Abstract
Purpose
To identify causes of symptomatic band keratopathy, and assess the results and long-term recurrence rates following chelation with topical ethylene-diamine-tetra-acetic acid (EDTA).
Patients and methods
A retrospective review of surgical logbooks identified patients managed by EDTA chelation for symptomatic band keratopathy from 2009 to 2015.
Results
We identified 108 cases; 89 case notes were available for analysis. Most cases of band keratopathy were idiopathic (36%). The most commonly identified underlying diagnosis was long-term topical glaucoma therapy (27%). Median presenting visual acuity was 6/18 (range 6/6-NPL) with the visual axis affected in 97.8% of cases. Treatment involved corneal epithelium removal, recurrent application of topical EDTA, and subsequent debridement. The mean duration of the operation was 20 min (range 10–45). Mean initial follow-up time was 40 days, and the visual axis was clear in 97.8%. Visual acuity was maintained or improved in 79.8%, with 13.5% improving by two lines or more. The mean length of follow-up was 581 days (median 374, maximum 2438). Twenty-five eyes (28.1%) showed localised recurrence of calcium with a mean time of 546 days (median 374), but only four cases required repeat EDTA chelation. The median time between operations was 430 days. Thirty-two per cent of the recurrence cases were associated with hypotony or chronic presence of silicone oil.
Conclusions
Chelation of calcium with topical EDTA is a safe and effective treatment for band keratopathy. Visual acuity improves in most eyes and while the rate of recurrence is moderate, the need for retreatment is low (4.5% overall).
Similar content being viewed by others
Introduction
Band keratopathy was first described by Dixon in 1848 as a degenerative condition of the cornea, characterised by deposition of grey-white opacities in the superficial corneal layers, most commonly in the exposed inter-palpebral region.1, 2 It can be classified as primary or secondary, with a variety of predisposing systemic and ocular conditions, including renal failure, elevated serum calcium levels, chronic ocular inflammation, corneal ulcers, alkali chemical burns (and phosphate-based irrigating solutions), and a compromised ocular surface due to adverse reactions to preservatives/buffers in ocular drops, particularly in patients on life-long therapy.1, 2, 3, 4, 5 Familial band keratopathy has also been reported.6 The natural history of band keratopathy is gradual progression over many months/years; however, there have been isolated reports of acute incidence following intra-cameral injection of tissue plasminogen activator to treat post-operative fibrinous reaction.7, 8
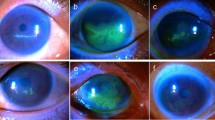
In most cases band keratopathy is superficial. Histological analysis of affected corneas have revealed a selective, fine-granular calcification of Bowman’s membrane, while other studies have reported deep-situated calcium plaques also involving the anterior stroma.1, 2 In the early stages of the condition, the patients remain asymptomatic; however, extension into the visual axis can result in glare and reduced visual acuity. Furthermore, a breakdown of the ocular surface can result in painful recurrent corneal erosion-like symptoms and predispose to corneal ulceration. It is our recommendation that band keratopathy should only be removed when visually significant or causing ocular surface symptoms. (See Figure 1 for a range of clinical presentations).
Causes of band keratopathy. Clinical picture demonstrating range of clinical presentations of band keratopathy. (a) Peripheral, not visually significant; (b) Central, mild visual symptoms; (c): Dense central band, visually significant; (d) Prominent in a blind eye causing ocular surface discomfort/pain.
Previous generations used mechanical debridement to remove superficial calcium deposition for the cornea.9 The most popular current method involves the use of a metal ion chelator known as ethylene-diamine-tetra-acetic acid (EDTA). Studies have reported EDTA chelation to be a safe, reliable method of removing band keratopathy, resulting in improvements in visual acuity and ocular comfort.1, 2 This procedure can be used in isolation or supplemented with superficial keratectomy (manual or excimer laser PTK) or amniotic membrane transplant.10, 11, 12, 13, 14 A technique using alcohol to elevate and preserve the epithelium, use of EDTA and then replacement of the epithelial sheet similar to LASEK to provide better analgesia has also been described.15
We wished to review the use of EDTA chelation in our department, assess the results and long-term recurrence rates following treatment, and identify the causes of symptomatic band keratopathy in our population.
Methods
We conducted a retrospective case note review of patients who underwent ‘EDTA chelation’ in a university hospital setting in Glasgow, UK, between 2009 and 2015. Inclusion criteria comprised any patients undergoing EDTA chelation to treat band keratopathy over the age of 18 years. Exclusion criteria included manual removal of calcium without EDTA, or use of adjuvant therapy such as amniotic membrane or conjunctival grafts.
Patient demographics included patient’s age, laterality, diagnosis, presenting visual acuity (initial VA) and indication for EDTA chelation. The clinic visit closest to the 4 weeks post-operative review was evaluated for VA and clearance of visual axis. Long-term measures such as time to recurrence and subsequent retreatment were also investigated.
The improvement or worsening of VA was noted at 4-week follow-up and a change of two or more lines of Snellen VA was considered clinically significant. Visual acuity was not considered to have changed significantly if it remained within one line of initial VA. Statistical analysis involved use of a Wilcoxon signed rank test to assess change in VA and a Mann–Whitney test to evaluate if presenting or final median VA had a correlation with recurrence of band keratopathy.
The EDTA chelation procedure was as follows: After informed consent, topical anaesthetic was administered and a lid speculum inserted in theatre. A solution of 20% alcohol was applied to the cornea within a LASIK well for 40 s and the epithelium removed. EDTA (in the form of disodium edetate 0.37% eye drops) was applied to the surface for 3 min intervals and the loosened calcium deposits were debrided with a curved blade. A bandage contact lens was placed at the end of the procedure, and preservative-free dexamethasone 0.1% and chloramphenicol 0.5% drops were prescribed for the post-operative period. (See Figure 2 for illustration of EDTA vial).
Results
A total of 108 cases of EDTA chelation were identified in the period 2009–2015. Of the 108 cases, 10 additionally had an amniotic membrane transplant while one had a conjunctival flap, so these 11 cases were excluded from the series. A further eight case notes were irretrievable. A total of 89 eyes from 72 patients were available for analysis. All patients underwent EDTA chelation by five different surgeons in NHS Greater Glasgow and Clyde, so these results are representative of a typical university hospital setting and population.
Demographics
There were 41 right eyes and 48 left eyes. The mean age was 71 years (range 18–93). Most cases of band keratopathy were idiopathic (36%, 32/89). The most common underlying diagnosis was long-term topical glaucoma therapy (27%). (See Table 1 for other causes.) The median presenting visual acuity was 6/18 (range 6/6-NPL) with the visual axis affected in 97.8% of cases.
The mean duration of the operation was 20 min (range 10–45); which involved corneal epithelium removal, recurrent application of topical EDTA, and mechanical debridement as previously detailed.
Outcomes
There were no documented surgical complications, although one case had a residual epithelial defect at 4 weeks follow-up, which subsequently healed. The mean initial follow-up time was 40 days. At follow-up, the visual axis was found to be clear in 97.8% of cases. Visual acuity was maintained or improved in 79.8%, with 13.5% improving by two lines or more (see Figure 3). However, there was no evidence of a difference between the initial VA and final VA following treatment (Wilcoxon signed rank test P=0.441).
Recurrence
The mean length of total follow-up was 581 days (median 374, maximum 2438). Twenty-five eyes (28.1%) showed localised recurrence of calcium with a mean time of 546 days (median 374), but only four cases (4.5% overall) required repeat EDTA chelation. The median time between operations was 430 days. Of note, 32% of cases of recurrence were associated with hypotony or chronic presence of silicone oil, and 10 of the recurrent cases (40%) had an initial VA of less than 6/120.
There was no evidence of a difference between the median initial VA values between those with and without recurrence (P=0.843, Mann–Whitney), or between the median final VA values between those with and without recurrence (P=0.288, Mann–Whitney) (see Figures 4 and 5: box plots).
Discussion
This large study of 89 treated eyes has documented the main causes of band keratopathy, and the effectiveness of EDTA chelation therapy with good long-term follow-up.
The previous largest review of band keratopathy in the published literature consisted of 65 treated eyes from Wills Eye Hospital, Philadelphia, USA in 2004.2 In their series, they reported the most common cause of band keratopathy to be chronic corneal oedema (28%) followed by idiopathic (25.9%), then phthisis (16.4%). Although not all of their patients underwent EDTA surgical therapy (they identified 230 patients with band keratopathy, but only treated 65 eyes), the range of diagnoses bear similarity to our findings.1, 2
The majority of our cases of band keratopathy needing EDTA chelation had no underlying local or systemic cause identified (idiopathic 36%). The most common cause we identified for band keratopathy was chronic topical glaucoma treatment (27%). Long-term use of topical medications (including active drug, preservatives, and excipients/buffers) have been implicated in the formation of band keratopathy in many studies, and particularly if the ocular surface is unhealthy.16, 17, 18, 19 We have previously published on the potential detrimental impact of chronic excipient exposure in glaucoma patients, best considered to be a low grade ‘lifelong chemical injury’ related to the pH and chronic free radical damage from their ocular anti-hypertensives.20, 21 Both superficial and deep band keratopathy have been linked with the presence of phosphate excipients in topical ophthalmic medications.22 Popiela et al surveyed 78 commonly used ophthalmic medications for information regarding their excipients, and discovered that 22 of the drugs contained phosphates, and a further 43 drugs contained buffers (excipients) other than phosphates.23 The European Medicines Agency has evaluated 117 possible or confirmed case reports of band keratopathy associated with use of phosphate-containing formulations and concluded that prescribers and patients should be aware of a potential causative link to calcification, particularly in those with ocular surface disease.24 This warning has been added to the Summary of Product Characteristics, Section 4.8 (undesirable effects) for phosphate-based medicines.
Visual acuity was seen to be maintained or improved in 79.8% of our cases, with 13.5% improving by two lines or more. However, we were surprised to note that initial and final median visual acuity improvements did not reach statistical significance. This was particularly relevant for patients at the extremes of initial presenting vision, as sub-group analysis revealed that all 29 patients presenting with VA 6/9 or better, and all 22 patients presenting with initial VA less than 6/120 did not achieve an improvement of 2 or more Snellen lines. This observation replicates the published literature that those patients with ‘sick eyes’ are likely to have other ocular comorbidities causing the poor vision, and therefore treatment should be planned for comfort rather than vision.1, 2 Consistent with this, 80% of those patients with counting fingers or worse vision reported improved ocular surface comfort following treatment.
In our series, 28.9% (11/38) of cases with initial visual acuity from 6/12 to 6/120 had a clinically significant (two-line) improvement in visual acuity. Najjar et al reported that 47% of patients presenting with visual acuity 20/50 to 20/400 improved by two or more lines of vision.2 Other studies have reported greater visual improvements, but they included patients who only improved by one line.10 Like the group from Philadelphia, we did not consider this to be visually or clinically significant.1, 2
Recurrence of calcium is a common problem, as EDTA chelation is only treating the calcium precipitated on the cornea, and not the underlying cause.1 We defined recurrence is defined as reformation of band keratopathy affecting the central visual axis, and we found this to occur in 28.1% of cases overall. Nearly half of these cases of recurrence (11/25) had an initial visual acuity of less than 6/120, and the most common diagnoses in this recurrence group were phthisis/hypotony or chronic silicone oil retention. Najjar et al reported a recurrence rate of 17.8%, and their primary diagnoses associated with recurrence were uveitis and herpes simplex keratitis, although they had smaller numbers (65 vs 89 cases) and shorter mean follow-up (432 vs 581 days) than this present series.2
One potential confounding factor to appreciate is that unlike other published studies we did not identify or separate cases of superficial smooth from deep calcareous band keratopathy which probably contributed to the higher recurrence rate observed in our population.10, 12
We were also interested to note that the average time for the surgical procedure was 20 min (range 10–45 min). It has been our observed clinical experience that initial attempts at debridement are often unsuccessful, and patience is required on the part of the surgeon to allow the EDTA to chelate and so clear the calcium chemically. One of our reasons for performing alcohol keratectomy at the start of the procedure is that it is a non-traumatic method of removing the epithelium without breaching Bowman’s. This also allows the EDTA to come into contact with any sub-clinical calcium and remove it chemically. This knowledge should be reflected in the expected scheduling operation time for this procedure, which is often longer than anticipated. The philosophical conflict between the proportional contribution of EDTA chelation of calcium or mechanical debridement of loosened calcium has been debated previously, and would be worthy of further study by analytical chemists.25 However, it is interesting to note that in 1954 Breinin and Devoe26 described the EDTA chelation process as requiring 20 min of contact with the band keratopathy prior to the calcium being amenable to mechanical removal from the stroma, identical to our findings nearly 70 years later!
Conclusion
In this large study with good long-term follow-up, chelation of calcium with topical EDTA was shown to be a safe, well-tolerated and effective treatment for visually significant or symptomatic band keratopathy. The outcomes were favourable as it was successful in clearing the visual axis in 97.8% of cases. Visual acuity was maintained or improved in the majority of cases, with 13.5% improving by two lines or more. The rate of recurrence was seen to be moderate (28.1%); however, the need for retreatment was low (4.5% overall).

References
Jhanji V, Rapuano CJ, Vajpayee RB . Corneal calcific band keratopathy. Curr Opin Ophthalmol 2011; 22 (4): 283–289.
Najjar DM, Cohen EJ, Rapuano CJ, Laibson PR . EDTA chelation for calcific band keratopathy: results and long-term follow-up. Am J Ophthalmol 2004; 137 (6): 1056–1064.
Weng SF, Jan RL, Chang C, Wang JJ, Su SB, Huang CC et al. Risk of band keratopathy in patients with end-stage renal disease. Sci Rep 2016; 6: 28675.
Galor A, Leder HA, Thorne JE, Dunn JP . Transient band keratopathy associated with ocular inflammation and systemic hypercalcemia. Clin ophthalmol 2008; 2 (3): 645–647.
Moisseiev E, Gal A, Addadi L, Caspi D, Shemesh G, Michaeli A . Acute calcific band keratopathy: case report and literature review. J Cataract Refract Surg 2013; 39 (2): 292–294.
Arora R, Shroff D, Kapoor S, Nigam S, Narula R, Chauhan D et al. Familial calcific band-shaped keratopathy: report of two new cases with early recurrence. Indian J Ophthalmol 2007; 55 (1): 55–57.
Althaus C, Schelle C, Sundmacher R . Acute band-shaped keratopathy after intraocular fibrinolysis with recombinant tissue plasminogen activator (rt-PA). Klin Monatsbl Augenheilkd 1996; 209 (5): 318–321.
Wollensak G, Meyer JH, Loffler KU, Funk J . Band keratopathy after tissue-plasminogen activator treatment of postoperative fibrin reaction. Klin Monatsbl Augenheilkd 1996; 209 (1): 43–46.
Bokosky JE, Meyer RF, Sugar A . Surgical treatment of calcific band keratopathy. Ophthalmic Surg 1985; 16 (10): 645–647.
Dighiero P, Boudraa R, Ellies P, Saragoussi JJ, Legeais JM, Renard G . Therapeutic photokeratectomy for the treatment of band keratopathy. J Fr Ophtalmol 2000; 23 (4): 345–349.
Kwon YS, Song YS, Kim JC . New treatment for band keratopathy: superficial lamellar keratectomy, EDTA chelation and amniotic membrane transplantation. J Korean Med Sci 2004; 19 (4): 611–615.
O'Brart DPS, Gartry DS, Lohmann CP, Patmore AL, Kerr Muir MG, Marshall J . Treatment of band keratopathy by excimer laser phototherapeutic keratectomy: surgical techniques and long term follow up. Br J Ophthalmol 1993; 77 (11): 702–708.
Im SK, Lee KH, Yoon KC . Combined ethylenediaminetetraacetic acid chelation, phototherapeutic keratectomy and amniotic membrane transplantation for treatment of band keratopathy. Korean J Ophthalmol 2010; 24 (2): 73–77.
Najjar DM . Management of band keratopathy with excimer phototherapeutic keratectomy. Eye (Lond) 2006; 20 (2): 252.
De Ortueta D, Schreyger F, Baatz H . Band keratopathy: a modified treatment. Eur J Ophthalmol 2006; 16 (4): 618–620.
Schrage NF, Kompa S, Ballmann B, Reim M, Langefeld S . Relationship of eye burns with calcifications of the cornea? Graefes Arch Clin Exp Ophthalmol 2005; 243 (8): 780–784.
Schrage NF, Schlossmacher B, Aschenbernner W, Langefeld S . Phosphate buffer in alkali eye burns as an inducer of experimental corneal calcification. Burns 2001; 27 (5): 459–464.
Schrage NF, Frentz M, Reim M . Changing the composition of buffered eye-drops prevents undesired side effects. Br J Ophthalmol 2010; 94 (11): 1519–1522.
Pavicić-Astalos J, Lacmanović-Loncar V, Petric-Vicković I, Sarić D, Mandić Z, Csik T et al. Eye drops preservative as the cause of corneal band keratopathy in long-term pilocarpine hydrochloride treatment. Acta Clin Croat 2012; 51 (1): 107–111.
Lockington D, Macdonald E, Stewart P, Young D, Caslake M, Ramaesh K . Free radicals and the pH of topical glaucoma medications: a lifetime of ocular chemical injury? Eye 2012; 26 (5): 734–741.
Lockington D, Macdonald E, Gregory M, Stewart P, Caslake M, Ramaesh K . Presence of free radicals in commonly used ophthalmic preparations. Br J Ophthalmol 2010; 94 (4): 525–526.
Bernauer W, Thiel MA, Kurrer M, Heiligenhaus A, Rentsch KM, Schmitt A et al. Corneal calcification following intensified treatment with sodium hyaluronate artificial tears. Br J Ophthalmol 2006; 90 (3): 285–288.
Popiela MZ, Hawksworth N . Corneal calcification and phosphates: do you need to prescribe phosphate free? J Ocul Pharmacol Therapeut 2014; 30 (10): 800–802.
European Medicines Agency. European Medicines Agency. Committee for Medicinal Products for Human Use (CHMP) Questions and answers on the use of phosphates in eye drops. Available at: www.ema.europa.eu/docs/en_GB/document_library/Medicine_QA/2012/12/WC500136247.pdf. Accessed 12 July 2017.
Arjamaa O . EDTA chelation for calcific band keratopathy. Am J Ophthalmol 2005; 139 (1): 216 author reply 216.
Breinin GM, Devoe AG . Chelation of calcium with edathamil calcium-disodium in band keratopathy and corneal calcium affections. AMA Arch Ophthalmol 1954; 52 (6): 846–851.
Acknowledgements
We would like to thank David Young (University of Strathclyde) for his assistance with the statistics. A version of the study was an oral presentation at SOE Congress, Barcelona, Spain in June 2017.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Al-Hity, A., Ramaesh, K. & Lockington, D. EDTA chelation for symptomatic band keratopathy: results and recurrence. Eye 32, 26–31 (2018). https://doi.org/10.1038/eye.2017.264
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2017.264
This article is cited by
-
Bandkeratopathie bei ektoper Vitamin-D-Produktion bei Sarkoidose
Die Ophthalmologie (2023)
-
Clinical outcomes of simultaneous phototherapeutic keratectomy and photoastigmatic keratectomy
Scientific Reports (2021)
-
Advanced Technology IOL Cataract Surgery Optimized for Pre-existing Corneal Disease
Current Ophthalmology Reports (2018)