. In this regard, using JMJD1C-transfected HEK293F cells that we have co-transfected with MECP2-Flag, we observed the interaction between JMJD1C and MECP2 by using the immunoprecipitation assay ( Figure 3e ). Importantly, the JMJD1C-Pro163Leu mutant protein cannot efficiently bind to MECP2 ( Figure 3e ). These findings could explain the role of JMJD1C in Rett syndrome, a disease mainly associated with de novo mutations in MECP2.3
Differential subcellular localization, processing, and chromatin binding of JMJD1C-Pro163. (a) Upper panels show immunofluorescence with anti-JMJD1C antibody in untransfected cells showing that the endogenous protein is localized predominantly in the cytoplasm; middle panels show clones expressing JMJD1C-WT- GFP, which is also localized in the cytoplasm; bottom panels show that clones expressing JMJD1C-P163-GFP have a nuclear signal. (b) Clones and control cells were fractionated to separate the cytoplasm (C), nuclear (N), and chromatin (Chr) fractions. Proteins were processed for western blot and blotted with compartment markers (upper panel) and with anti-JMJD1C (lower panel). JMJD1C-WT-GFP looks like endogenous protein (black arrow), but JMJD1C-P163-GFP shows several processed bands in nucleus and chromatin (green arrows). (c) Normalized fluorescence recovery after photobleaching assay (FRAP) in clones overexpressing JMJD1C-WT-GFP and JMJD1C-P163-GFP. The bleached area was situated above the nuclei. The mutant P163L recovers more slowly than WT and has a larger immobile fraction. (d) Chromatin fractions from Hek293 control cells and Hek293 clones expressing JMJD1C-WT-GFP and JMJD1C-Pro163Leu-GFP were immunoprecipitated with anti-Methyl-lysine antibodies and blotted with anti-MDC1. (e) JMJD1C transfected HEK293F cells were co-transfected with MeCP2-Flag and subjected to immunoprecipitation assay with anti-JMJD1C antibody, anti-MeCP2 antibody, and control rabbit IgG. The western blot using anti-Flag shows the interaction between JMJD1C and MeCP2 when JMJD1C is immunoprecipitated (top). JMJD1C-Pro163Leu mutant protein cannot efficiently bind to MECP2 (below). B, bound fraction; UB, unbound fraction.
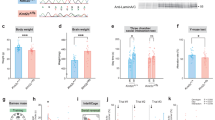
Once we had shown the aberrant functions of the mutant JMJD1C protein in the above model, we wondered whether the disruption of the wild-type JMJD1C protein had any cellular effect on a neuronal system. We first confirmed that JMJD1C was expressed in different brain regions by examining the Allen Brain Atlas Database (http://www.brain-map.org/) ( Figure 4a ).23 We also validated the widespread expression of JMJD1C throughout distinct regions of the mouse ( Figure 4b ) and human ( Figure 4c ) brain by quantitative RT-PCR. Then, we used the short hairpin RNA (shRNA) approach to analyze the impact of its loss in neurons. Lentiviral shRNA-mediated depletion of JMJD1C was performed in primary neuronal cultures obtained from neonatal mouse hippocampus and JMJD1C protein downregulation was observed using western blot ( Figure 5a ) and immunofluorescence ( Figure 5b ). We studied the existence of changes in the dendrites in these JMJD1C-impaired cells. A significant reduction in the complexity of the dendritic process is known to be a common hallmark in Rett syndrome.24 In this regard, and using immunofluorescence for the dendrite marker Map2, we found that JMJD1C shRNA-depleted neurons showed diminished dendritic branching in comparison with shRNA-scrambled control neurons ( Figure 5c , d ). Thus, a functional JMJD1C protein seems to be required for a correct dendritic pattern, and a defect in this gene could be associated with the dendritic impairment observed in autistic patients.
JMJD1C expression in human and mouse brain regions. CXF refers to frontal cortex; CP, Caudatus-Putamen; TH, thalamus; HIP, hippocampus; MB, midbrain; CB, cerebellum. (a) ISH analysis performed using a 2-month-old male mouse sample extracted from the Allen Brain Atlas Database (©2014 Allen Institute for Brain Science. Allen Mouse Brain Atlas [Internet]. Available at http://mouse.brain-map.org/). (b) Quantitative RT-PCR using three 18-month-old female samples. The y axis shows the fold-change in JMJD1C RNA expression, normalized with respect to thalamus expression, in relation to the RPL38 housekeeping gene. Error bars represent the SEM across different samples. (c) Quantitative RT-PCR using a sample from a 64-year-old male. The y axis shows the fold-change in JMJD1C RNA expression, normalized with respect to thalamus expression, in relation to the GUSB housekeeping gene. Error bars represent the SEM across different experiments.
JMJD1C expression in primary neuron culture and shRNA knockdown. 3DIV neuron primary cultures were infected with lentiviral vectors expressing shRNAs against JMJD1C or scramble shRNA. Neurons were maintained in 14DIV culture. (a) Western blot shows the bands of JMJD1C and the effect of a scramble shRNA and shRNA against JMJD1C on the exon 24. Only the shRNA24 reduces the expression. Anti-H3 histone antibodies were used for normalization. (b) Immunofluorescence against JMJD1C. Neurons infected with shRNA24 reduce the expression of JMJD1C; infected neurons were identified with GFP tracer under CMV promoter in viral vector and nuclei stained with Hoechst3342. (c) Map2 immunostaining showing dendritic branching in primary cell culture neurons transfected with JMJD1C shRNA24 or scramble shRNA (SCR; Control). JMJD1C downregulation via shRNA24 induced a marked reduction of the Map2 signal, demonstrating a decrease in the length of neuronal dendrites (red). (d) The dendritic length (Map2 staining) in primary cell culture neurons transfected with JMJD1C shRNA24 or scramble shRNA was calculated using IMAGEJ software. Five fields per condition were analyzed and the results are plotted as a bar graph. Bar errors represent SEM across different fields and P values are indicated. shRNA-mediated downregulation of JMJD1C significantly decreased dendritic length.
Discussion
Our findings suggest that mutations in the candidate histone demethylase JMJD1C contribute to the development of intellectual disability, including well-defined clinical entities such as Rett syndrome in those cases in which the usual mutations in MECP2, CDKL5, and FOXG1 are not present. Importantly, from a functional standpoint, a JMJD1C mutant protein is unable to correctly demethylate the MDC1 DNA repair-response protein but the wild-type JMJD1C protein plays a key role in dendritic activity.
It is interesting to note that many of the genes mutated in intellectual disability have an epigenetic component,25,26 and JMJD1C can now be included in this growing list. Epigenetics can be broadly defined as the inheritance of gene activity that does not depend on the “naked” DNA sequence, and it includes the control of gene transcription by several chemical marks, such as DNA methylation and histone modifications. Examples include the methyl-CpG-binding protein MeCP2 in Rett syndrome, the chromatin remodeling protein CHD7 in CHARGE syndrome, and the histone methyltransferase NSD1 in Sotos syndrome.25,26 The latter disorder is a representative case of how the importance of post-translational modifications of histone N-terminal tails in the genetic origin of neurodevelopmental disorders is becoming increasingly recognized. In this context, two mental retardation syndromes have been linked to mutations in the histone lysine demethylases KDM5C (previously known as JARID1C)27 and PHF8 (also known as JHDM1F).28 Thus, it is possible that mutations in many other members of the large histone demethylase family are involved in the development of intellectual disability.29 In this regard, although further research is required to understand the molecular pathways that involve JMJD1C, our work highlights the increasing contribution of the genetic disruption of epigenetic genes to human neurodevelopmental disorders.
Disclosure
The authors declare no conflict of interest.
References
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. American Psychiatric Publishing: Arlington, VA, 2013.
Anderson DK, Lord C, Risi S, et al. Patterns of growth in verbal abilities among children with autism spectrum disorder. J Consult Clin Psychol 2007;75:594–604.
Bedogni F, Rossi RL, Galli F, et al. Rett syndrome and the urge of novel approaches to study MeCP2 functions and mechanisms of action. Neurosci Biobehav Rev 2014;46:187–201.
Bailey A, Le Couteur A, Gottesman I, et al. Autism as a strongly genetic disorder: evidence from a British twin study. Psychol Med 1995;25:63–77.
Rosenberg RE, Law JK, Yenokyan G, McGready J, Kaufmann WE, Law PA. Characteristics and concordance of autism spectrum disorders among 277 twin pairs. Arch Pediatr Adolesc Med 2009;163:907–914.
Zafeiriou DI, Ververi A, Dafoulis V, Kalyva E, Vargiami E. Autism spectrum disorders: the quest for genetic syndromes. Am J Med Genet B Neuropsychiatr Genet 2013;162B:327–366.
Jeste SS, Geschwind DH. Disentangling the heterogeneity of autism spectrum disorder through genetic findings. Nat Rev Neurol 2014;10:74–81.
Ronemus M, Iossifov I, Levy D, Wigler M. The role of de novo mutations in the genetics of autism spectrum disorders. Nat Rev Genet 2014;15:133–141.
Flore LA, Milunsky JM. Updates in the genetic evaluation of the child with global developmental delay or intellectual disability. Semin Pediatr Neurol 2012;19:173–180.
Castermans D, Vermeesch JR, Fryns JP, et al. Identification and characterization of the TRIP8 and REEP3 genes on chromosome 10q21.3 as novel candidate genes for autism. Eur J Hum Genet 2007;15:422–431.
Wolf SS, Patchev VK, Obendorf M. A novel variant of the putative demethylase gene, s-JMJD1C, is a coactivator of the AR. Arch Biochem Biophys 2007;460:56–66.
Kim SM, Kim JY, Choe NW, et al. Regulation of mouse steroidogenesis by WHISTLE and JMJD1C through histone methylation balance. Nucleic Acids Res 2010;38:6389–6403.
Watanabe S, Watanabe K, Akimov V, et al. JMJD1C demethylates MDC1 to regulate the RNF8 and BRCA1-mediated chromatin response to DNA breaks. Nat Struct Mol Biol 2013;20:1425–1433.
Neale BM, Kou Y, Liu L, et al. Patterns and rates of exonic de novo mutations in autism spectrum disorders. Nature 2012;485:242–245.
Iossifov I, O’Roak BJ, Sanders SJ, et al. The contribution of de novo coding mutations to autism spectrum disorder. Nature 2014;515:216–221.
Guerrini R, Parrini E. Epilepsy in Rett syndrome, and CDKL5- and FOXG1-gene-related encephalopathies. Epilepsia 2012;53:2067–2078.
Consortium T 1000 GP. An integrated map of genetic variation from 1,092 human genomes. Nature 2012;491:56–65.
Sherry ST, Ward MH, Kholodov M, et al. dbSNP: the NCBI database of genetic variation. Nucleic Acids Res 2001;29:308–311.
Tennessen JA, Bigham AW, O’Connor TD, et al.; Broad GO; Seattle GO; NHLBI Exome Sequencing Project. Evolution and functional impact of rare coding variation from deep sequencing of human exomes. Science 2012;337:64–69.
Vollmar M, Johansson C, Krojer T, et al. Crystal structure of the Jumonji domain of human Jumonji domain containing 1C protein. Protein Data Base PDB Id: 2YPD. 2012.
Samocha KE, Robinson EB, Sanders SJ, et al. A framework for the interpretation of de novo mutation in human disease. Nat Genet 2014;46:944–950.
Ferrer-Costa C, Gelpí JL, Zamakola L, Parraga I, de la Cruz X, Orozco M. PMUT: a web-based tool for the annotation of pathological mutations on proteins. Bioinformatics 2005;21:3176–3178.
Lein ES, Hawrylycz MJ, Ao N, et al. Genome-wide atlas of gene expression in the adult mouse brain. Nature 2007;445:168–176.
Percy AK. Rett syndrome. Current status and new vistas. Neurol Clin 2002;20:1125–1141.
Sanchez-Mut JV, Huertas D, Esteller M. Aberrant epigenetic landscape in intellectual disability. Prog Brain Res 2012;197:53–71.
Urdinguio RG, Sanchez-Mut JV, Esteller M. Epigenetic mechanisms in neurological diseases: genes, syndromes, and therapies. Lancet Neurol 2009;8:1056–1072.
Jensen LR, Amende M, Gurok U, et al. Mutations in the JARID1C gene, which is involved in transcriptional regulation and chromatin remodeling, cause X-linked mental retardation. Am J Hum Genet 2005;76:227–236.
Laumonnier F, Holbert S, Ronce N, et al. Mutations in PHF8 are associated with X linked mental retardation and cleft lip/cleft palate. J Med Genet 2005;42:780–786.
De Rubeis S, He X, Goldberg AP, et al.; DDD Study; Homozygosity Mapping Collaborative for Autism; UK10K Consortium. Synaptic, transcriptional and chromatin genes disrupted in autism. Nature 2014;515:209–215.
Acknowledgements
This study was supported by the European Community’s Seventh Framework Program (FP7/2007–2013) under grant agreement PITN-GA-2012–316758 of the EPITRAIN project and PITN-GA-2009–238242 of DISCHROM; ERC grant agreement 268626 of the EPINORC project; the E-RARE EuroRETT network (Carlos III Health Institute project PI071327); the Fondation Lejeune (France); MINECO projects SAF2011-22803 and CSD2006-00049; the Cellex Foundation; the Botín Foundation; the Catalan Association for Rett Syndrome; Fundación Alicia Koplowitz 2011 Grant AKOPLOWITZ11_006; the FIS project PI1002512; Grants PI10/01422, PI13/00285, CA10/01474, RD06/0020/1050, RD12/0036/008, and 2009-SGR293; and the Health and Science Departments of the Catalan government (Generalitat de Catalunya). K.S. and P.P. are EPITRAIN Research Fellows. M.E. is an ICREA Research Professor.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
(PDF 308 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Sáez, M., Fernández-Rodríguez, J., Moutinho, C. et al. Mutations in JMJD1C are involved in Rett syndrome and intellectual disability. Genet Med 18, 378–385 (2016). https://doi.org/10.1038/gim.2015.100
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gim.2015.100
Keywords
This article is cited by
-
Copy Number Variants in Patients with Autism and Additional Clinical Features: Report of VIPR2 Duplication and a Novel Microduplication Syndrome
Molecular Neurobiology (2017)
-
Whole exome sequencing of Rett syndrome-like patients reveals the mutational diversity of the clinical phenotype
Human Genetics (2016)