Abstract
Abstract. Osteogenesis imperfecta is a systemic heritable disorder of connective tissue whose cardinal manifestation is bone fragility. In approximately 90% of individuals with osteogenesis imperfecta, mutations in either of the genes encoding the pro-α1 or pro-α2 chains of type I collagen (COL1A1 or COL1A2) can be identified. Of those without collagen mutations, a number of them will have mutations involving the enzyme complex responsible for posttranslational hydroxylation of the position 3 proline residue of COL1A1. Two of the genes encoding proteins involved in that enzyme complex, LEPRE1 and cartilage-associated protein, when mutated have been shown to cause autosomal recessive osteogenesis imperfecta, which has a moderate to severe clinical phenotype, often indistinguishable from osteogenesis imperfecta types II or III. Mutations in COL1A1 or COL1A2 which result in an abnormal protein still capable of forming a triple helix cause a more severe phenotype than mutations that lead to decreased collagen production as a result of the dominant negative effect mediated by continuous protein turnover. The current standard of care includes a multidisciplinary approach with surgical intervention when necessary, proactive physiotherapy, and consideration for the use of bisphosphonates all in attempts to improve quality of life.
Similar content being viewed by others
OVERVIEW
Osteogenesis imperfecta (OI) is a genetic disorder of the connective tissue matrix caused by abnormal collagen microfibril assembly. Several clinical subtypes of OI have been described based on the clinical, biochemical, and molecular nature of the disorder (Table 1). New research is emphasizing the structural interaction within the microfibril and identifying regions within the collagen, which play greater or lesser roles in the structural properties of the triple helix. In taking this information into account, clinical phenotypes resulting from certain mutations can be predicted because of this pathogenetic correlation.1 The clinical manifestations vary considerably, ranging from a severe perinatal lethal form to a mild disorder which only becomes evident in adulthood, manifesting as premature osteoporosis. Most commonly, however, OI presents in childhood with multiple fractures and related complications. The precise incidence of OI is unknown and reports vary from approximately 1/100,000 to 1/25,000 dependant on the criterion used to define OI.2,3 Severe forms and milder disease occur with approximately similar incidence. Severe and mild forms share the cardinal feature of bone fragility, which is characterized by bone fractures often after little or no trauma. Several findings in OI are common to other disorders of connective tissues; hypermobile joints and a blue hue to the sclerae are among these features frequently described. The incorporation of abnormal type 1 collagen in teeth results in brittle opalescent teeth, the hallmark of Dentinogenesis Imperfecta (DI), often seen in OI. Progressive conductive hearing loss in early adulthood is the result of damage to the ossicles in the middle ear; over time, hearing loss typically progresses and combined conductive and sensorineural hearing loss may be seen in adults, similar to that of otosclerosis. Short stature and bone deformity are common features of the disorder.
The mainstay of treatment is orthopedic management along with physiotherapy.4 Bisphosphonates are being evaluated for efficacy and clinical trials have shown improvement at least in bone mineral density (BMD). Gene-based therapies are still actively being pursued.
Clinical manifestations
In 1979, before the molecular understanding of the underlying collagen disorder, Sillence and Rimoin5 formulated a clinical classification, which is still in common use. This classification was largely based on clinical and radiologic subgrouping and has more recently been updated to reflect current pathogenetic understanding (Table 1).
Although this classification into types of OI is helpful in providing information about prognosis and management, the features of different types of OI overlap and subclassification is not necessarily an easy task. It should be noted that the severity of clinical and radiographic features lies on a continuum and that the “types” are defined using characteristics that appear to form clinical “nodes.” Interfamilial variability is apparent among individuals with the same OI type and intrafamilial variability is apparent among individuals with the same mutation. It should be noted, however, that there are essentially two pathogenic molecular mechanisms resulting in OI caused by collagen mutations. Chain exclusion, in which the mutant chain is not incorporated into the collagen triple helix results in the milder OI type I as the abnormal collagen microfibril is unable to be incorporated into the triple helix and is thus degraded leaving the remaining allele to produce less structurally intact collagen triple helix. The second mechanism is via chain nonexclusion in which the abnormal collagen chain results in a defective helix. The resultant dominant negative effect manifests as more severe OI types II, III, and IV.
Notwithstanding the advances in genetic and biochemical pathophysiology of OI, there is probably still utility in typing OI to provide patients and families with information about the expected clinical course.
OI type I
OI type I is characterized by blue sclerae and normal stature although affected individuals are often shorter than predicted by parental height. A small proportion of infants with OI type I have femoral bowing at birth. The first fractures may occur at birth or with diapering. More often, the first fractures occur when the infant begins to walk and, more importantly, to fall. Fractures generally occur at a rate of a few to several per year and then decrease in frequency after puberty. Fracture frequency often increases again in the 5th decade of life and accounts for more than 25% of lifetime fractures. This is especially noted in postmenopausal women and men beyond the 5th decade. Affected individuals may have anywhere from a few fractures to more than 100, but the fractures usually heal normally with no resulting deformity after appropriate orthopedic management.
Joint hypermobility may be present and may increase the risk of premature joint degeneration with resultant osteoarthritis and chronic joint pain. There may also be predisposition to develop scoliosis and chronic back pain is not uncommon.
DI is rare and distinguishes a subset of type I OI. DI predisposes teeth to premature wearing down, which interferes with chewing and has cosmetic implications. Dental eruption in OI type I can sometimes occur early.
Progressive hearing loss occurs in about 50% of adults with OI type I, beginning as a conductive hearing loss but becoming a mixed hearing loss in time.
OI type II
This is the perinatal lethal form of OI and although some fetuses die in utero or are spontaneously aborted, more typically, infants die in the immediate perinatal period. More than 60% of affected infants die on the first day; 80% die within the first week; survival beyond 1 year is exceedingly rare and usually involves intensive support such as continuous assisted ventilation. Death usually results from pulmonary insufficiency related to the small thorax, rib fractures, or flail chest because of unstable ribs. Those who survive the first few days of life may not be able to take in sufficient calories because of respiratory distress.
Abnormalities characteristic of OI type II are evident on prenatal ultrasound and at birth. Weight and length are small for gestational age. The sclerae are typically dark blue and connective tissue is extremely fragile. The skull is large for the body size and soft to palpation because of poor mineralization, historically referred to as a “ping-pong ball” skull. The anterior fontanelle is large and usually extends down to the metopic ridge. Callus formation on the ribs may be palpable. Extremities are short, bowed, or deformed. Hips are usually flexed and abducted in a “frog-leg” position. Radiographs show “crumpled” long bones and fractures in various stages of healing.
The inheritance of type II OI is typically autosomal dominant and thus most affected neonates represent new spontaneous mutations; however, germ line mosaicism has been described on several occasions and thus there is a risk of recurrence. Newly described autosomal recessive forms of OI may show clinical overlap with OI type II (see later).
OI type III
This is the most severe form of OI, which is compatible with survival into adulthood. The diagnosis of OI type III is often apparent at birth. Fractures in the newborn period, simply with handling of the infant, are common. Rarely, the number and severity of rib fractures lead to death from pulmonary failure within the first few weeks or months of life.
This disorder has also been referred to as the Progressive Deforming type of OI as many affected individuals have as many as 200 fractures and deformity progresses even in the absence of fracture. These deformities can reach 70–90° in long bones, either from mechanical forces exerted by muscle/tendon or angulation from prior fractures. Coxa vara, which has an overall incidence of 10% in OI, has been reported with increased frequency (55%) in type III OI.6 OI type III can be difficult to manage. The majority of affected individuals do not walk without assistance and many use a wheelchair because of severe bone fragility and marked bone deformity. Use of bisphosphonates may be changing the natural history of OI type III but this has not yet been well documented.
Growth velocity is slow and adult height is characterized by marked short stature, with some individuals achieving final adult height of less than 1 m.
Intellect is normal unless intracerebral hemorrhage has occurred.
Even within OI type III, considerable heterogeneity is observed at the clinical level. The distinction between an individual with mild type III OI and type IV OI is often subtle, and many clinicians use the ability to ambulate independently to differentiate the two. The mechanical strength of the bone as determined by nano indentation is the same in both subtypes and independent of bone type.7 Some individuals have normal-appearing teeth and facial appearance, whereas others have DI and relative macrocephaly with frontal bossing. Enlarged ventricles reflecting the soft calvarium and a flattened midface are frequently observed. Barrel chest deformity and kyphoscoliosis are also common. Scleral hue is variable, often blue or gray, and sclera can be blue in infancy but lighten with age. Hearing loss generally begins in the teenage years.
Basilar impression, an abnormality of the craniovertebral junction caused by descent of the skull on the cervical spine, is a known complication, the symptomatic form occurring rarely. Basilar impression is characterized by invagination of the margins of the foramen magnum upward into the skull, resulting in protrusion of the odontoid process into the foramen magnum. Basilar impression may progress to brainstem compression, obstructive hydrocephalus, or syringomyelia because of direct mechanical blockage of normal CSF flow. Basilar impression can cause headache with coughing, trigeminal neuralgia, loss of function of the extremities, or parasthesias. When swimming, individuals with basilar impression may perceive that water temperature differs below and above the umbilicus. Lhermitte's sign (tingling on neck flexion) can be demonstrated at any stage. At its most severe levels of involvement, sleep apnea and death can occur. Symptomatic basilar impression requires corrective surgery.8
OI type IV
OI type IV is characterized by variability in phenotype, ranging in severity from moderately severe to so mild that it may be difficult to make the diagnosis. Hallmarks of OI type IV are DI, which is common but may be mild; adult-onset hearing loss, and normal-to-gray sclerae. Stature is variable and may vary markedly within the family. Basilar impression can occur.
Newly described types of OI
The following subtypes of OI continue on the Sillence numerical classification. They have been subcategorized on the basis of the bone histology and certain clinical characteristics which aid differentiation. This classification scheme is likely to change with additional research on pathophysiology of OI.
OI type V
The distinguishing clinical features of OI type V include the common occurrence of radial head dislocation/subluxation and calcification of the interosseous membrane between the radius and ulna.9,10 These changes are not congenital but develop over time and they may not be found in all Type V patients. Affected individuals frequently have hyperplastic callus formation. An irregular pattern of bone lamellation is noted on polarized light microscopy. Although molecular pathogenesis has not been elucidated, it is important to note at this time that collagen electrophoresis is normal in these individuals which suggests that an associated protein within the matrix is most likely affected. OI type V accounts for about 5% of people with OI.
OI type VI
OI type VI has a moderate to severe phenotype associated with frequent fractures but is clinically indistinguishable from type IV OI. The hallmark is abnormal bone mineralization on histological examination.
OI type VII and VIII
These subtypes of OI are distinguished by autosomal recessive inheritance. Dysregulation of posttranslational hydroxylation of the proline residue at position 986 of COL1A1 has been implicated in the pathogenesis of these types of OI. This alpha hydroxylation is dependent on three proteins; cartilage-associated protein (CRTAP), prolyl-3-hydroxylase-1 (LEPRE1) and Cyclophylin B which form an active complex in the endoplasmic recticulum. Mutations in CRTAP have been assigned as being causative of OI VII, whereas mutations in LEPRE1 which encodes prolyl-3-hydroxylase-1 have been assigned to OI VIII, although these assignments seem tentative.11–16 To date, no pathogenic mutations in Cyclophylin B have been described in association with OI. The clinical features of these two subtypes are dependent on the remaining functional proteins and overlap in clinical severity with OI II and OI III. There are a few clinical indicators which suggest the diagnosis within the spectrum of moderately severe OI, and it may thus be difficult to differentiate these subtypes. Infants tend to have congenital fractures and significant undermineralization of their bones. Abnormal, “popcorn,” epiphyses have been described and speculated to result from abnormal hydroxylation of type I collagen in cartilage. This finding is not pathognomonic for these types of OI as it has been seen in families with apparent OI type III; it may be that when those individuals with apparent type III OI and popcorn epiphyses undergo CRTAP or LEPRE1 mutation analysis it will be found that they do not have OI type III. Affected individuals with recessive OI tend to be of short stature and rhizomelic with coxa vara and white sclera. The populations originally identified to harbor mutations in CRTAP or LEPRE1 were the Africans, African-Americans and Irish Traveler populations, but other ethnicities are represented as well. It remains unclear what percentage of OI is caused by CRTAP or LEPRE1 mutations.
Allellic connective tissue disorders
There have been several patients described with joint hypermobility and skin laxity, a clinical phenotype more in keeping with Ehlers Danlos Syndrome but who have an increased disposition to fractures. Mutations not involving the glycine residues within the triple helix of COL1 and mutations involving cysteine residues have been described in a subset of these patients.17–19 It is also felt that the N-terminal mutations in COL1 result in abnormal protein folding, which leads to a milder OI/Ehlers Danlos Syndrome phenotype.20
General clinical considerations
Facial features
Infants and children with OI are often described as having a triangular-shaped face. The skull is relatively large compared with body size and frontal bossing is frequently noted. Platybasia is commonly associated with basilar impression, a feature present in approximately 11% of individuals with types III and IV OI. Prognathism has also been frequently reported. The hue of the sclera was emphasized in previous classifications; this feature is, however, markedly variable and merely an artifact of collagen defects as a whole. It is believed to result from a combination of the increased opacity of the scleral tissues which allow varying degrees of retinal pigmentation to be visualized and altered light diffraction properties of the abnormal matrix proteins. Although this feature should alert the diagnostician to the possibility of an underlying collagen defect, it is not always useful to differentiate subtypes of OI.
Teeth
DI is characterized by translucent, discolored teeth which are fragile. They erupt earlier than in normal age matched control populations. If the deciduous are normal, the permanent teeth will also be normal, indicating that DI is inherent to the teeth and not a secondarily acquired anomaly. The abnormal dentin is believed to result from dysregulation of ADAMTS2 and COL1A1.21
Skin
Easy bruising is a generalized feature of connective tissue fragility and so can be a feature of OI.
Hearing
Mixed conductive and sensorineural hearing loss afflicts the majority of adults with OI. Childhood onset hearing loss affects approximately 7% of affected children between the ages of 5 and 9; progressive postpubertal hearing loss is more typical in OI. The initial conductive hearing loss results from fractures of the bones of the middle ear with contracture and scarring of the incus. With aging, sensorineural hearing loss compounds the preexisting conductive element. The fixation of the stapes is not unlike otosclerosis and surgical techniques such as stapedotomy used to treat otosclerosis have shown similar success in treating hearing loss in OI.22–24 It is interesting to note that in an animal model, changes in OI murine otic capsule are similar to those seen in otosclerosis.25 These changes have been noted along with concomitant changes in BMP3/NFKB1 gene expression, an avenue for exploration for future therapies. Some short-term gains have been described with cochlear implants.26 Biphosphonate therapy has not been shown to influence hearing loss.
Eyes
Visual acuity is usually normal, but there is a predisposition to both spontaneous and traumatic eye injury. Spontaneous scleral rupture and uveal prolapse has been documented and an awareness of the potential to eye injury may be emphasized.27 Scleral hue is often used as a diagnostic indicator for OI. The variability of this finding is marked and at risk of subjective assessment. It has been suggested to standardize scleral color using the Munsell system,28 but the value of this approach remains undetermined. It is not unusual for unaffected infants to exhibit blue sclera until approximately 15 months of age.
Gastrointestinal
Although complaints of constipation are common in children and adults with OI who are mobile in wheelchairs, it is not clear if this is a complication of OI itself or of the mode of transport. Bowel obstruction can occur as a result of protrusio acetabuli but seems to be uncommon.
Cardiopulmonary
Pulmonary insufficiency is the most common cause of demise in type II OI and affects a number of individuals with more severe type III OI. Both chest wall pathology and severe kyphoscoliosis associated with vertebral compression can contribute to restrictive lung disease. This may progress to pulmonary hypertension and subsequent cor pulmonale, requiring oxygen support. Mitral valve prolapse and aortic dilatation with or without regurgitation have also been reported. A recent report indicated figures as high as 95% with valvular regurgitation but this rarely causes serious morbidity unless there are compounding cardiovascular risks. Although both atrial and/or aortic rupture have been reported in OI; it is unclear whether they occur more commonly in individuals with OI than in the general population. A minority of individuals with OI have also been reported with coronary artery dissection or cerebral aneurysms, both of which are relatively common within the general population, the association thus remains unclear. Reduced mobility and wheel chair dependency can predispose to vascular stasis and an increased risk of pneumonias.
Musculoskeletal
Short stature is a feature of all types of OI, even in its mildest representation wherein adult height is only marginally reduced by familial standards. In the more severe presentations, vertical growth falls off the growth curves by the end of the first year of life and actual growth velocity is slower than the general population. A rare association with spondylolysthesis secondary to elongated pedicles has been described in the context of OI.29
Development
Delays in gross motor development are common in association with significant joint hypermobility and deforming types of OI, but this can improve with age. Development in other areas is usually normal. The increase in head circumference is most notable between the ages of 2 and 3. The presence of basilar invagination (BI) and its neurological sequelae have been described above (see Type III OI section).
Life expectancy
The most severely affected children with type II OI typically do not survive the neonatal period. Life expectancy for OI I and IV is normal. In OI III, the mean life expectancy may be shortened because of severe kyphoscoliosis and abnormal thoracic shape with attendant restrictive pulmonary disease and cardiac insufficiency.
Pregnancy
Fertility is normal in OI. For most women who have OI, pregnancy is uncomplicated. The exception is in those women with OI who are very small and require preterm cesarean section because of respiratory compromise. Infants with OI are more frequently delivered by cesarean section as a result of nonvertex presentation. No difference in the frequency of complications was found between vaginal or cesarean delivery.30 Joint laxity may increase, as it does with unaffected women, and reduce mobility in small moderately affected women.31,32 Bleeding is probably not more common than usual and complications of vaginal tearing during delivery are not common. It is uncertain whether postpartum pelvic relaxation is more common than usual. No adverse pregnancy outcomes have been recorded to date in woman with OI using bisphosphonates during pregnancy.33
Serum biochemistry
Serum concentrations of Vitamin D, calcium, phosphorous, and alkaline phosphatase are typically normal; however, the latter is often elevated in response to fracture and rarely alkaline phosphatase levels may be decreased in OI.
Establishing a diagnosis
As with any disorder, a detailed history is a crucial element of the diagnostic workup. This in combination with a physical examination focused toward anomalies encountered in connective tissue disorders and a radiologic review of the skeleton aids in formulating a diagnosis.
The following list of features would be typical in an individual presenting with OI.
-
Family history of OI or recurrent fractures
-
Fractures with minimal or no trauma in the absence of other factors, such as inflicted physical abuse or other known disorders of bone
-
Short stature or stature shorter than predicted based on stature of unaffected family members, often with bone deformity
-
Blue sclerae
-
DI
-
Progressive, postpubertal hearing loss
-
Ligamentous laxity and other signs of connective tissue abnormality
-
Family history of OI, usually consistent with autosomal dominant inheritance
-
Fractures of varying ages and stages of healing, often of the long bones but may also involve ribs and skull. The metaphyseal chip fractures characteristic of child physical abuse can be seen in a small number of children with OI
-
“Codfish” vertebrae, which are the consequence of spinal compression fractures, seen primarily in the adult
-
Wormian bones, defined as “sutural bones that are 6 mm by 4 mm (in diameter) or larger, in excess of 10 in number, with a tendency to arrangement in a mosaic pattern.” Wormian bones are suggestive of, but not pathognomic, for OI. They are present in up to 60% of affected children
-
Protrusio acetabuli, in which the socket of the hip joint is too deep and the acetabulum bulges into the cavity of the pelvis causing intrapelvic protrusion of the acetabulum
-
Low bone density detected by dual energy x-ray absorptiometry (DEXA). There seems to be a correlation between BMD and fracture risk in OI34,35
Note:
-
1
Bone density can be normal, especially in OI type I, as DEXA measures mineral content rather than collagen properties. DEXA, however, is not a straightforward quantitative measurement as the DEXA z-score reflects both the quantity and structural arrangement of the mineral content in the bone matrix which is qualitatively abnormal in types II, III, and IV OI.
-
2
A major determinant of bone density may be the affected individual's ability to ambulate.
-
3
Bone density standards for children are not yet available universally but with increased use of this modality in children, DEXA is gaining popularity as a useful modality in diagnosis and monitoring of OI.
A selective skeletal survey should be requested.
-
1
AP and lateral views of the long bones are examined for general bone quality, bowing, and healing fractures.
-
2
AP and lateral views of the spine are examined for kyphoscoliosis and/or vertebral compressions.
-
3
Two or three views of the skull should also be obtained to detect wormian bones.
Differential diagnosis
The differential diagnosis of OI is largely determined by the age of presentation and the clinical severity.
In utero
Prenatal ultrasound examination of severe OI may lead to a consideration of hypophosphatasia, thanatophoric dysplasia, campomelic dysplasia, and achondrogenesis as they all present with relative macrocephaly and rhizomelic shortening of the limbs. The appearance of the hands tends to be normal in OI. Experienced sonographers usually have little difficulty in identifying the crumpled long bones and beaded ribs which differentiate OI type II from these other disorders. Ventriculomegaly is often noted in OI.
Infancy and childhood
Nonaccidental trauma (child abuse)
OI needs to be distinguished from child physical abuse/nonaccidental trauma. The prevalence of physical abuse is much greater than the prevalence of OI and it has been reported that only 7% of children with unexplained fractures have an underlying medical condition. On rare occasion, both can occur in the same child. Patient history, family history, physical examination, radiographic imaging, and the clinical course all contribute to the distinction of OI from child abuse. The overlap in clinical features includes multiple or recurrent fractures, fractures that do not match the history of trauma, and the finding of fractures of varying ages and at different stages of healing. The continued occurrence of fractures in a child who has been removed from a possibly abusive situation lends support to the possibility of OI. Metaphyseal and rib fractures, thought to be virtually pathognomonic for child abuse, can occur in OI. The presence or absence of blue sclerae is unreliable in distinguishing OI from child abuse because blue sclerae are often found in unaffected normal infants until about 15 months of age; also children with OI type IV and recessive forms of OI may not have blue sclerae. Family history is often unrevealing; families suspected of possible child abuse often provide an unverified family history of frequent fractures; conversely, the family history of individuals with OI often does not reveal any other affected individuals because of a de novo mutation in the proband or the presence of a mild phenotype in relatives. Laboratory testing (protein-based studies or molecular genetic testing of COL1A1, COL1A2 and potentially LEPRE and CRTAP) often is not needed, and in some cases, the time required to perform such testing can delay proper disposition of child abuse cases.36
Infantile hypophosphatasia
Infantile hypophosphatasia (OMIM 2415000) is an autosomal recessive disorder that presents in infancy with an osteogenic defect characterized by micromelia, bowed limbs, and severe osteoporosis, including a poorly mineralized cranium. The sclerae are often blue and fractures are not uncommon. The distinction from severe OI is facilitated by the observation that serum biochemistry is abnormal in hypophosphatasia. Infantile hypophosphatasia is caused by mutations in tissue nonspecific alkaline phophatase, and thus, levels of the enzyme are low in association with high-inorganic pyrophosphate, whereas levels of alkaline phosphatase tend to be normal or raised in OI because of the increased rate of bone turnover.
Bruck syndrome
Bruck syndrome (OMIM 259450) is an autosomal recessive condition characterized by bone fragility, congenital joint contractures, clubfeet, normal or blue sclerae, and wormian bones. It results from defects in the lysyl hydroxylase (PLOD2) that hydroxylates the amino-terminal lysyl residues involved in crosslink formation.
Osteoporosis pseudoglioma syndrome
Osteoporosis pseudoglioma syndrome (OMIM 259770) includes bone fragility and fractures, other skeletal deformities, pseudoglioma with blindness in infancy, and other anomalies. It is caused by mutations in the gene encoding the lipoprotein receptor-related protein 5.
Cole-Carpenter syndrome
Cole-Carpenter syndrome (OMIM 112240) is characterized by bone deformities, multiple fractures, ocular proptosis, shallow orbits, orbital craniosynostosis, frontal bossing, and hydrocephalus.
Hadju-Cheney syndrome
Hadju-Cheney syndrome (OMIM 102500) is characterized by short stature, failure to thrive, conductive hearing loss, dysmorphic features, early tooth loss, genitourinary anomalies, osteopenia, pathologic fractures, wormian bones, failure of suture ossification, basilar impression, vertebral abnormalities, kyphoscoliosis, cervical instability, joint laxity, dislocation of the radial head, long-bowed fibulae, pseudoclubbing, short-distal digits, acroosteolysis, and hirsutism.
Gerodermia osteodysplastica
Gerodermia osteodysplastica (OMIM 231070) is characterized by dwarfism, lax skin, osteoporosis, wormian bones, fractures, vertebral compression, wizened facial appearance because of facial skin laxity.
Idiopathic juvenile osteoporosis
Idiopathic juvenile osteoporosis typically presents in preadolescents with fractures and osteoporosis. The fracture susceptibility and osteoporosis usually resolve spontaneously with puberty. The etiology of idiopathic juvenile osteoporosis is unknown.
Dentiogenesis imperfect
DI can occur separately from OI as an isolated familial condition as a result of mutations in the DSPP gene on chromosome 4.
TESTING
Protein, biochemical testing
Historically, biochemical testing was the first line test for confirming a clinical diagnosis of OI. The advent of sequencing technologies has shifted the focus, and it is now possible to perform collagen and noncollagen gene sequencing as a first line approach, only investigating the biochemical properties of the collagens should this testing be negative (Fig. 2). Collagen biochemical analysis and mutation analysis have similar sensitivity and specificity, although sequencing has the advantage of being able to confirm defects in all four genes implicated in causing OI. Collagen analysis may be normal or abnormal in CRTAP or LEPRE mutation patients. Collagen analysis proceeds as follows: fibroblasts derived from a skin biopsy are cultured in vitro to provide a source of protein for electrophoretic analysis of type 1 collagen. The collagens are labeled and assayed on SDS-PAGE to assess their electrophoretic mobility. Abnormal proteins will migrate differently on the gel when compared with control samples. The sensitivity of biochemical testing is 87% in nonlethal forms of OI and about 98% in the lethal form. The sensitivity varies with clinical phenotype and reflects the small effect of some COL1A1 or COL1A2 mutations on type I collagen structure and quantity. A screening test that may be both specific and sensitive for identifying nondeforming OI is the ratio of alkaline phosphatase to phospho inositol phosphate, but this is not in widespread use.42
Gene sequence analysis
Sequence analysis of COL1A1 and COL1A2 cDNA to detect mutations in the coding sequence and sequence analysis of COL1A1 and COL1A2 genomic DNA to detect mutations that alter either sequence or stability of mRNA identify between 80 and 85% of mutations in the type I collagen genes. The mutations in most families are unique; only a few recurrent mutations (mostly CpG dinucleotides) are seen in more than one family. Mutation detection rate varies by COLA1-related OI type, a comprehensive database of all 850 known mutations has been published along with a genotype-phenotype correlation.
For patients in whom no collagen type I mutation is identified, sequencing of CRTAP and LEPRE1 is available commercially and is a reasonable next step in sequential sequencing to identify a mutation. The importance of identifying these mutations is to establish a genetic risk for future offspring as the parents of an affected individual would be at a 25% risk of conceiving another child with the disorder and it would be imperative for them to be counseled accurately, and adequately, of antenatal services available to them.
Genomic DNA can be isolated from peripheral leukocytes in blood or from any other tissue sample from the individual being tested.
Molecular mechanisms
Genotype-phenotype
Structural studies analyzing the effects of glycine substitutions with bulkier, more hydrophilic amino acid side chains on collagen triple helix assembly have shown progressive disruption of the triple helix and molecular assembly.38,39 This is in keeping with predicted theory that the greater the disruption of the triple helix assembly, the more severe the phenotype. A recent report highlights that the lethal mutations occur in eight clusters, which are regularly spaced along two thirds of the chain.40 The severity of the mutations within these clusters are independent of the actual amino acid substitution as they result in exon skipping and thus rely more on the specific position. The “regional model” of lethal mutations has been proposed to identify such mutations and theoretical assignment of phenotype is accurate 86% of the time. These domains seem to be important sites for interaction between proteoglycans and the collagen microfibril.
Mutations affecting the disulfide bonds, which affect C-peptide stability have been shown to result in the milder OI type IV phenotype. This type of mutation can result in formation of a COL1A1 homotrimer.41
Management/treatment
The mainstay of OI management is orthopedic surgery when needed and rehabilitative physiotherapy. The optimal care environment would be a specialized multidisciplinary clinic. Promoting general physical wellbeing is an important aspect of OI management and must be initiated from a very early age. General fatigue during daily activities is a common complaint and because muscle strength and exercise tolerance is reduced in OI, maintaining physical health is paramount to good general health. A regular exercise program also encourages bone growth and improves joint stability.4
Some attention should be focused on the psychosocial aspects of being diagnosed with a chronic disorder, which interferes with daily activities and general acceptance/tolerance by peers and school authorities.
Bisphosphonate therapy
Bisphosphonates are analogs of pyrophosphate that act by inactivating osteoclasts thereby inhibiting bone resorption.42 They are widely used in postmenopausal women to treat osteoporosis, where they have been shown to increase bone density, decrease bone turnover,43 and reduce fractures.44 Although their method of action does not predict amelioration of collagen abnormalities found in OI, the use of bisphosphonates has been extended to people with OI in hopes that increased bone density may positively impact the clinical course of OI. Bisphosphonates have been and continue to be evaluated in both uncontrolled and controlled trials to assess the extent to which they can increase bone mass and strength, reduce fractures and bone deformity, and improve function in children and adults with OI.
Attempts to use bisphosphonates as a treatment for OI were pioneered by Glorieux et al.45 An open-label trial of cyclical intravenous Pamidronate (a bisphosphonate) was reported by Glorieux et al. and the effects of relatively long-term use in adults by Astrom and Soderhall46 and Zeitlin et al.47 Falk et al.48 replicated the study of Glorieux et al. in children over 22 months of age, but of note did report one child with fracture nonunion after treatment with Pamidronate. Nonunion is known to occur in individuals with OI not receiving Pamidronate and the relevance of this observation is thus not clear. However, caution is warranted such that it is reasonable to temporarily discontinue bisphosphonates for several weeks before and after orthopedic surgery procedures in an effort to reduce the risk of bone nonunion. Recently, Pamidronate has been offered even to very young children with OI, but complications including transient asymptomatic hypocalcemia49 and symptomatic hypocalcemia50 have been noted. Pamidronate use is invasive and inconvenient, typically requiring intravenous infusions every 3 months 4 hours a day for 3 consecutive days and has real and potential complications. Several other bisphosphonates have thus been investigated. Oral bisphosphonates have been used in treatment protocols and a comprehensive analysis of the results of controlled clinical trials of bisphosphonates in OI is discussed later. Zoledronic acid is a very potent bisphosphonate that was studied in a clinical trial for OI, but the results of the trial are not yet available. Zoledronic acid can be given over a short infusion period once every 6–12 months. The long-term consequences of lowering bone turnover in children with OI are unknown, but a severe complication of bisphosphonates, avascular necrosis of the jaw, although noted in patients who do not have OI, has not been reported in children or in OI patients, and its risk seems to be highest in the elderly cancer patient treated with the most potent bisphosphonates.51
One of the authors of this review, R.D.S with coauthors recently completed a Cochrane review of bisphosphonate use for OI.3 The objective was to assess the effectiveness of pharmacologic agents in increasing BMD, reducing fractures, and improving quality of life as measured by decreased bone pain and increased function. Cochrane reviews focus on controlled clinical trials, mainly randomized controlled clinical trials (RCTs). Fourteen potentially relevant studies were identified; eight RCTs were qualified for inclusion, six pediatric and two adult studies.52–59 Bisphosphonates use led to increased BMD in OI in all studies in the Cochrane review, where BMD was systematically measured. Effects of bisphosphonates on fracture incidence were inconsistent between studies. Functional outcome and quality of life were inadequately and inconsistently assessed. Even though a few studies did attempt to look at the important parameters, none of the studies were long term. There was insufficient evidence to compare efficacy of oral versus intravenous bisphosphonates.
Interestingly, multiple studies reported the largest gains in BMD in the first year of therapy52–55 and gains in BMD were independent of administration of therapy in a placebo-controlled crossover trial.58 These data possibly argue for short-course bisphosphonate therapy, but further evaluation is certainly indicated.
Bisphosphonate therapy administered for 1–3 years seems to be safe and generally well tolerated. Adverse effects of bisphosphonates are few and minor in this population (gastrointestinal complaints, fever, headache, small decreases in lymphocyte counts).54,59,60 Flu-like symptoms described as “acute phase reactions” are common with IV administration of bisphosphonates with the first infusion.54,59 The results presented in the Cochrane review provide evidence, albeit in a relatively small population, of significant improvement in BMD in individuals with OI when treated with either oral or IV bisphosphonates. However, BMD is a surrogate marker, not a clinical outcome measure. It is still unclear whether this increase in BMD results in lower fracture rates. There are methodological problems with estimates of fracture frequency in studies of bisphosphonates published to date in OI such as inadequate sample size and different methods of estimating fractures before versus during bisphosphonate use (i.e., retrospective recall vs. active surveillance). Effects of bisphosphonates on growth, bone deformity, pain, mobility, and quality of life have not yet been adequately studied.61–63 The long-term safety of bisphosphonates in OI, particularly when used in children, have not been sufficiently evaluated. There was no clear correlation with poor fracture healing but some experts recommend that bisphosphonates not be used within a 6-week period after surgery (same before surgery if elective) because of potential effects on perioperative bone healing. The optimal method, dose, initiation, and duration of use of bisphosphonates in OI remain unclear and require further investigation.
Another therapeutic modality being investigated is the safety and efficacy of cyclical parathyroid hormone as an adjunct treatment for adults with OI. Current medications in this class are unsafe for use in children.
Future considerations for treatment include the bone morphogenic protein modulators targeting BMP2 and BMP7, RANKL inhibitors and the development of stem-cell based therapies.
Screening
-
1
Hearing should be tested by formal audiology evaluation and revisited periodically to assess severity. Stapes surgery has been shown to be of benefit in several series and cochlear implants have shown promise as outcomes are similar to results obtained with other causes of SNHL.
-
2
Imaging for BI: There is a high frequency of BI in patients with severe OI, which is generally slowly progressive in childhood. Patients who present early with minor symptoms have good long-term outcomes with ventral decompression surgery. It is recommended that children with symptoms be referred for neurosurgical evaluation and CT and MRI imaging. Janus et al.64 reported that periodic MRI scans did not influence the clinical decision-making process.
-
3
Dental: early dental assessment is important and involvement of an orthodontist is essential for the dental health of individuals presenting with DI. Reconstruction of teeth with a composite veneer has been shown to improve both esthetics and function in young adults with DI. Dental evaluation should always be performed before bisphosphonates are prescribed to children who have dentition.
Summary
Disease characteristics
OI is a disorder of bone mineralization secondary to abnormal COL1A1 or COL1A2 protein microfibril assembly. The clinical sequelae of this anomaly is brittle bones, which predispose affected individuals to recurrent fractures. The clinical severity ranges from a severe perinatal lethal form to a mild adult variant. The clinical severity correlates with the region of the collagen protein affected and the type of mutation that occurs at these regional nodes. Recently, mutations in two additional genes have been shown to cause some cases of OI, CRTAP, and LEPRE.
Diagnosis/testing
OI is primarily diagnosed on clinical and radiologic findings. The diagnosis may be confirmed by biochemical and/or molecular genetic testing in some instances.
Management
Orthopedic management is the mainstay of interventional therapies. Physiotherapy and regular exercise are essential for proactive health care and general well being. Pharmacotherapy with bisphosphonates may reduce the overall morbidity associated with this disorder and improve quality of life and function in affected individuals, but this has not been well documented in controlled clinical trials.
Counseling
OI is predominantly inherited as an autosomal dominant disorder. The risk to an affected individual of having children with the same disorder is thus 50%. Although the majority of infants with the severe type II OI represent new de novo mutations, a minority result from germline mozaicism and the recurrence risk for parents is thus approximately higher than would otherwise be suspected (approximately 6%). Autosomal recessive forms of OI have been documented, in association with mutations in CRTAP and LEPRE1. Parents of affected children would carry a 25% recurrence risk for future offspring.
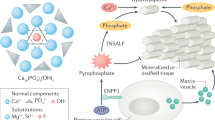
Pathogenic changes resulting in osteogenesis imperfecta can occur at any point of collagen synthesis. Beginning with transcription, followed by translation and subsequent posttranslational modification and transport. This figure outlines the basic processes involved in the molecular synthesis of collagen. 1, transcription (most common cause of OI is DNA mutation); 2, translation; 3, CRTAP-P3H1-CycB (CRTAP and P3H1 have recently been identified as causative mechanisms in OI. CRTAP, P3H1, and Cyclophylin B form a complex, which is responsible for the 3-OH of specific COL1 residues) other: prolyl-4-OH; 4, gal transferase/glc transferase; 5, assembly of procollagen chains; 6, disulfide bonds at N terminus; 7, triple helix assembly; 8, protein suicide (after spontaneous triple helix formation, the newly formed microfibril could undergo protein suicide and result in a milder clinical form of OI or the aberrant protein could be included in collagen fibril assembly, where the dominant negative effect results in more severe disease phenotype); 9, secretion; 10, N and C proteinase cleavage of propeptide; 11, collagen fibril assembly.
References
Makareeva E, Mertz EL, Kuznetsova NV, et al. Structural heterogeneity of type I collagen triple helix and its role in osteogenesis imperfecta. J Biol Chem 2008; 283: 4787–4798.
Martin E, Shapiro JR . Osteogenesis impcrfccta:cpidcmiology and pathophysiology. Curr Osteoporos Rep 2007; 5: 91–97.
Phillipi CA, Remmington T, Steiner RD . Bisphosphonate therapy for osteogenesis imperfecta. Cochrane Database Syst Rev 2008; 4: CD005088.
Cheung MS, Glorieux FH . Osteogenesis imperfecta: update on presentation and management. Rev Endocr Metab Disord 2008; 9: 153–156.
Sillence DO, Rimoin DL . Classification of osteogenesis imperfect. Lancet 1978; 1: 1041–1042.
Aarabi M, Rauch F, Hamdy RC, Fassier F . High prevalence of coxa vara in patients with severe osteogenesis imperfecta. J Pediatr Orthop 2006; 26: 24–28.
Fan Z, Smith PA, Harris GF, Rauch F, Bajorunaite R . Comparison of nanoindentation measurements between osteogenesis imperfecta type III and type IV and between different anatomic locations (femur/tibia versus iliac crest). Connect Tissue Res 2007; 48: 70–75.
Ibrahim AG, Crockard HA . Basilar impression and osteogenesis imperfecta: a 21-year retrospective review of outcomes. J Neurosurg Spine 2007; 7: 594–600.
Fassier AM, Rauch F, Aarabi M, Janelle C, Fassier F . Radial head dislocation and subluxation in osteogenesis imperfecta. J Bone Joint Surg Am 2007; 89: 2694–2704.
Lee DY, Cho TJ, Choi IH, et al. Clinical and radiological manifestations of osteogenesis imperfecta type V. J Korean Med Sci 2006; 21: 709–714.
Marini JC, Cabral WA, Barnes AM, Chang W . Components of the collagen prolyl 3-hydroxylation complex are crucial for normal bone development. Cell Cycle 2007; 6: 1675–1681.
Cabral WA, Chang W, Barnes AM, et al. Prolyl 3-hydroxylase 1 deficiency causes a recessive metabolic bone disorder resembling lethal/severe osteogenesis imperfecta. Nat Genet 2007; 39: 359–365.
Barnes AM, Chang W, Morello 11, et al. Deficiency of cartilage-associated protein in recessive lethal osteogenesis imperfecta. N Engl J Med 2006; 355: 2757–2764.
Morello R, Bertin TK, Chen Y, et al. CRTAP is required for prolyl 3- hydroxylation and mutations cause recessive osteogenesis imperfecta. Cell 2006; 127: 291–304.
Cabral WA, Chang W, Barnes AM, et al. Erratum: prolyl 3-hydroxylase 1 deficiency causes a recessive metabolic bone disorder resembling lethal/severe osteogenesis imperfecta. Nat Genet 2008; 40: 927.
Baldridge D, Schwarze U, Morello R, et al. CRTAP and LEPRE1 mutations in recessive osteogenesis imperfecta. Hum Mutat 2008; 29: 1435–1442.
Malfait F, Symoens S, Coucke P, Nunes L, Be Almeida S, De Paepe A . Total absence of the alpha2(I) chain of collagen type I causes a rare form of Ehlers-Danlos syndrome with hypermobility and propensity to cardiac valvular problems. J Med Genet 2006; 43: e36.
Maifait F, Symoens S, Be Backer J, et al. Three arginine to cysteine substitutions in the pro-alpha (1)-collagen chain cause Ehlers-Danlos syndrome with a propensity to arterial rupture in early adulthood. Hum Mutat 2007; 28: 387–395.
Cabral WA, Makareeva E, Letocha AD, et al. Y-position cysteine substitution in type I collagen (alphal(I) R888C/p.R1066C) is associated with osteogenesis imperfecta/Ehlers-Danlos syndrome phenotype. Hum Mutat 2007; 28: 396–405.
Makareeva E, Cabral WA, Marini JC, Leikin S . Molecular mechanism of alpha 1(I)-osteogenesis imperfecta/Ehlers-Danlos syndrome: unfolding of an N-anchor domain at the N-terminal end of the type I collagen triple helix. J Biol Chem 2006; 281: 6463–6470.
De Coster PS, Cornelissen M, De Paepe A, Martens LC, Vral A . Abnormal dentin structure in two novel gene mutations [COL1A1, Arg134Cys] and [ADAMTS2, Trp795-to-ter] causing rare type I collagen disorders. Arch Oral Biol 2007; 52: 101–109.
Doi K, Nishimura H, Ohta Y, Kubo T . Stapes surgery in Japanese patients with osteogenesis imperfecta. EA Adv Otorhinolaryngol 2007; 65: 226–230.
Van der Rijt A, Cremers C . Stapes surgery in osteogenesis imperfecta: results of a new series. Otol Neurotol 2003; 24: 717–722.
Kuurila K, Pynnonen S, Grenman R . Stapes surgery in osteogenesis imperfecta in Finland. Ann Otol Rhinol Laryngol 2004; 113: 187–193.
Stankovic KM, Kristiansen AG, Bizaki A, Lister M, Adams SC, McKenna MS . Studies of otic capsule morphology and gene expression in the Movl3 mouse—an animal model of type I osteogenesis imperfecta. Audiol Neurootol 2007; 12: 334–343.
Rotteveel U, Beynon AS, Mens LH, Snik AF, Mulder JJ, Mylanus EA . Cochlear Implantation in 3 Patients with Osteogenesis imperfecta: imaging. Surgery and Programming Issues Audiol Neurootol 2007; 13: 73–85.
Pirouzian A, O'Halloran H, Scher C, Jockin Y, Yaghmai R . Traumatic and spontaneous scleral rupture and uveal prolapse in osteogenesis imperfecta. J Pediatr Ophthalmol Strabismus 2007; 44: 315–317.
Zack P, Zack Lit, Surtees R, Neville BG . A standardized tool to measure and describe scleral colour in osteogenesis imperfecta. Ophthalmic Physiol Opt 2007; 27: 174–178.
Ivo R, Fuerderer S, Eysel P . Spondylolisthesis caused by extreme pedicle elongation in osteogenesis imperfecta. Eur Spine J 2007; 16: 1636–1640.
Cubert R, Cheng EY, Mack S, Pepin MG, Byers PH . Osteogenesis imperfecta: mode of delivery and neonatal outcome. Obstet Gynecol 2001; 97: 66–69.
Bird HA . Joint hypermobility. Musculoskeletal Care 2007; 5: 4–19.
McAllion S, Paterson C . Musculo-skeletal problems associated with pregnancy in women with osteogenesis imperfecta. J Obst Gyn 2002; 22: 169–172.
Chan B, Zacharin M . Maternal and infant outcome after pamidronate treatment of polyostotic fibrous dysplasia and osteogenesis imperfecta before conception: a report of four cases. J Clin Endocrinol Metab 2006; 91: 2017–2020.
Huang RP, Ambrose CG, Sullivan E, Haynes RJ . Functional significance of bone density measurements in children with osteogenesis imperfecta. J Bone Joint Surg Am 2006; 88: 1324–1330.
Bachrach LK . Consensus and controversy regarding osteoporosis in the pediatric population. Endocr Pract 2007; 13: 513–520.
Steiner RD, Pepin M, Byers PH . Studies of collagen synthesis and structure in the differentiation of child abuse from osteogenesis imperfecta. J Pediatr 1996; 128: 542–547.
Cundy T, Home A, Bolland M, Gamble G, Davidson J . Bone formation markers in adults with mild osteogenesis imperfecta. Clin Chem 2007; 53: 1109–1114.
Xu P, Huang J, Cebe P, Kaplan DL . Osteogenesis imperfecta collagen-like peptides: self-assembly and mineralization on surfaces. Biomacromolecules 2008; 9: 1551–1557.
Bodian DL, Madhan B, Brodsky B, Klein TE . Predicting the clinical lethality of osteogenesis imperfecta from collagen glycine mutations. Biochemistry 2008; 47: 5424–5432.
Marini JC, Forlino A, Cabral WA, et al. Consortium for osteogenesis imperfecta mutations in the helical domain of type I collagen: regions rich in lethal mutations align with collagen binding sites for integrins and proteoglycans. Hum Mutat 2007; 28: 209–221.
Pace JM, Wiese M, Drenguis AS, et al. Defective C-propeptides of the proalpha2(I) chain of type I procollagen impede molecular assembly and result in osteogenesis imperfecta. J Biol Chem 2008; 283: 16061–16067.
Fisher JE, Rogers MJ, Halasy JM, et al. Alendronate mechanism of action: geranylgeraniol, an intermediate in the mevalonate pathway, prevents inhibition of osteoclast formation, bone resorption, and kinase activation in vitro. Proc Natl Acad Sci USA 1999; 96: 133–138.
Reid IR, Brown JP, Burckhardt P, et al. Intravenous zoledronic acid in postmenopausal women with low bone mineral density. N Engl J Med 2002; 346: 653–661.
Black DM, Cummings SR, Karpf DB, et al. Randomized trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Lancet 1996; 348: 1535–1541.
Glorieux FH, Bishop NJ, Plotkin H, Chabot G, Lanoue G, Travers R . Cyclic administration of pamidronate in children with severe osteogenesis imperfecta. N Engl J Med 1998; 339: 947–952.
Astrom E, Soderhall S . Beneficial effect of long term intravenous bisphosphonate treatment of osteogenesis imperfecta. Arch Dis Child 2002; 86: 356–364.
Zeitlin L, Rauch F, Plotkin H, Glorieux FH . Height and weight development during four years of therapy with cyclical intravenous pamidronate in children and adolescents with osteogenesis imperfecta types I, III, and IV. Pediatrics 2003; 111: 1030–1036.
Falk MJ, Heeger S, Lynch KA, et al. Intravenous bisphosphonate therapy in children with osteogenesis imperfecta. Pediatrics 2003; 111: 573–578.
Plotkin H, Rauch F, Bishop NJ, et al. Pamidronate treatment of severe osteogenesis imperfecta in children under 3 years of age. J Clin Endocrinol Metab 2000; 85: 1846–1850.
Chien YH, Chu SY, Hsu CC, Hwu WL . Pamidronate treatment of severe osteogenesis imperfecta in a newborn infant. J Inherit Metab Dis 2002; 25: 593–595.
Woo S, Hellstein JW, Kalmar JR . Systematic Review: bisphosphonates and Osteonecrosis of the Jaws. Ann Intern Med 2006; 144: 753–761.
Adami S, Gatti D, Colapietro F, et al. Intravenous neridronate in adults with osteogenesis imperfecta. J Bone Miner Res 2003; 18: 126–130.
Gatti D, Antoniazzi F, Prizzi R, et al. Intravenous neridronate in children with osteogenesis imperfecta: a randomized controlled study. J Bone Miner Res 2005; 20: 758–763.
Letocha AD, Cintas HL, Troendle JF, et al. Controlled trial of pamidronate in children with types III and IV osteogenesis imperfecta confirms vertebral gains but not short-term functional improvement. J Bone Miner Res 2005; 20: 977–986.
Chevrel G, Schott AM, Fontanges E, et al. Effects of oral alendronate on BMD in adult patients with osteogenesis imperfecta: a 3-year randomized placebo-controlled trial. J Bone Miner Res 2006; 21: 300–306.
Glorieux FH, Rauch F, Ward LM, et al. Alendronate in the treatment of pediatric osteogenesis imperfecta. J Bone Miner Res 2004; 20 ( suppl): 1043.
Sakkers R, Kok D, Engelbert R, et al. Skeletal effects and functional outcome with olpadronate in children with osteogenesis imperfecta: a 2-year randomised placebo-controlled study. Lancet 2004; 363: 1427–1431.
Seikaly MG, Kopanati S, Salhab N, et al. Impact of alendronate on quality of life in children with osteogenesis imperfecta. J Pediatr Orthopaed 2005; 25: 786–791.
DiMeglio LA, Peacock M . Two-year clinical trial of oral alendronate versus intravenous pamidronate in children with osteogenesis imperfecta. J Bone Miner Res 2006; 21: 132–140.
Ward LM, Denker AE, Porras A, et al. Single-dose pharmacokinetics and tolerability of alendronate 35- and 70-milligram tablets in children and adolescents with osteogenesis imperfecta type I. J Clin Endocrinol Metabol 2005; 90: 4051–4056.
Huber MA . Osteogenesis imperfecta. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2007; 103: 314–320.
Malmgren B, Aström E, Söderhäll S . No osteonecrosis in jaws of young patients with osteogenesis imperfecta treated with bisphosphonates. J Oral Pathol Med 2008; 37: 196–200.
Schwartz S, Joseph C, Iera D, Vu DD . Bisphosphonates, osteonecrosis, osteogenesis imperfecta and dental extractions: a case series. J Can Dent Assoc 2008; 74: 537–542.
Janus GJ, Engelbert RH, Beek E, Gooskens RH, Pruijs JE . Osteogenesis imperfecta in childhood: MR imaging of basilar impression. Eur J Radiol 2003; 47: 19–24.
Acknowledgements
The authors acknowledge the important contributions of Melanie Pepin and Peter Byers to this manuscript by way of their coauthorship with Robert Steiner of the OI GeneReview chapter on which this manuscript was based.
Author information
Authors and Affiliations
Corresponding author
Additional information
Disclosure: The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Basel, D., Steiner, R. Osteogenesis imperfecta: Recent findings shed new light on this once well-understood condition. Genet Med 11, 375–385 (2009). https://doi.org/10.1097/GIM.0b013e3181a1ff7b
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1097/GIM.0b013e3181a1ff7b
Keywords
This article is cited by
-
Mutation spectrum of COL1A1/COL1A2 screening by high-resolution melting analysis of Chinese patients with osteogenesis imperfecta
Journal of Bone and Mineral Metabolism (2020)
-
Assessing disease experience across the life span for individuals with osteogenesis imperfecta: challenges and opportunities for patient-reported outcomes (PROs) measurement: a pilot study
Orphanet Journal of Rare Diseases (2019)
-
Incorporating the patient perspective in the study of rare bone disease: insights from the osteogenesis imperfecta community
Osteoporosis International (2019)
-
Fatigue and disturbances of sleep in patients with osteogenesis imperfecta – a cross-sectional questionnaire study
BMC Musculoskeletal Disorders (2018)
-
The clinical features of osteogenesis imperfecta in Vietnam
International Orthopaedics (2017)