Abstract
Purpose
To evaluate the ocular manifestations of congenital toxoplasmosis at the first ophthalmological examination of children up to the age of 12 months.
Methods
Cross-sectional study of 44 children with a confirmed diagnosis of congenital toxoplasmosis. In all patients, complete ophthalmological examinations were performed under sedation. The patients underwent biomicroscopy of the anterior segment, skiascopy under cyclopegia, and indirect binocular ophthalmoscopy with maximum mydriasis.
Results
The mean age of patients was 4.2 months. Of the 44 children evaluated, 31 (70.4%) presented ocular involvement and 29 (65.9%) of them had retinochoroiditis lesions. The retinochoroiditis lesions were bilateral in 22 (75.8%) patients and unilateral in 7 (24.2%). The retinochoroiditis lesions were active in 8 (15.7%) eyes and had healed in 43 (84.3%). Most of the lesions were concentrated in the papillomacular area (76.3%). Other associated ocular alterations were present in 22 children, the most prevalent being cataract, microphthalmia, and strabismus.
Conclusion
Ocular involvement in congenital toxoplasmosis might be much more frequent and occurs earlier than previously described.
Similar content being viewed by others
Introduction
Congenital toxoplasmosis, a fetal infection caused by the parasite Toxoplasma gondii, is considered as a major cause of child morbidity and mortality.1 It is distributed worldwide, but its prevalence varies from region to region, according to climate and habits of the population. In recent studies conducted in Rio Grande do Sul, Brazil, a high incidence of congenital toxoplasmosis was shown, ranging from 6 to 8 per 10 000 live births.2, 3, 4
Vertical transmission of toxoplasmosis occurs during primary infection in pregnant women, and generally the maternal disease goes unnoticed. When pregnant women become infected in the first trimester, the frequency of fetal infection goes up to 15–20%, in the second up to 25%, and in the third up to 65–70%. The most compromised fetuses are those who are infected earlier.5, 6, 7
In congenital toxoplasmosis, newborns often do not present clear signs of the systemic disease at birth, but a more detailed investigation may show severe ocular and neurological manifestations.8 Even patients with a subclinical infection run a major risk of developing long-term sequelae, including retinochoroiditis and neurological abnormalities.8, 9, 10, 11, 12, 13
Ocular lesions are the most frequent manifestations of congenital toxoplasmosis.14, 15 Prevalence of retinochoroiditis lesions widely vary in literature.1, 10, 12, 14 These lesions may already be present at birth or they may appear later.10 It should be mentioned that this ocular involvement is serious because, in many cases, it affects the macular region.16, 17, 18
Besides retinochoroiditis, other ocular manifestations of congenital toxoplasmosis are described, such as microphthalmia, optic nerve atrophy, and abnormalities of the iris, cataract, and strabismus.12, 15, 19, 20, 21, 22
A significant reduction in prevalence and severity of the disease has been attributed to prenatal and neonatal treatment maintained throughout the first year of life.23, 24, 25, 26, 27 However, there is currently an active discussion about the benefits of the treatment of congenital toxoplasmosis; it is thus important to know in detail the natural history of the disease.28, 29
The purpose of this work is to study the ocular manifestations in the first examination of confirmed cases of congenital toxoplasmosis in children under the age of 12 months.
Materials and methods
This cross-sectional study included all patients with a definite diagnosis of congenital toxoplasmosis who were referred to the Uveitis Section at Hospital de Clínicas de Porto Alegre, Brazil, between January 2000 and December 2004. All patients were referred by paediatricians from three different services throughout the city. To be included, the children must be 12 months old or younger and present at least one of the following items.30
-
Detection of IgM anti-toxoplasma antibodies in peripheral blood within the first 6 months of life;
-
Increased IgG anti-toxoplasma antibodies in successive dosages within the first year of life;
-
Maintenance of IgG anti-toxoplasma antibodies in peripheral blood after the age of 12 months.
The diagnostic suspicion of congenital toxoplasmic infection was raised because of the following situations: (1) prenatal screening or screening of the mother at delivery. In these situations, the mother seroconverted during pregnancy or had a serological profile suggestive of recent toxoplasmosis—presence of IgM anti-toxoplasma antibodies with low-avidity IgG; (2) neonatal screening when the mother was not suspected of having toxoplasmosis during pregnancy, but positive IgM anti-toxoplasma antibodies were found in the infant at routine neonatal screening.
The time when the mother acquired the infection and first produced a serological (IgM) response was rarely identified precisely, but the trimester of fetal infection could be identified by placing the time of maternal infection between the last negative serological test and the first positive one. To avoid selection bias, children referred because of ocular symptoms without previous suspicion of congenital toxoplasmosis were not included.
The initial investigation consisted of delivering the term of consent and interviewing the family member(s) responsible. The ophthalmological examination of the patient was performed under chloral hydrate sedation with a dosage of 50 mg/kg to perform a more thorough examination. The patients underwent biomicroscopy of the anterior segment, skiascopy under cyclopegia with cyclopentolate at 1%, and indirect binocular ophthalmoscopy with maximum mydriasis and scleral indentation when needed.
The following data were catalogued during this study: period of maternal infection, presence and characteristics of all ocular alterations found in the children, treatment performed after toxoplasmic diagnosis in the pregnant women, and treatment of the patients before the first examination. Patients who were treated received pirimetamine (1 mg/kg per day), sulfadiazine (100 mg/kg per day), and folinic acid (10 mg thrice weekly). In the presence of active lesions, prednisone (1 mg/kg per day) was introduced.
The retinal lesions were analysed in terms of laterality, activity, and topographic location. For this purpose, the retinal surface was divided into zones 1, 2, and 3. Zone 1 corresponds to the retinal area between the superior and inferior temporal arcades, including the macula, the optic disc, and the peridiscal region in an extension of the disc diameter. Zone 2 extends to the equator at the level of the ampullae of the vorticose veins. The remainder of the peripheral retina was considered as zone 3 (Figure 1). This division of the retinal surface is similar to that described by Holland et al31 in a study on cytomegalovirus retinopathy.
Data on the patients were obtained by using frequency tables for qualitative variables and by using mean and standard deviation for quantitative variables.
This study was approved by the Ethics Committee at the Hospital de Clínicas de Porto Alegre and an informed consent was obtained from the legal representatives of all patients included in the study.
Results
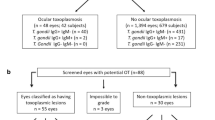
Data from the first ophthalmological consultation of 44 children with congenital toxoplasmosis, born between January 2000 and December 2004, were evaluated. Congenital toxoplasmosis was previously suspected and diagnosed at the hospital where the child was born or by the paediatrician after the positive result of neonatal screening, and then referred to the Ophthalmology Service at Hospital de Clínicas de Porto Alegre for ophthalmological follow-up.
The diagnosis was confirmed in 38 (86.3%) patients because they presented positive IgM anti-toxoplasma antibodies in peripheral blood, in 5 (11.4%) because of the persistence of IgG anti-toxoplasma antibodies after the age of 12 months, and in 1 (2.3%) because of the increased IgG anti-toxoplasma titres in successive dosages during the first year.
The average age of the patients was 4.2 months (2 days–12 months) (Table 1).
With regard to gender, 24 (54.5%) were female; 41 (93.2%) were white, and 3 (6.8%) were black or brown. With regard to origin, 21 (47.7%) were from the city of Porto Alegre and 23 (52.3%) from other regions of the state of Rio Grande do Sul.
With regard to the period of maternal infection, 21 (47.7%) mothers were infected during the third trimester of pregnancy, 3 (6.8%) during the second trimester, and 1 (2.3%) during the first trimester. A total of 19 mothers (43.2%) have no information on the period of infection owing to lack of prenatal care (Table 1). In the latter cases, suspicion of toxoplasmosis was raised at delivery, when the mother presented serological evidence of recent toxoplasmosis, or post-delivery, when the infant had positive T. gondii-specific IgM found by neonatal screening. Only one pregnant woman received treatment for toxoplasmosis.
Before the first ophthalmological visit, 36 (81.8%) children were receiving treatment for congenital toxoplasmosis and 8 (18.2%) were not. A total of 44 children were examined and 31 (70.4%) presented ocular involvement, 29 (65.9%) showed retinochoroiditis lesions, and 5 patients had media opacities that prevent fundoscopy, 2 of them in both eyes. In addition to the retinal lesions, other alterations were found in 22 children, 12 of them with strabismus, 7 with nystagmus, 6 with cataract, and 5 with microphthalmia.
A total of 81 eyes could be examined fundoscopically, and 51 (62.7%) of them had retinal lesions. Of a total of 88 eyes examined, 7 (7.9%) presented media opacities, which prevented fundus examination, and 30 (34%) did not show retinal lesions (Table 1).
The lesions were bilateral in 22 (75.8%) and unilateral in 7 (24.2%) children. The morphological aspect of the lesions varied widely from the typical wagon-wheel-shaped lesion (Figure 2) to retinal necrotic focal lesions of different sizes and with a variable presence of pigment (Figure 3).
The retinal lesions showed signs of activity in 8 (15.7%) eyes (Figure 4) and they were healed in 43 (84.3%). The mean age of the patients with active lesions was 1.4 months.
With regard to the location of the retinal lesions, 31.3% were present only in zone 1, 17.6% in zone 2, and 3.9% in zone 3. The lesions were found simultaneously in zones 1 and 2 in 35.3% of the eyes, in zones 1+3 and 2+3 affected 4.1% of the eyes; all the three zones were affected (Figure 5).
Discussion
Congenital toxoplasmosis is a worldwide prevalent disease capable of causing severe manifestations, in which ophthalmological alterations are the most frequent. Such alterations are often factors that prevent the affected individual from having a normal life, frequently resulting in damage to the family and subsequently to the society as a whole.15
This study included patients from several areas of the state of Rio Grande do Sul, which is located in the southern region of Brazil. Previous studies show that this region has a high prevalence of ocular acquired toxoplasmosis, and concomitantly, the rate of congenital toxoplasmosis currently can be considered as one of the highest in the world.2, 3, 4, 32, 33
It has been widely accepted that approximately 90% of newborns with a serological diagnosis of congenital toxoplasmosis have a normal physical examination at birth and only about 40% present ocular and/or neurological alterations after further investigation.6, 15, 18 This study definitely showed that ocular involvement may be more frequent and occurs earlier than previously described. The mean age of our patients was 4.2 months and, when considering only the patients with active lesions, the mean age is lower—1.4 months. Our study showed that 70.4% of the patients already presented some kind of ocular involvement at the first examination. This means that more patients are affected than the data from standard clinical examination by a paediatrician reveal.
If we only consider the retinochoroiditis lesions found in our study (65.9%), the rates found are similar to those described in other studies (Table 2). However, it should be pointed out that the findings in our study were obtained at the first ophthalmological examination of children up to the age of 12 months and only the paper of Mets et al14 showed results similar to ours.
In other studies that gave similar results, patient follow-up was done for up to 27 years. As it is known that congenital toxoplasmosis may present relapses in its evolution, the coincident results indicate that the patients in our study presented a more severe and precocious involvement. Another possibility to explain the similarities of our results with those in the literature, which studies older patients, is the difficulty in performing fundoscopy among the newborns. The examination in the present study was carried out with indirect binocular ophthalmoscopy, extreme mydriasis, and with sedation, to enable complete visualisation of the peripheral retina, thus avoiding the underdiagnosis of retinal lesions. Therefore, it may be assumed that some of the lesions diagnosed late in these studies have already existed, but have not been detected in the initial investigations.14
The presence of retinochoroiditis lesions in 65.9% of our patients confirms the observation that this is the most frequent alteration of congenital toxoplasmosis.14, 20 With regard to the location of the lesions in this study, 76.3% involves zone 1, which corresponds to the macular and peripapillary areas.
We highlight the fact that involvement of the macular area leads to greatest damages, and thus, more than 75% of our patients are at a high risk of major, irreversible visual loss. However, we stress that the foveal region in some cases can be spared, which ensures good vision for the patient in the future, even in cases of zone 1 involvement.14, 34
The importance of lesions located in zones 2 and 3, however, cannot be attenuated. Considering that ocular toxoplasmosis is a relapsing disease, peripheral lesions may originate other active foci later, which could cause great visual loss, mainly in younger children who usually do not inform others of their sight symptoms.
The other ophthalmological alterations observed, such as cataract, microphthalmia, strabismus, and nystagmus, showed the parasite's preference also for other ocular structures, and these had already been described in the literature. Some of these findings could also be related to intracranial neurological damage or complications of retinal lesions. In the cases with cataract, we cannot rule out the existence of concomitant retinal lesions. The high active uveitis rate found in our study probably may be related to the young age of the patients and the early initial consultation.
The severity of congenital infection by T. gondii found in this study may be established by the high number of patients with such severe ocular alterations that they prevented fundoscopy. It can be presumed that most of these patients would also have retinochoroiditis lesions, which would increase the total number of patients with this type of alteration. To explain this fact, it can be postulated that the parasite genotypes presented in our region are more virulent than the ones existing in other regions of the world.32, 35, 36, 37 Bilateral ocular involvement occurred in 75.8% of the cases, thus showing the broad involvement of the nervous system by the parasite during the fetal stages of development. This finding had already been noted since the first descriptions of the disease, which showed that bilateral involvement is a typical characteristic of congenital toxoplasmosis.16
Most of the patients in this study received specific treatment. Only eight patients were not treated before our first examination and no differences were found in the lesions with respect to the remaining patients. The effectiveness of treatment was not an outcome evaluated in this work, but the benefit of treatment in congenital toxoplasmosis has already been widely shown.23, 24, 25 We stress that 17.6% of the patients studied presented active lesions at the time of the examination, which led us to add the use of corticosteroids in addition to the specific treatment of the disease to minimise the damage caused by the accompanying inflammatory reaction.
Contrary to what has been accepted so far, this study shows that ocular involvement in congenital toxoplasmosis, in Brazil, has reached approximately 70% in the first year of life. The patients must remain under follow-up to monitor recurrences and other ocular alterations caused over the long term by toxoplasmosis.
Owing to the difficulty in the complete visualisation of the peripheral retina in infants, we believe that a complete ophthalmological examination with indirect binocular ophthalmoscope, extreme mydriasis, and under sedation must be carried out in every child with suspected congenital infection by T. gondii.
References
Remington JS, McLeod R, Thulliez P, Desmonts G . Toxoplasmosis. In: Remington JS, Klein JO, Wilson CB, Baker CJ (eds). Infectious Diseases of the Fetus and Newborn Infant. 6th ed. Elsevier-Saunders: Philadelphia, 2006, pp 948–1091.
Spalding SM, Amendoeira MR, Ribeiro LC, Silveira C, Garcia AP, Camillo-Coura L . Estudo prospectivo de gestantes e seus bebês com risco de transmissão de toxoplasmose congênita em município do Rio Grande do Sul. Rev Soc Bras Med Trop 2003; 36 (4): 483–491.
Mozzatto L, Procianoy RS . Incidence of congenital toxoplasmosis in southern Brazil: a prospective study. Rev Inst Med Trop Sao Paulo 2003; 45 (3): 147.
Lago EG, Neto EC, Melamed J, Rucks AP, Presotto C, Coelho JC et al. Congenital toxoplasmosis: late pregnancy infections detected by neonatal screening and maternal serological testing at delivery. Paediatr Perinat Epidemiol 2007; 21: 525–531.
Dunn D, Wallon M, Peyron F, Petersen E, Peckham C, Gilbert R . Mother-to-child transmission of toxoplasmosis: risk estimates for clinical counseling. Lancet 1999; 353: 1829–1833.
Gilbert RE . Toxoplasmosis. In: Newel ML, McIntyre J (eds). Congenital and Perinatal Infections. Cambridge University Press: Cambridge, 2000, p 305.
Montoya JG, Liesenfeld O . Toxoplasmosis. Lancet 2004; 363: 1965–1976.
Wilson CB, Remington JS, Stagno S, Reynolds DW . Development of adverse sequelae in children born with subclinical congenital toxoplasma infection. Pediatrics 1980; 66: 767–774.
Lower-Sieger DH, Rothova A, Koppe JG, Kylstra A . Congenital toxoplasmosis, a prospective study based on 1821 pregnant women. In: Saari KM (ed). Uveitis Update. Elsevier Science Publishers; Amsterdam-New York, 1984, pp 203–207.
Koppe JG, Loewer Sieger DH, de Roever Bonnet H . Results of 20-year follow-up of congenital toxoplasmosis. Lancet 1986; 1: 254–255.
Figueiredo ARP, Oréfice F, Caiaffa WT, Antunes CMF, Chiari CA . Aspectos oftalmológicos da associação toxoplasmose congênita - deficiência mental. Arq Bras Oftalmol 1989; 52: 84–89.
Peyron F, Wallon M, Bernardoux C . Long-term follow-up of patients with congenital ocular toxoplasmosis. N Engl J Med 1996; 334: 993–994.
Melamed J, Dornelles F, Eckert GU . Alterações tomográficas cerebrais em crianças com lesões oculares por toxoplasmose congênita. J Pediatr 2001; 77: 475–480.
Mets MB, Holfels E, Boyer KM, Swisher CN, Roizen N, Stein M et al. Eye manifestations of congenital toxoplasmosis. Am J Ophthalmol 1996; 122: 309–324.
Meenken C, Assies J, van Nieuwenhuizen O, Holwerda-van der Maat WG, van Schooneveld MJ, Delleman WJ et al. Long-term ocular and neurological involvement in severe congenital toxoplasmosis. Br J Ophthalmol 1995; 79: 581–584.
François J . La Toxoplasmose et ses Manifestations Oculars. Editeurs Masson et Cie: Paris, 1963.
Pinheiro SRA, Orefice F, Andrade GMQ, Caiaffa WT . Estudo da toxoplasmose ocular em famílias de pacientes portadores de toxoplasmose congênita sistêmica e ocular. Arq Bras Oftalmol 1990; 53: 4–6.
Guerina NG, Hsu HW, Meissner HC, Maguire JH, Lynfield R, Stechenberg B et al. Neonatal serologic screening and early treatment for congenital Toxoplasma gondii infection. N Engl J Med 1994; 330: 1858–1863.
Orefice F, Pinheiro SRA, Ferreira CD . Calcificações no plano coróide –retina de pacientes com lesões cicatrizadas de toxoplasmose congênita diagnosticadas pela tomografia computadorizada. Rev Bras Oftalmol 1989; 48: 49–53.
Bahia MD, Oréfice F, Andrade CMQ . Análise clínica das lesões de retinocoroidite em crianças portadoras de toxoplasmose congênita. Rev Bras Oftalmol 1992; 51: 265–271.
Vutova K, Peicheva Z, Popova A, Markova V, Mincheva N, Todorov T . Congenital toxoplasmosis: eye manifestations in infants and children. Ann Trop Paediatr 2002; 22: 213–218.
Kodjikian L, Wallon M, Fleury J, Denis P, Binquet C, Peyron F et al. Ocular manifestations in congenital toxoplasmosis. Graefes Arch Clin Exp Ophthalmol 2006; 244: 14–21.
Couvreur J, Desmonts G, Aron-Rosa D . Le pronostic oculaire de la toxoplasmose congénitale: rôle du traitement. Ann Pédiatr 1984; 31: 855–858.
Hohlfeld P, Daffos F, Thulliez P, Aufrant C, Couvreur J, MacAleese J et al. Fetal toxoplasmosis: outcome of pregnancy and infant follow-up after in utero treatment. J Pediatr 1989; 115: 765–769.
Mc Auley J, Boyer KM, Patel D, Mets M, Swisher C, Roizen N et al. Early and longitudinal evaluations of treated infants and children and untreated historical patients with congenital toxoplasmosis: the Chicago collaborative treatment trial. Clin Infect Dis 1994; 18: 38–72.
Brézin AP, Thulliez P, Couvreur J, Nobré R, McLeod R, Mets MB . Ophthalmic outcomes after prenatal and postnatal treatment of congenital toxoplasmosis. Am J Ophthalmol 2003; 135: 779–784.
McLeod R, Boyer K, Karrison T, Kasza K, Swisher C, Roizen N et al. Outcome of treatment for congenital toxoplasmosis, 1981–2004: The National Collaborative Chicago-based, Congenital Toxoplasmosis Study. Clin Infect Dis 2006; 42: 1383–1394.
Gras L, Gilbert RE, Ades AE, Dunn DT . Effect of prenatal treatment on the risk of intracranial and ocular lesions in children with congenital toxoplasmosis. Int J Epidemiol 2001; 30: 1309–1316.
Petersen E . Toxoplasmosis. Semin Fetal Neonatal Med 2007; 12: 214–223.
Lebech M, Joynson DHM, Seitz HM, Thulliez P, Gilbert RE, Dutton GN et al. Classification system and case definitions of Toxoplasma gondii infection in immunocompetent pregnant women and their congenitally infected offspring. Eur J Clin Microbiol Infect Dis 1996; 15: 799–805.
Holland GN, Buhles WC, Mastre B, Kaplan HJ . A controlled retrospective study of gancyclovir treatment for cytomegalovirus retinopathy. Arch Ophthalmol 1989; 107: 1759–1766.
Melamed J . Peculiarities of Ocular Toxoplasmosis in Rio Grande do Sul, Brazil. World Uveitis Symposium. Roca. Roca Publishers; São Paulo, 1989, pp 339–348.
Neves JM, Nascimento LB, Ramos JGL, Martins-Costa SH . Toxoplasmose na gestação. Rev Bras Ginecol Obstet 1994; 16: 197–202.
Tan HK, Schmidt D, Stanford M, Teär-Fahnehjelm K, Ferret N, Salt A et al. Risk of visual impairment in children with congenital toxoplasmic retinochoroiditis. Am J Ophthalmol 2007; 144: 648–653.
Boothhroid JC, Grigg ME . Population biology of Toxoplasma gondii and its relevance to human infections: do different strains cause different disease? Curr Opin Microbiol 2002; 5: 438–442.
Kahn A, Jordan C, Muccioli C, Vallochi AL, Rizzo LV, Belfort R et al. Genetic divergence of Toxoplasma gondii strains associated with ocular toxoplasmosis, Brazil. Emerg Infect Dis 2006; 12: 942–949.
Gilbert RE, Freeman K, Lago EG, Bahia-Oliveira LMG, Tan HK, Wallon M et al. Ocular sequelae of congenital toxoplasmosis in Brazil compared with Europe. PLoS, Negl Trop Dis 2008; 2: 1–7.
Acknowledgements
This study was designed by JM, GUE, and VSS. This study was conducted by JM, VSS, EGL, and FU. The collection, management, analysis, and interpretation of data was done by JM, GUE, and EGL. The preparation, review, or approval of the paper was done by JM, GUE, VSS, EGL, FU.
Author information
Authors and Affiliations
Corresponding author
Additional information
Funding/support: None.
Conflict of interest: None.
Rights and permissions
About this article
Cite this article
Melamed, J., Eckert, G., Spadoni, V. et al. Ocular manifestations of congenital toxoplasmosis. Eye 24, 528–534 (2010). https://doi.org/10.1038/eye.2009.140
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2009.140
Keywords
This article is cited by
-
The Brazilian Toxoplasma gondii strain BRI caused greater inflammation and impairment in anxiogenic behavior in mice, which was reverted by rosuvastatin treatment
Parasitology Research (2024)
-
Ocular toxoplasmosis: a review of the current diagnostic and therapeutic approaches
International Ophthalmology (2022)
-
Einseitige Mikrophthalmie bei einem 4 Monate alten Säugling
Monatsschrift Kinderheilkunde (2021)
-
Evaluation of colostrum as an alternative biological sample for the diagnosis of human congenital toxoplasmosis
BMC Infectious Diseases (2015)