Abstract
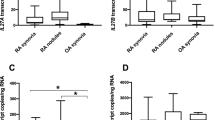
Matrix metalloproteinases (MMPs) contribute to the joint damage in rheumatoid arthritis (RA). Less is known of the involvement of MMPs at extra-articular sites of rheumatoid inflammation. We assessed the relative contribution from MMP-1, MMP-3, MMP-7 and MMP-12 to joint and extra-articular tissue destruction and inflammation by comparing gene expression in joint synovia and subcutaneous rheumatoid nodules from RA patients. Expression of MMP-1 and MMP-3 predominated in synovia, whereas MMP-12 expression was significantly higher in rheumatoid nodules. Markedly higher MMP-7 expression distinguished a subgroup of nodules that featured infiltrating monocyte/macrophage-producing MMP-7 protein. The high MMP-7 expression in nodules was associated with the single-nucleotide polymorphism (SNP) rs11568818 (−181A>G, MMP-7 promoter) and more active inflammation within the nodule lesions. Patients with such nodules had significantly earlier age of RA onset. Our findings indicate that the expression of MMP-1 and MMP-3 occurs relatively independent of the tissue microenvironment with substantial expression also at extra-articular sites. MMP-12 expression reflects the involvement of monocyte/macrophages in rheumatoid inflammation. Evidence for the association between the rs11568818 SNP and increased MMP-7 expression is restricted to nodules, which indicates that consequences of the MMP-7 polymorphism are likely to manifest within aspects of immune/inflammatory activity that are monocyte/macrophage-mediated.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Wolfe F, Michaud K, Gefeller O, Choi HK . Predicting mortality in patients with rheumatoid arthritis. Arthritis Rheum 2003; 48: 1530–1542.
Ziff M . The rheumatoid nodule. Arthritis Rheum 1990; 33: 761–767.
Highton J, Hung N, Hessian P, Wilsher M . Pulmonary rheumatoid nodules demonstrating features usually associated with rheumatoid synovial membrane. Rheumatology 2007; 46: 811–814.
Palmer DG, Hogg N, Allen CA, Highton J, Hessian PA . A mononuclear phagocyte subset associated with cell necrosis in rheumatoid nodules: identification with monoclonal antibody 5.5. Clin Immunol Immunopathol 1987; 45: 17–28.
Highton J, Kean A, Hessian PA, Thomson J, Rietveld J, Hart DN . Cells expressing dendritic cell markers are present in the rheumatoid nodule. J Rheumatol 2000; 27: 339–346.
Duke OL, Hobbs S, Panayi GS, Poulter LW, Rasker JJ, Janossy G . A combined immunohistological and histochemical analysis of lymphocyte and macrophage subpopulations in the rheumatoid nodule. Clin Exp Immunol 1984; 56: 239–246.
Mellbye OJ, Forre O, Mollnes TE, Kvarnes L . Immunopathology of subcutaneous rheumatoid nodules. Ann Rheum Dis 1991; 50: 909–912.
Highton J, Hessian PA, Stamp L . The rheumatoid nodule: peripheral or central to rheumatoid arthritis? Rheumatology 2007; 46: 1385–1387.
Hessian PA, Highton J, Kean A, Sun CK, Chin M . Cytokine profile of the rheumatoid nodule suggests that it is a Th1 granuloma. Arthritis Rheum 2003; 48: 334–338.
Wikaningrum R, Highton J, Parker A, Coleman M, Hessian PA, Roberts-Thompson PJ et al. Pathogenic mechanisms in the rheumatoid nodule: comparison of proinflammatory cytokine production and cell adhesion molecule expression in rheumatoid nodules and synovial membranes from the same patient. Arthritis Rheum 1998; 41: 1783–1797.
Vincenti MP, Clark IM, Brinckerhoff CE . Using inhibitors of metalloproteinases to treat arthritis. Easier said than done? Arthritis Rheum 1994; 37: 1115–1126.
Ahrens D, Koch AE, Pope RM, Stein-Picarella M, Niedbala MJ . Expression of matrix metalloproteinase 9 (96-kd gelatinase B) in human rheumatoid arthritis. Arthritis Rheum 1996; 39: 1576–1587.
Pakozdi A, Amin MA, Haas CS, Martinez RJ, Haines GK, Santos LL et al. Macrophage migration inhibitory factor: a mediator of matrix metalloproteinase-2 production in rheumatoid arthritis. Arthritis Res Ther 2006; 8: R132.
Matsushita I, Uzuki M, Matsuno H, Sugiyama E, Kimura T . Rheumatoid nodulosis during methotrexate therapy in a patient with rheumatoid arthritis. Mod Rheumatol 2006; 16: 401–403.
Crouser ED, Culver DA, Knox KS, Julian MW, Shao G, Abraham S et al. Gene expression profiling identifies MMP-12 and ADAMDEC1 as potential pathogenic mediators of pulmonary sarcoidosis. Am J Respir Crit Care Med 2009; 179: 929–938.
Sheen P, O’Kane CM, Chaudhary K, Tovar M, Santillan C, Sosa J et al. High MMP-9 activity characterises pleural tuberculosis correlating with granuloma formation. Eur Respir J 2009; 33: 134–141.
Vaalamo M, Kariniemi AL, Shapiro SD, Saarialho-Kere U . Enhanced expression of human metalloelastase (MMP-12) in cutaneous granulomas and macrophage migration. J Invest Dermatol 1999; 112: 499–505.
Burke B . The role of matrix metalloproteinase 7 in innate immunity. Immunobiology 2004; 209: 51–56.
Busiek DF, Baragi V, Nehring LC, Parks WC, Welgus HG . Matrilysin expression by human mononuclear phagocytes and its regulation by cytokines and hormones. J Immunol 1995; 154: 6484–6491.
Busiek DF, Ross FP, McDonnell S, Murphy G, Matrisian LM, Welgus HG . The matrix metalloprotease matrilysin (PUMP) is expressed in developing human mononuclear phagocytes. J Biol Chem 1992; 267: 9087–9092.
Halpert I, Sires UI, Roby JD, Potter-Perigo S, Wight TN, Shapiro SD et al. Matrilysin is expressed by lipid-laden macrophages at sites of potential rupture in atherosclerotic lesions and localizes to areas of versican deposition, a proteoglycan substrate for the enzyme. Proc Natl Acad Sci USA 1996; 93: 9748–9753.
Shapiro SD, Kobayashi DK, Ley TJ . Cloning and characterization of a unique elastolytic metalloproteinase produced by human alveolar macrophages. J Biol Chem 1993; 268: 23824–23829.
Shipley JM, Wesselschmidt RL, Kobayashi DK, Ley TJ, Shapiro SD . Metalloelastase is required for macrophage-mediated proteolysis and matrix invasion in mice. Proc Natl Acad Sci USA 1996; 93: 3942–3946.
Sternlicht MD, Werb Z . How matrix metalloproteinases regulate cell behavior. Annu Rev Cell Dev Biol 2001; 17: 463–516.
Parks WC, Wilson CL, Lopez-Boado YS . Matrix metalloproteinases as modulators of inflammation and innate immunity. Nat Rev Immunol 2004; 4: 617–629.
Jormsjo S, Whatling C, Walter DH, Zeiher AM, Hamsten A, Eriksson P . Allele-specific regulation of matrix metalloproteinase-7 promoter activity is associated with coronary artery luminal dimensions among hypercholesterolemic patients. Arterioscler Thromb Vasc Biol 2001; 21: 1834–1839.
Jormsjo S, Ye S, Moritz J, Walter DH, Dimmeler S, Zeiher AM et al. Allele-specific regulation of matrix metalloproteinase-12 gene activity is associated with coronary artery luminal dimensions in diabetic patients with manifest coronary artery disease. Circ Res 2000; 86: 998–1003.
Chen Y, Nixon NB, Dawes PT, Mattey DL . Influence of variations across the MMP-1 and -3 genes on the serum levels of MMP-1 and -3 and disease activity in rheumatoid arthritis. Genes Immun 2012; 13: 29–37.
Li Y, Jin X, Kang S, Wang Y, Du H, Zhang J et al. Polymorphisms in the promoter regions of the matrix metalloproteinases-1, -3, -7, and -9 and the risk of epithelial ovarian cancer in China. Gynecol Oncol 2006; 101: 92–96.
Zhang J, Jin X, Fang S, Wang R, Li Y, Wang N et al. The functional polymorphism in the matrix metalloproteinase-7 promoter increases susceptibility to esophageal squamous cell carcinoma, gastric cardiac adenocarcinoma and non-small cell lung carcinoma. Carcinogenesis 2005; 26: 1748–1753.
Peng B, Cao L, Ma X, Wang W, Wang D, Yu L . Meta-analysis of association between matrix metalloproteinases 2, 7 and 9 promoter polymorphisms and cancer risk. Mutagenesis 2010; 25: 371–379.
Ye S, Patodi N, Walker-Bone K, Reading I, Cooper C, Dennison E . Variation in the matrix metalloproteinase-3, -7, -12 and -13 genes is associated with functional status in rheumatoid arthritis. Int J Immunogenet 2007; 34: 81–85.
Palmer DG, Hogg N, Highton J, Hessian PA, Denholm I . Macrophage migration and maturation within rheumatoid nodules. Arthritis Rheum 1987; 30: 728–736.
Hessian PA, Edgeworth J, Hogg N . MRP-8 and MRP-14, two abundant Ca(2+)-binding proteins of neutrophils and monocytes. J Leukoc Biol 1993; 53: 197–204.
Brinckerhoff CE . Joint destruction in arthritis: metalloproteinases in the spotlight. Arthritis Rheum 1991; 34: 1073–1075.
Mohammed FF, Smookler DS, Khokha R . Metalloproteinases, inflammation, and rheumatoid arthritis. Ann Rheum Dis 2003; 62 (Suppl 2): ii43–ii47.
Mattey DL, Nixon NB, Dawes PT, Ollier WE, Hajeer AH . Association of matrix metalloproteinase 3 promoter genotype with disease outcome in rheumatoid arthritis. Genes Immun 2004; 5: 147–149.
Suzuki K, Enghild JJ, Morodomi T, Salvesen G, Nagase H . Mechanisms of activation of tissue procollagenase by matrix metalloproteinase 3 (stromelysin). Biochemistry 1990; 29: 10261–10270.
Klein T, Bischoff R . Physiology and pathophysiology of matrix metalloproteases. Amino Acids 2010; 41: 271–290.
Ancuta P, Liu KY, Misra V, Wacleche VS, Gosselin A, Zhou X et al. Transcriptional profiling reveals developmental relationship and distinct biological functions of CD16+ and CD16− monocyte subsets. BMC Genomics 2009; 10: 403.
Wang X, Liang J, Koike T, Sun H, Ichikawa T, Kitajima S et al. Overexpression of human matrix metalloproteinase-12 enhances the development of inflammatory arthritis in transgenic rabbits. Am J Pathol 2004; 165: 1375–1383.
Dean RA, Cox JH, Bellac CL, Doucet A, Starr AE, Overall CM . Macrophage-specific metalloelastase (MMP-12) truncates and inactivates ELR+ CXC chemokines and generates CCL2, -7, -8, and -13 antagonists: potential role of the macrophage in terminating polymorphonuclear leukocyte influx. Blood 2008; 112: 3455–3464.
Feldmann M, Maini RN . Lasker Clinical Medical Research Award. TNF defined as a therapeutic target for rheumatoid arthritis and other autoimmune diseases. Nat Med 2003; 9: 1245–1250.
Li Q, Park PW, Wilson CL, Parks WC . Matrilysin shedding of syndecan-1 regulates chemokine mobilization and transepithelial efflux of neutrophils in acute lung injury. Cell 2002; 111: 635–646.
Kelly MM, Leigh R, Gilpin SE, Cheng E, Martin GE, Radford K et al. Cell-specific gene expression in patients with usual interstitial pneumonia. Am J Respir Crit Care Med 2006; 174: 557–565.
Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 1988; 31: 315–324.
Acknowledgements
This work was supported by Arthritis New Zealand and the Health Research Council of New Zealand.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies this paper on Genes and Immunity website
Supplementary information
Rights and permissions
About this article
Cite this article
Kazantseva, M., Hung, N., Highton, J. et al. MMP expression in rheumatoid inflammation: the rs11568818 polymorphism is associated with MMP-7 expression at an extra-articular site. Genes Immun 14, 162–169 (2013). https://doi.org/10.1038/gene.2012.65
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gene.2012.65
Keywords
This article is cited by
-
Natural carboxyterminal truncation of human CXCL10 attenuates glycosaminoglycan binding, CXCR3A signaling and lymphocyte chemotaxis, while retaining angiostatic activity
Cell Communication and Signaling (2024)
-
A review of the pleiotropic actions of the IFN-inducible CXC chemokine receptor 3 ligands in the synovial microenvironment
Cellular and Molecular Life Sciences (2023)
-
Pulsatilla decoction suppresses matrix metalloproteinase-7-mediated leukocyte recruitment in dextran sulfate sodium-induced colitis mouse model
BMC Complementary Medicine and Therapies (2022)
-
IgG anti-hinge antibodies against IgG4 F(ab’)2 fragments generated using pepsin are useful diagnostic markers for rheumatoid arthritis: implications of the possible roles of metalloproteinases and IgG subclasses in generating immunogenic hinge epitopes
Arthritis Research & Therapy (2020)
-
Gait speed and handgrip strength as predictors of all-cause mortality and cardiovascular events in hemodialysis patients
BMC Nephrology (2020)