Abstract
Purpose Ptosis is common in patients with mitochondrial disease. Whilst surgical shortening of the levator muscle can mechanically elevate the lid, this procedure does not restore normal movement and leaves patients at risk of corneal exposure due to concomitant ophthalmoparesis. Recent studies have shown that bupivacaine-induced muscle regeneration is capable of reversing the molecular genetic and biochemical defect in patients with mitochondrial myopathies. This study was undertaken to assess the potential of this approach in restoring levator muscle function in patients with mitochondrial disease and ptosis.
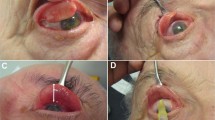
Methods The levator muscle of one eye in five patients with molecularly genetically confirmed mitochondrial DNA disease and ptosis was directly injected with 3 ml of bupivacaine hydrochloride (0.75%). Levator function was compared before and 3 months after the injection.
Results No objective clinical improvement in levator function was detected following bupivacaine administration.
Discussion The lack of functional recovery seen in our patients is most likely to result from a failure of bupivacaine to induce sufficient regeneration necessary to improve levator muscle function. This result indicates that consideration now needs to be given to the use of alternative and more potent myotoxic agents capable of inducing a more widespread regenerative response from the endogenous muscle satellite cells which contain low or undetectable amounts of mutant mitochondrial DNA.
Similar content being viewed by others
Article PDF
References
Chinnery PF, Turnbull DM . Clinical features, investigation, and management of patients with defects of mitochondrial DNA. J Neurol Neurosurg Psychiatry 1997;63:559–63.
Newman NJ . Mithochondril diseases and the eye. Ophthalmol Clin North Am 1992;5:405–24.
Pavlakis SG, Rowland LP, DeVivo DC, Bonillo E, DiMauro S . Mitochondrial myopathies and encephalomyopathies. In: Plum F, editor. Contemporary neurology. Philadelphia: Davis, 1988:95–133.
Fu K, Hartlen R, Johns T, Genge A, Karparti G, Shoubridge EA . A novel heteroplasmic tRNAleu(CUN) mtDNA point mutation in a sporadic patient with mitochondrial encephalomyopathy segregates rapidly in skeletal muscle and suggests an approach to therapy. Hum Mol Genet 1996;5:1835–40.
Clark K, Bindoff LA, Lightowlers RN, et al. Reversal of a mitochondrial DNA defect in human skeletal muscle. Nature Genet 1997;16:222–4.
Wallace DC . Mitochondrial genetics: a paradigm for aging and degenerative diseases? Science 1992;256:628–32.
Hayashi J-I, Ohta S, Kikuchi A, Takemitsu M, Goto Y-I, Nonaka I . Introduction of disease-related mitochondrial DNA deletions into HeLa cells lacking mitochondrial DNA results in mitochondrial dysfunction. Proc Natl Acad Sci USA 1991;88:10614–8.
Boulet I, Karpati G, Shoubridge EA . Distribution and threshold expression of the tRNALys mutation in skeletal muscle of patients with myoclonic epilepsy and ragged-red fibres (MERRF). Am J Hum Genet 1992;51:1187–200.
Chomyn A, Martinuzzi A, Yoneda M, et al. MELAS mutation in mtDNA binding site for transcription termination factor causes defects in protein synthesis and in respiration but no change in levels of upstream and downstream mature transcripts. Proc Natl Acad Sci USA 1992;89:422–15.
Sciacco M, Bonilla E, Schon EA, DiMauro S, Moraes CT . Distribution of wild-type and common deletion forms of mtDNA in normal and respiration-deficient muscle fibers from patients with mitochondrial myopathy. Hum Mol Genet 1994;3:13–9.
Porteous WK, James AM, Sheard PW, et al. Bioenergetic consequences of accumulating the common 4977-bp mitochondrial DNA deletion. Eur J Biochem 1998;257:192–201.
Moraes CT, Schon EA, DiMauro S, Miranda AF . Heteroplasmy of mitochondrial genomes in clonal cultures from Kearns-Sayre syndrome. Biochem Biophys Res Commun 1989;160:765–71.
Shoubridge EA . Molecular histology of mitochondrial diseases. In: DiMauro S, Wallace DC, editor. Mitochondrial DNA in human pathology. New York: Raven Press, 1993:109–23.
Weber K, Wilson JN, Taylor L, Johnson MA, Turnbull DM, Bindoff LA . A new mtDNA mutation showing accumulation with time and restriction to skeletal muscle. Am J Hum Genet 1997;60:373–80.
Bischoff R . The satellite cell and muscle regeneration. In: Engel AG, Franzini-Armstrong C, editor. Myology, Vol 1. 2nd ed.. New York: McGraw-Hill, 1994:97–118.
Benoit PW, Belt WD . Destruction and regeneration of skeletal muscle after treatment with a local anaesthetic, bupivacaine. J Anat 1970;107:547–56.
Carlson BM, Emerick S, Komorowski TE, Rainin EA, Shepard BM . Extraocular muscle regeneration in primates: local anaesthetic-induced lesions. Ophthalmology 1992;99:582–9.
Foster AH, Carlson BC . Myotoxicity of local anaesthetics and regeneration of damaged muscle fibers. Anesth Analg 1980;59:727–36.
Carlson BM, Edgar EA . Rat extraocular muscle regeneration. Arch Ophthalmol 1985;103:1373–7.
Wardell TM, Chrzanowska-Lightowlers ZMA, Andrews RM, Griffiths PG, Chinnery PF, Turnbull DM . Low levels of mutant mtDNA in extraocular muscle myoblasts. In preparation.
Johnson MA, Bindoff LA, Turnbull DM . Cytochrome c oxidase activity in single muscle fibres: assay techniques and diagnostic applications. Ann Neurol 1993;33:28–35.
Birch-Machin M, Jackson S, Singh Kler R, Turnbull DM . Study of skeletal muscle mitochondrial dysfunction. Methods Toxicol 1993;2:51–69.
Hall-Craggs ECB . Rapid degeneration and regeneration of a whole skeletal muscle following treatment with bupivacaine (Marcaine). Exp Neurol 1974;43:349–58.
Porter JD, Edney DP, McMahon EJ, Burns LA . Extraocular myotoxicity of the retrobulbar anaesthetic bupivacaine hydrochloride. Invest Ophthalmol Vis Sci 1988;29:163–74.
Porter JD, Baker RS, Ragusa RJ, Brueckner JK . Extraocular muscles: basic and clinical aspects of structure and function. Surv Ophthalmol 1995;39:451–84.
Author information
Authors and Affiliations
Corresponding author
Additional information
Commercial or proprietary interest: None
Rights and permissions
About this article
Cite this article
Andrews, R., Griffiths, P., Chinnery, P. et al. Evaluation of bupivacaine-induced muscle regeneration in the treatment of ptosis in patients with chronic progressive external ophthalmoplegia and Kearns-Sayre syndrome. Eye 13, 769–772 (1999). https://doi.org/10.1038/eye.1999.225
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/eye.1999.225
Keywords
This article is cited by
-
Mitochondrial Diseases: Therapeutic Approaches
Bioscience Reports (2007)
-
Chronic progressive external ophthalmoplegia
Current Neurology and Neuroscience Reports (2002)
-
Gene therapy for mitochondrial disease by delivering restriction endonucleaseSmaI into mitochondria
Journal of Biomedical Science (2002)
-
Mitochondrial disease
Current Treatment Options in Neurology (2001)