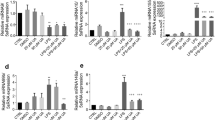
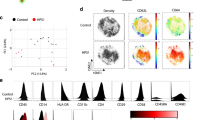
Abstract
Recent clinical trials in patients with inflammatory diseases like multiple sclerosis (MS) or inflammatory bowel disease (IBD) have shown the beneficial effects of probiotic helminth administration, although the underlying mechanism of action remains largely unknown. Potential cellular targets may include innate immune cells that propagate inflammation in these diseases, like pro-inflammatory macrophages. We here investigated the effects of the helminth Trichuris suis soluble products (SPs) on the phenotype and function of human inflammatory (granulocyte-macrophage colony-stimulating factor (GM-CSF)-differentiated) macrophages. Interestingly, we here show that T. suis SPs potently skew inflammatory macrophages into a more anti-inflammatory state in a Toll-like receptor 4 (TLR4)-dependent manner, and less effects are seen when stimulating macrophages with TLR2 or -3 ligands. Gene microarray analysis of GM-CSF-differentiated macrophages further revealed that many TLR4-induced inflammatory mediators, including interleukin (IL)-12B, CCL1 and CXCL9, are downregulated by T. suis SPs. In particular, we observed a strong reduction in the expression and function of P2RX7, a purinergic receptor involved in macrophage inflammation, leading to reduced IL-1β secretion. In conclusion, we show that T. suis SPs suppress a broad range of inflammatory pathways in GM-CSF-differentiated macrophages in a TLR4-dependent manner, thereby providing enhanced mechanistic insight into the therapeutic potential of this helminth for patients with inflammatory diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Bach JF . The effect of infections on susceptibility to autoimmune and allergic diseases. N Engl J Med 2002; 347: 911–920.
Zaccone P, Cooke A . Vaccine against autoimmune disease: can helminths or their products provide a therapy? Curr Opin Immunol 2013; 25: 418–423.
Strachan DP . Hay fever, hygiene, and household size. BMJ 1989; 299: 1259–1260.
Weinstock JV, Elliott DE . Helminths and the IBD hygiene hypothesis. Inflamm Bowel Dis 2009; 15: 128–133.
Fleming JO, Cook TD . Multiple sclerosis and the hygiene hypothesis. Neurology 2006; 67: 2085–2086.
Bager P, Arnved J, Ronborg S, Wohlfahrt J, Poulsen LK, Westergaard T et al. Trichuris suis ova therapy for allergic rhinitis: a randomized, double-blind, placebo-controlled clinical trial. J Allergy Clin Immunol 2010; 125: 123–30 e1-3.
Feary JR, Venn AJ, Mortimer K, Brown AP, Hooi D, Falcone FH et al. Experimental hookworm infection: a randomized placebo-controlled trial in asthma. Clin Exp Allergy 2010; 40: 299–306.
Correale J, Farez M . Association between parasite infection and immune responses in multiple sclerosis. Ann Neurol 2007; 61: 97–108.
Correale J, Farez MF . The impact of parasite infections on the course of multiple sclerosis. J Neuroimmunol 2011; 233: 6–11.
Fleming JO, Isaak A, Lee JE, Luzzio CC, Carrithers MD, Cook TD et al. Probiotic helminth administration in relapsing-remitting multiple sclerosis: a phase 1 study. Mult Scler 2011; 17: 743–754.
Kuijk LM, Klaver EJ, Kooij G, van der Pol SM, Heijnen P, Bruijns SC et al. Soluble helminth products suppress clinical signs in murine experimental autoimmune encephalomyelitis and differentially modulate human dendritic cell activation. Mol Immunol 2012; 51: 210–218.
Murray PJ, Wynn TA . Protective and pathogenic functions of macrophage subsets. Nat Rev 2011; 11: 723–737.
Mosser DM, Edwards JP . Exploring the full spectrum of macrophage activation. Nat Rev 2008; 8: 958–969.
Elliott DE, Weinstock JV . Helminth-host immunological interactions: prevention and control of immune-mediated diseases. Ann N Y Acad Sci 2012; 1247: 83–96.
Vogel DY, Vereyken EJ, Glim JE, Heijnen PD, Moeton M, van der Valk P et al. Macrophages in inflammatory multiple sclerosis lesions have an intermediate activation status. J Neuroinflammation 2013; 10: 35.
Klaver EJ, Kuijk LM, Laan LC, Kringel H, van Vliet SJ, Bouma G et al. Trichuris suis-induced modulation of human dendritic cell function is glycan-mediated. Int J Parasitol 2013; 43: 191–200.
Verreck FA, de Boer T, Langenberg DM, van der Zanden L, Ottenhoff TH . Phenotypic and functional profiling of human proinflammatory type-1 and anti-inflammatory type-2 macrophages in response to microbial antigens and IFN-gamma- and CD40L-mediated costimulation. J Leukoc Biol 2006; 79: 285–293.
Codarri L, Gyulveszi G, Tosevski V, Hesske L, Fontana A, Magnenat L et al. RORgammat drives production of the cytokine GM-CSF in helper T cells, which is essential for the effector phase of autoimmune neuroinflammation. Nat Immunol 2011; 12: 560–567.
McQualter JL, Darwiche R, Ewing C, Onuki M, Kay TW, Hamilton JA et al. Granulocyte macrophage colony-stimulating factor: a new putative therapeutic target in multiple sclerosis. J Exp Med 2001; 194: 873–882.
Schlee M . Master sensors of pathogenic RNA - RIG-I like receptors. Immunobiology 2013; 218: 1322–1335.
Gouwy M, Struyf S, Leutenez L, Portner N, Sozzani S, Van Damme J . Chemokines and other GPCR ligands synergize in receptor-mediated migration of monocyte-derived immature and mature dendritic cells. Immunobiology 2014; 219: 218–229.
Donnelly-Roberts DL, Jarvis MF . Discovery of P2 × 7 receptor-selective antagonists offers new insights into P2 × 7 receptor function and indicates a role in chronic pain states. Br J Pharmacol 2007; 151: 571–579.
Hiemstra IH, Klaver EJ, Vrijland K, Kringel H, Andreasen A, Bouma G et al. Excreted/secreted Trichuris suis products reduce barrier function and suppress inflammatory cytokine production of intestinal epithelial cells. Mol Immunol 2014; 60: 1–7.
Fava F, Danese S . Intestinal microbiota in inflammatory bowel disease: friend of foe? World J Gastroenterol 2011; 17: 557–566.
Diaz A, Allen JE . Mapping immune response profiles: the emerging scenario from helminth immunology. Eur J Immunol 2007; 37: 3319–3326.
Ambarus CA, Krausz S, van Eijk M, Hamann J, Radstake TR, Reedquist KA et al. Systematic validation of specific phenotypic markers for in vitro polarized human macrophages. J Immunol Methods 2012; 375: 196–206.
Fassbender K, Ragoschke A, Rossol S, Schwartz A, Mielke O, Paulig A et al. Increased release of interleukin-12p40 in MS: association with intracerebral inflammation. Neurology 1998; 51: 753–758.
Longbrake EE, Racke MK . Why did IL-12/IL-23 antibody therapy fail in multiple sclerosis? Expert Rev Neurother 2009; 9: 319–321.
Shin T, Kang B, Tanuma N, Matsumoto Y, Wie M, Ahn M et al. Intrathecal administration of endothelin-1 receptor antagonist ameliorates autoimmune encephalomyelitis in Lewis rats. NeuroReport 2001; 12: 1465–1468.
Cunningham ME, Huribal M, Bala RJ, McMillen MA . Endothelin-1 and endothelin-4 stimulate monocyte production of cytokines. Crit Care Med 1997; 25: 958–964.
Majetschak M . Extracellular ubiquitin: immune modulator and endogenous opponent of damage-associated molecular pattern molecules. J Leukoc Biol 2011; 89: 205–219.
Cesaro A, Brest P, Hofman V, Hebuterne X, Wildman S, Ferrua B et al. Amplification loop of the inflammatory process is induced by P2 × 7R activation in intestinal epithelial cells in response to neutrophil transepithelial migration. Am J Physiol 2010; 299: G32–G42.
Yiangou Y, Facer P, Durrenberger P, Chessell IP, Naylor A, Bountra C et al. COX-2, CB2 and P2 × 7-immunoreactivities are increased in activated microglial cells/macrophages of multiple sclerosis and amyotrophic lateral sclerosis spinal cord. BMC Neurol 2006; 6: 12.
Dujmovic I, Mangano K, Pekmezovic T, Quattrocchi C, Mesaros S, Stojsavljevic N et al. The analysis of IL-1 beta and its naturally occurring inhibitors in multiple sclerosis: The elevation of IL-1 receptor antagonist and IL-1 receptor type II after steroid therapy. J Neuroimmunol 2009; 207: 101–106.
Narcisse L, Scemes E, Zhao Y, Lee SC, Brosnan CF . The cytokine IL-1beta transiently enhances P2 × 7 receptor expression and function in human astrocytes. Glia 2005; 49: 245–258.
Ambrosini E, Remoli ME, Giacomini E, Rosicarelli B, Serafini B, Lande R et al. Astrocytes produce dendritic cell-attracting chemokines in vitro and in multiple sclerosis lesions. J Neuropathol Exp Neurol 2005; 64: 706–715.
Wagner DR, Kubota T, Sanders VJ, McTiernan CF, Feldman AM . Differential regulation of cardiac expression of IL-6 and TNF-alpha by A2- and A3-adenosine receptors. Am J Physiol 1999; 276 (6 Pt 2): H2141–H2147.
Zhang J, Tachado SD, Patel N, Zhu J, Imrich A, Manfruelli P et al. Negative regulatory role of mannose receptors on human alveolar macrophage proinflammatory cytokine release in vitro. J Leukoc Biol 2005; 78: 665–674.
Fleming JO . Helminth therapy and multiple sclerosis. Int J Parasitol 2013; 43: 259–274.
Fabriek BO, Van Haastert ES, Galea I, Polfliet MM, Dopp ED, Van Den Heuvel MM et al. CD163-positive perivascular macrophages in the human CNS express molecules for antigen recognition and presentation. Glia 2005; 51: 297–305.
Schirmer SH, Fledderus JO, van der Laan AM, van der Pouw-Kraan TC, Moerland PD, Volger OL et al. Suppression of inflammatory signaling in monocytes from patients with coronary artery disease. J Mol Cell Cardiol 2009; 46: 177–185.
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA 2005; 102: 15545–15550.
Kooij G, Mizee MR, van Horssen J, Reijerkerk A, Witte ME, Drexhage JA et al. Adenosine triphosphate-binding cassette transporters mediate chemokine (C-C motif) ligand 2 secretion from reactive astrocytes: relevance to multiple sclerosis pathogenesis. Brain 2011; 134 (Pt 2): 555–570.
Acknowledgements
This work was supported by grants from the National Multiple Sclerosis Society (PP1686) and the Dutch MS Research Foundation (MS 11-771).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Genes and Immunity website
Supplementary information
Rights and permissions
About this article
Cite this article
Ottow, M., Klaver, E., van der Pouw Kraan, T. et al. The helminth Trichuris suis suppresses TLR4-induced inflammatory responses in human macrophages. Genes Immun 15, 477–486 (2014). https://doi.org/10.1038/gene.2014.38
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gene.2014.38
This article is cited by
-
Extracellular vesicles secreted by Brugia malayi microfilariae modulate the melanization pathway in the mosquito host
Scientific Reports (2023)
-
Whipworm secretions and their roles in host-parasite interactions
Parasites & Vectors (2022)
-
Analysis of the Trichuris suis excretory/secretory proteins as a function of life cycle stage and their immunomodulatory properties
Scientific Reports (2018)
-
Inhibition of CD40-TRAF6 interactions by the small molecule inhibitor 6877002 reduces neuroinflammation
Journal of Neuroinflammation (2017)
-
Trichuris suis soluble products induce Rab7b expression and limit TLR4 responses in human dendritic cells
Genes & Immunity (2015)