Abstract
Granulocyte-macrophage colony-stimulating factor (GM-CSF, also called CSF-2) is best known for its critical role in immune modulation and hematopoiesis. A large body of experimental evidence indicates that GM-CSF, which is frequently upregulated in multiple types of human cancers, effectively marks cancer cells with a ‘danger flag’ for the immune system. In this context, most studies have focused on its function as an immunomodulator, namely its ability to stimulate dendritic cell (DC) maturation and monocyte/macrophage activity. However, recent studies have suggested that GM-CSF also promotes immune-independent tumor progression by supporting tumor microenvironments and stimulating tumor growth and metastasis. Although some studies have suggested that GM-CSF has inhibitory effects on tumor growth and metastasis, an even greater number of studies show that GM-CSF exerts stimulatory effects on tumor progression. In this review, we summarize a number of findings to provide the currently available information regarding the anticancer immune response of GM-CSG. We then discuss the potential roles of GM-CSF in the progression of multiple types of cancer to provide insights into some of the complexities of its clinical applications.
Similar content being viewed by others
Introduction
The hematopoietic growth factor granulocyte-macrophage colony-stimulating factor (GM-CSF) regulates hematopoietic stem cell/progenitor cell differentiation into dendritic cells (DCs), granulocytes, and macrophages in the bone marrow.1 In clinical oncology, immune responses against multiple infectious agents or cancer cells are activated by GM-CSF acting as an immune stimulant to increase various immune cell activities.2 These encouraging results have led to numerous clinical trials of GM-CSF to evaluate whether it can enhance antitumor immune responses against a number of cancer types by promoting the activation, maturation and migration of various immune cells that may result in successful tumor treatment. In addition, a growing body of evidence suggests that GM-CSF is produced and secreted by a wide variety of non-immune cell types, including fibroblasts, keratinocytes and endothelial cells in response to appropriate stimuli.3, 4 GM-CSF promotes the growth and migration of tumor cells by enhancing the expression of MMPs,5 and it induces keratinocyte growth, thereby accelerating wound healing.6, 7 Because of these effects, GM-CSF has been used in adjuvant tumor therapies. However, the value of GM-CSF is still highly controversial because of its different effects on tumor progression depending on the tumor type or cancer model. A large body of experimental evidence indicates that GM-CSF can act as a tumor-derived factor that may promote tumor growth and progression. In multiple cancer models, constitutive GM-CSF protein expression and secretion has been observed, frequently together with its conjugate receptors.8, 9, 10, 11 An increased level of GM-CSF in serum is considered a potential diagnostic and prognostic marker indicating poor prognosis in colorectal cancer patients.12 Enhanced GM-CSF protein levels, together with platelet-derived growth factor (PDGF) and vascular endothelial growth factor, were found to be significantly associated with invasion and poor prognosis in patients with head and neck cancers.13 Consistent with this finding, previous studies have suggested that GM-CSF promotes cancer cell proliferation and migration in a variety of solid tumors and cancer cell lines.5, 14, 15, 16 These results suggest that in addition to its immune-stimulatory functions, GM-CSF may have direct effects on tumor progression and invasion. Therefore, in the current review, we provide an overview of the existing empirical findings and summarize both the advantages and disadvantages of the growing influence of GM-CSF on tumorigenesis to give directions for future research.
Discovery of GM-CSF and its biological functions
GM-CSF was first purified from the conditioned medium of mouse lung tissue treated with endotoxin lipopolysaccharide as a small glycoprotein (24–33 kDa), which was able to stimulate the proliferation of bone marrow-derived macrophages and granulocytes.17 GM-CSF isolated from mouse lung conditioned medium stimulates the proliferation of multiple types of hematopoietic cells, including macrophages, eosinophils, erythroid cells, granulocytes and megakaryocytes, in a concentration-dependent manner.18 GM-CSF is also able to stimulate the development and maturation of leukemic hematopoietic cells into neutrophils, eosinophils and monocytes.18 In addition, GM-CSF is produced and secreted by a number of different cell types, including activated T cells, B cells, macrophages, mast cells, vascular endothelial cells and fibroblasts, generally in response to inflammatory cytokines and innate immune activation.17, 19, 20 GM-CSF may also have an important role in regulating the extracellular matrix by modulating the metabolism of vascular collagens. Moreover, GM-CSF can promote the proliferation and migration of vascular endothelial cells, thus contributing to angiogenic processes,21 and induces keratinocyte proliferation and migration, which, in turn, stimulates wound healing.22, 23
Molecular mechanisms underlying GM-CSF regulation
GM-CSF exerts all of its biological activities by binding and activating its cognate heteromeric receptor (also known as CD116), which is present on multiple cell types, including endothelial cells, granulocytes, lymphocytes, macrophages and monocytes.24 The GM-CSF receptor is composed of at least two different subunits, the alpha chain and the beta chain, which are also present in the common receptors for interleukin-3 (IL-3) and IL-5.25 The alpha subunit contains ligand-binding sites,26 whereas the beta subunit complexes with the alpha protein and mediates receptor signal transduction.27 Onetto-Pothier et al.28 demonstrated the presence of two classes of GM-CSF receptors on acute myeloid leukemic cells: a high-binding affinity receptor for the ligand with a dissociation constant (kd) of 3–73 pmol l−1; and a second class of receptor with a low-binding affinity (a kd of 1–10 nmol l−1) for the ligand. Interestingly, both subunits lack intrinsic catalytic domains such as tyrosine kinase domains.18 The cytoplasmic domains of the GM-CSF receptor beta chain are constitutively associated with the kinase Janus kinase 2 (JAK2).29 These GM-CSF receptor beta-chain-bound JAK2 molecules may cross-phosphorylate both each other and the receptor beta chain itself.29 This phosphorylation is sufficient to trigger multiple intracellular signaling pathways, including STAT5 and MAPK.1, 30 Subsequently, multiple GM-CSF target genes are constitutively activated, including the transcription factor PU.1, which regulates the appropriate differentiation and maturation of macrophages.31
Regulation of GM-CSF production
GM-CSF is produced and secreted by a number of different cell types, including activated T cells, B cells, macrophages, mast cells, vascular endothelial cells, fibroblasts and a wide variety of cancer cell types.18 GM-CSF expression is rapidly stimulated in response to lipopolysaccharide and pro-inflammatory cytokines, including IL-1,32 IL-633 and tumor necrosis factor-α,34 whereas its expression can be successfully inhibited by IL-4,35 IL-1036 and IFN-γ.37 In addition, immunosuppressive agents, including cyclosporine A, potently reduced GM-CSF production.38 Similarly, a synthetic glucocorticoid, dexamethasone, markedly inhibited GM-CSF secretion in human retinal pericytes, monocytes and retinal endothelial cells.39 Normally, the serum GM-CSF concentrations are extremely low or even undetectable, whereas the immunoreactive circulating levels are significantly elevated in response to inflammatory stimuli, including bacterial endotoxins and local infections. Consistent with these results, elevated GM-CSF levels are seen in the skin of lesions from atopic dermatitis patients. Correspondingly, enhanced GM-CSF secretion from keratinocytes may contribute to the chronicity of inflammatory lesions by enhancing the antigen-presenting functions of DCs.40 In synovial fluid from patients with rheumatoid arthritis, measurable levels of GM-CSF support the differentiation of an inflammatory DC population,41 which may, in turn, influence bone loss and joint destruction. Noster et al.42 showed that synovial GM-CSF production by human CD4+ T cells is stimulated in response to the Th1-polarising cytokine IL-12 and the T-cell survival factor IL-15. However, it is not fully understood how the GM-CSF secretion from helper T cells is regulated.
Immune-dependent antitumor activity of GM-CSF
GM-CSF as an adjuvant in immunotherapy
The human immune system, which maintains healthy barrier homeostasis against diverse insults and minimizes inflammation and cellular dysregulation, is divided into two extremely broad categories: innate and adaptive immunity. The innate immune system evolved numerous defense mechanisms to quickly recognize and respond to a wide variety of preprogrammed inflammatory responses involving various soluble factors, including complement and antimicrobial peptides, as well as multiple cellular components, including DCs, macrophages, mast cells and natural killer cells. The adaptive immune response is a slower-acting, longer-lasting and more specific response than the innate response.43 The concept that the immune system can recognize and eliminate antigen-bearing cancer cells is known as cancer immuno-surveillance, which has a critical role in the host defense against the initiation and progression of cancer.44, 45 The ability of the immune system to eliminate abnormal or cancerous cells has been a major focus of cancer immunotherapy based on enhancing host protective antitumor immunity.46 Constitutive GM-CSF-producing cancer cells stimulate potent, long-lasting and specific anti-tumor immunity by priming CD4+ and CD8+ T cells to recognize circulating tumor-associated antigens, which in turn induce a systemic antitumor-specific immune response.47 The mechanism underlying this GM-CSF-mediated antitumor immunity is believed to rely in part on the enhanced local recruitment and activation of DCs,48 which may result in the enhancement of tumor antigen-associated presentation to T cells in tumor-draining lymph nodes49 and in the activation of other cellular elements of the immune response, including granulocytes, macrophages and NK cells.48, 50 Therefore, GM-CSF is critical to the regulation of anti-tumor immune responses, mainly by the activation of both innate and adaptive immunity.51, 52, 53
DC-mediated anti-tumor immunity of GM-CSF
Antigen presenting cells have an important role in the generation of protective immune responses to tumor-specific antigens. Due to their high constitutive levels of MHC and co-stimulatory molecules, DCs are the most potent antigen presenting cells and have a critical role in the host immune system.54 Marked numerical increases in DCs were detected in the thymus and spleen of mice injected with recombinant GM-CSF or transgenic mice that overexpress GM-CSF,55, 56 suggesting that GM-CSF can stimulate the in vivo expansion of DCs. In this context, GM-CSF efficiently stimulates higher levels of protective anti-tumor immunity via DC activation and accumulation.48 The increased anti-tumor immunity of GM-CSF-producing cells may be related to the ability to mature and recruit DCs,57 which are able to phagocytose apoptotic/necrotic tumor cells and display several co-stimulatory factors..58 In addition to their antigen-specific responses, several studies have identified specific DC functions for the induction of antitumor immunity of tumor vaccines.59 For example, Mach et al showed that GM-CSF-secreting tumor cells stimulated potent antitumor immunities by enhancing the expression levels of B7-1 and CD1d on DCs.48
Phase I clinical trial of GM-CSF-secreting tumor vaccines
The use of recombinant GM-CSF as an immune adjuvant to stimulate humoral or cellular immune responses to tumor antigens improves the survival of patients with various types of cancer. The activation of GM-CSF receptors promotes the survival, growth and differentiation of many different immune cell types, including neutrophils, macrophages and various T cells, in addition to the direct stimulatory effect on multiple immune functions. Obviously, these immunological properties make GM-CSF a potent immune adjuvant in cancer immunotherapy. Indeed, the subcutaneous injection of GM-CSF-producing cancer cells activated an intense local inflammatory response that stimulated DCs, macrophages and granulocytes.60, 61 The stimulation of these immune cells indicates that GM-CSF may enhance tumor-specific antigen presentation, thereby leading to improved anti-tumor activities by activating the immune system. To explore the effectiveness of the paracrine activity of the GM-CSF protein as a potent antitumor immune effector, Soiffer et al.62 conducted a phase I clinical trial investigating the biologic activity of engineered GM-CSF-producing autologous cancer cells in patients with metastatic melanoma. These autologous GM-CSF-secreting cells stimulated potent antitumor immunity and subsequently induced extensive tumor destruction (at least 80%) in 11 of the 16 patients with metastatic melanoma by recruiting CD4+ and CD8+ T cells into metastatic lesions.62 Consistent with these findings, Salgia et al.63 also conducted a phase I clinical trial, which revealed that metastatic lesions resected after vaccination with irradiated GM-CSF-secreting cells showed T lymphocyte and plasma cell infiltrates with tumor necrosis in three of the six patients with metastatic non-small-cell lung cancer. At a minimum of 36 months follow-up analysis, 10 of the 35 patients (29%) with metastatic melanoma were alive after vaccination, with a minimum follow-up of 36 months; further, 4 of these patients had no evidence of disease.64 In addition, Simons et al.65 demonstrated in their phase I clinical trial that included patients with immunocompetent prostate cancer that these GM-CSF-secreting tumor cells activated new T-cell and B-cell immune responses against prostate cancer antigens and the infiltration of effector cells consisting of CD45RO+ T cells. These results suggest that GM-CSF-secreting cells can create an advantageous environment for tumor antigen presentation.
Immune-independent effect of GM-CSF on multiple cancer types
Stimulatory effects on tumor progression
Interestingly, GM-CSF has also been described as a tumor-stimulating factor that acts in various cancer models in an autocrine or paracrine manner. Constitutive GM-CSF secretion has been found, frequently together with the GM-CSF receptor, in a variety of tumor models, including small-cell lung carcinomas,66 meningiomas,67 skin carcinoma,68, 69, 70 gliomas71 and head and neck squamous cell carcinomas (HNSCC).72 In various experiments, GM-CSF stimulated cancer cell proliferation and/or migration in vitro or in vivo in an immune-independent manner in multiple cancer types, including skin carcinoma,68, 70 gliomas,71 HNSCC14 and lung cancer cells.73 In summary, many previous studies have shown a tumor-promoting effect of GM-CSF in different cancer types and have raised exciting questions about the mechanisms of GM-CSF-driven cancer progression and metastasis.
Bladder cancer
When they are initially diagnosed, 70% of all bladder cancers are superficial (noninvasive), but most of them (60–70%) have a propensity to transform into invasive tumors following initial transurethral resection of bladder cancer. In ~15–25% of patients, bladder cancers recur and progress to invasive, high-risk tumors.74 Unexplained leukocytosis associated with bladder carcinoma has been described and is linked to poor prognosis.75 In some cases, this leukocytosis has been attributed to the inappropriate production and secretion of GM-CSF from bladder cancer cells.10 Some patients with urothelial carcinomas have been found to express GM-CSF receptors concomitantly, thus resulting in an autocrine and/or paracrine stimulation of growth, which may explain why some of these carcinomas behave aggressively.76 Recent microarray analysis revealed significant increases in the levels of GM-CSF and in the alpha-subunits of the GM-CSF receptor in bladder cancer patient samples compared with normal tissues.77
Brain cancer
Glioblastoma is a type of aggressive brain tumor that grows rapidly from glial cells and results in a very low 5-year survival rate for patients.78 Elevated expression levels of GM-CSF and its receptor have been reported in surgical specimens of malignant glioblastoma;79, 80 high tumoral levels of GM-CSF and its receptor are significantly correlated with a poor prognosis.9, 81, 82 In glioblastoma, the GM-CSF and its receptor promote tumor progression, possibly by upregulating anti-apoptotic and pro-angiogenic signals via the activation of the STAT3 signaling pathway or by increasing the expression of vascular endothelial growth factor and its receptor.83, 84 In the tumor environment, tumor cells and tumor-associated microglial cells, but not mesenchymal stem cells, are now known to secrete GM-CSF.38, 85, 86 Decreased GM-CSF levels significantly suppressed cancer cell growth and metastasis, suggesting a stimulatory effect of GM-CSF on glioblastoma progression.85
Colorectal cancer
Chronic inflammation is known to have an important regulatory role in the development of colorectal cancer in a variety of current experimental models.87, 88 Indeed, chronic inflammation, as has been commonly observed in various types of inflammatory bowel disorders, is known to be associated with an elevated incidence of colorectal cancer.87, 88 GM-CSF is known to be involved in regulating macrophage polarization.89 Interestingly, elevated levels of the soluble inflammatory cytokine GM-CSF in serum have been recognized in some patients with colorectal cancers, which suggest that GM-CSF may be an independent prognostic factor.15, 90 Consistently, gene expression arrays show that ~70% of human and murine colorectal cancers exhibit a consistent production and secretion of GM-CSF.11
Head and neck cancer
The increased expression of GM-CSF, together with platelet-derived growth factor and vascular endothelial growth factor, is significantly correlated with invasion and poor prognosis in patients with HNSCC.72 Tomita et al.91 reported that GM-CSF stimulates HNSCC cell invasion and metastasis by upregulating MMP-2 and MMP-14 expression. These findings have led to a careful re-evaluation to determine whether adjuvant GM-CSF therapy can prevent or stimulate tumor progression in patients with different types of cancer; however, there is a need for further investigations of the potential adverse effects of recombinant human GM-CSF in these patients.
Lung cancer
A remarkable tumor-related leukocytosis sometimes accompanies malignant lung cancer in the absence of apparent infection.92 The aggressive tumor growth and poor prognosis in some cases may be closely linked to the leukemoid reaction in lung cancer patients.93 Frequently, this leukocytosis can be caused by an unregulated production of hematological growth factors, including CSFs, IL-1, IL-6 and tumor necrosis factor-α.94, 95 Interestingly, increased serum levels of GM-CSF are considered markers for adverse clinical outcomes, especially in patients with non-small-cell lung carcinomas.96 Consistently, GM-CSF expression positively correlates with the tumorigenicity and spontaneous metastatic capability of human97 carcinomas, and the enhanced invasive ability of human lung cancer cells can be accompanied by an increased expression of extracellular matrix-degrading enzymes.73 Therefore, because GM-CSF may profoundly influence tumor progression and metastasis, caution is warranted when using recombinant GM-CSF as an adjuvant therapy for patients with lung cancer.98
Inhibitory effects on tumor progression
The ectopic secretion of GM-CSF has been observed in many different cancer cell lines derived from solid tumors,99 but the immune-independent effects of tumor cell-derived GM-CSF and the potential mechanisms underlying its direct effects on tumor progression remain unknown or poorly defined. Although some studies have suggested that GM-CSF inhibits tumor growth and metastasis, a greater number of studies have demonstrated that GM-CSF exerts stimulatory effects on tumor progression. The contradictory results obtained by a number of authors have revealed that GM-CSF can exert either significant anti-proliferative effects100, 101 or anti-apoptotic effects,102, 103, 104 depending on the tumor type and stage of development.
Anti-proliferative effect of GM-CSF
By performing clonogenic assay and suspension culture, Yamashita et al.101 demonstrated that GM-CSF treatment suppressed the proliferation of human small-cell lung cancer cells by blocking cell cycle progression from G0/G1 to the S phase. These anti-tumor effects of GM-CSF were attenuated by the addition of GM-CSF-neutralizing antibody. Ruff et al.100 also revealed that GM-CSF exerts antitumor effects by inhibiting the proliferation of SCLCs, as determined by 3H-thymidine incorporation assay and soft agar colony-formation assay. Interestingly, Urdinguio et al.11 demonstrated that this immune-independent antitumor effect seems to depend on the ectopic expression of GM-CSF receptor subunits in human colorectal cancer. Tumor cells expressing GM-CSF and its receptor failed to grow in vivo when they were transplanted into immunocompetent mice.11 Consistently, high levels of expression of GM-CSF and its receptor are associated with improved 5-year survival rates in patients with colorectal cancers.11 These findings strongly support the anti-proliferative functions of GM-CSF as potential immune-independent tumor suppressors.
GM-CSF as a differentiation inducer
Unlike bulk tumor cells, a tumor subpopulation with stem cell-like properties contributes to tumor initiation, metastasis and therapeutic resistance in various types of cancer.105 Therefore, accelerating the terminal differentiation process can be considered as an alternative therapeutic option to eradicate this stem cell-like subpopulation by modulating the expression of various terminal differentiation regulators. In this context, Yamashita et al.101 demonstrated that recombinant GM-CSF treatment increased the percentage of cells with surface marker Mo1, which exerts a rapid cell differentiation of immature cells, thus suggesting that GM-CSF inhibits tumor progression by inducing differentiation of SCLCs. Consistent with these findings, Ruff et al.100 revealed that GM-CSF exerts remarkable antitumor activity against SCLCs by enhancing the expression levels of differentiation antigenic phenotypes such as Leu-M3, Leu-7 and HLA-DR.
Conclusion
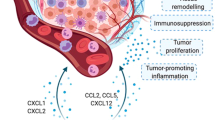
GM-CSF is secreted by many immune cell types, including macrophages, mast cells and T cells, mainly in response to immune activation and inflammatory cytokines, which in turn mediate immune responses. However, an increasing amount of evidence shows that in addition to the traditional immune modulating potential, GM-CSF is secreted by a number of non-immune cell types, including endothelial cells, keratinocytes and fibroblasts, following the appropriate stimuli. Interestingly, GM-CSF was recently described as an immune-independent tumor-promoting factor. GM-CSF stimulates tumor cell growth and/or migration in vitro and in vivo in multiple cancer types, including skin carcinoma, gliomas, HNSCCs and lung cancer cells. In contrast, some studies have suggested that GM-CSF has inhibitory effects on tumor progression. Therefore, the study of GM-CSF is one of the most interesting areas of cancer research, but further investigation is required for clinical applications. Although increased attention is now focused on the anti-tumor, immunostimulatory effects and immune-independent tumor-promoting effects of GM-CSF on tumor progression, the current knowledge about the immune-independent inhibitory effects of GM-CSF on tumor progression and the underlying mechanisms is still rudimentary. Therefore, more detailed knowledge about the mutual interactions between GM-CSF and tumor cells will undoubtedly lead to more efficient and successful clinical outcomes in the future. The schematic diagram summarizes the potential roles of GM-CSF in tumor progression (Figure 1).
Schematic diagram summarizing the potential roles of GM-CSF in tumor progression. GM-CSF exerts its function mainly by stimulating dendritic cell (DC) maturation and monocyte/macrophage activity as an immunomodulator. In addition, GM-CSF promotes immune-independent tumor progression by supporting tumor microenvironments and stimulating tumor growth and metastasis. GM-CSF also has inhibitory effects on tumor growth and metastasis.
References
van de Laar L, Coffer PJ, Woltman AM . Regulation of dendritic cell development by GM-CSF: molecular control and implications for immune homeostasis and therapy. Blood 2012; 119: 3383–3393.
Ohashi K, Kobayashi G, Fang S, Zhu X, Antonia SJ, Krieg AM et al. Surgical excision combined with autologous whole tumor cell vaccination is an effective therapy for murine neuroblastoma. J Pediatr Surg 2006; 41: 1361–1368.
Barreda DR, Hanington PC, Belosevic M . Regulation of myeloid development and function by colony stimulating factors. Dev Comp Immunol 2004; 28: 509–554.
Metcalf D . The colony-stimulating factors and cancer. Nat Rev Cancer 2010; 10: 425–434.
Gutschalk CM, Yanamandra AK, Linde N, Meides A, Depner S, Mueller MM . GM-CSF enhances tumor invasion by elevated MMP-2, -9, and -26 expression. Cancer Med 2013; 2: 117–129.
Mascia F, Cataisson C, Lee TC, Threadgill D, Mariani V, Amerio P et al. EGFR regulates the expression of keratinocyte-derived granulocyte/macrophage colony-stimulating factor in vitro and in vivo. J Invest Dermatol 2010; 130: 682–693.
Rho CR, Park MY, Kang S . Effects of granulocyte-macrophage colony-stimulating (GM-CSF) factor on corneal epithelial cells in corneal wound healing model. PLoS ONE 2015; 10: e0138020.
Lammel V, Stoeckle C, Padberg B, Zweifel R, Kienle DL, Reinhart WH et al. Hypereosinophilia driven by GM-CSF in large-cell carcinoma of the lung. Lung Cancer 2012; 76: 493–495.
Revoltella RP, Menicagli M, Campani D . Granulocyte-macrophage colony-stimulating factor as an autocrine survival-growth factor in human gliomas. Cytokine 2012; 57: 347–359.
Perez FA, Fligner CL, Yu EY . Rapid clinical deterioration and leukemoid reaction after treatment of urothelial carcinoma of the bladder: possible effect of granulocyte colony-stimulating factor. J Clin Oncol 2009; 27: e215–e217.
Urdinguio RG, Fernandez AF, Moncada-Pazos A, Huidobro C, Rodriguez RM, Ferrero C et al. Immune-dependent and independent antitumor activity of GM-CSF aberrantly expressed by mouse and human colorectal tumors. Cancer Res 2013; 73: 395–405.
Taghipour Fard Ardekani M, Malekzadeh M, Hosseini SV, Bordbar E, Doroudchi M, Ghaderi A . Evaluation of pre-treatment serum levels of IL-7 and GM-CSF in colorectal cancer patients. Int J Mol Cell Med 2014; 3: 27–34.
Montag M, Dyckhoff G, Lohr J, Helmke BM, Herrmann E, Plinkert PK et al. Angiogenic growth factors in tissue homogenates of HNSCC: expression pattern, prognostic relevance, and interrelationships. Cancer Sci 2009; 100: 1210–1218.
Gutschalk CM, Herold-Mende CC, Fusenig NE, Mueller MM . Granulocyte colony-stimulating factor and granulocyte-macrophage colony-stimulating factor promote malignant growth of cells from head and neck squamous cell carcinomas in vivo. Cancer Res 2006; 66: 8026–8036.
Nebiker CA, Han J, Eppenberger-Castori S, Iezzi G, Hirt C, Amicarella F et al. GM-CSF production by tumor cells is associated with improved survival in colorectal cancer. Clin Cancer Res 2014; 20: 3094–3106.
Rigo A, Gottardi M, Zamo A, Mauri P, Bonifacio M, Krampera M et al. Macrophages may promote cancer growth via a GM-CSF/HB-EGF paracrine loop that is enhanced by CXCL12. Mol Cancer 2010; 9: 273.
Shiomi A, Usui T . Pivotal roles of GM-CSF in autoimmunity and inflammation. Mediators Inflamm 2015; 2015: 568543.
Shi Y, Liu CH, Roberts AI, Das J, Xu G, Ren G et al. Granulocyte-macrophage colony-stimulating factor (GM-CSF) and T-cell responses: what we do and don't know. Cell Res 2006; 16: 126–133.
Ponomarev ED, Shriver LP, Maresz K, Pedras-Vasconcelos J, Verthelyi D, Dittel BN . GM-CSF production by autoreactive T cells is required for the activation of microglial cells and the onset of experimental autoimmune encephalomyelitis. J Immunol 2007; 178: 39–48.
Li R, Rezk A, Miyazaki Y, Hilgenberg E, Touil H, Shen P et al. Proinflammatory GM-CSF-producing B cells in multiple sclerosis and B cell depletion therapy. Sci Transl Med 2015; 7: 310ra166.
Tisato V, Secchiero P, Rimondi E, Gianesini S, Menegatti E, Casciano F et al. GM-CSF exhibits anti-inflammatory activity on endothelial cells derived from chronic venous disease patients. Mediators Inflamm 2013; 2013: 561689.
Barrientos S, Stojadinovic O, Golinko MS, Brem H, Tomic-Canic M . Growth factors and cytokines in wound healing. Wound Repair Regen 2008; 16: 585–601.
Szabowski A, Maas-Szabowski N, Andrecht S, Kolbus A, Schorpp-Kistner M, Fusenig NE et al. c-Jun and JunB antagonistically control cytokine-regulated mesenchymal-epidermal interaction in skin. Cell 2000; 103: 745–755.
Kingston D, Schmid MA, Onai N, Obata-Onai A, Baumjohann D, Manz MG . The concerted action of GM-CSF and Flt3-ligand on in vivo dendritic cell homeostasis. Blood 2009; 114: 835–843.
Murphy JM, Young IG . IL-3, IL-5, and GM-CSF signaling: crystal structure of the human beta-common receptor. Vitam Horm 2006; 74: 1–30.
McClure BJ, Hercus TR, Cambareri BA, Woodcock JM, Bagley CJ, Howlett GJ et al. Molecular assembly of the ternary granulocyte-macrophage colony-stimulating factor receptor complex. Blood 2003; 101: 1308–1315.
Geijsen N, Koenderman L, Coffer PJ . Specificity in cytokine signal transduction: lessons learned from the IL-3/IL-5/GM-CSF receptor family. Cytokine Growth Factor Rev 2001; 12: 19–25.
Onetto-Pothier N, Aumont N, Haman A, Bigras C, Wong GG, Clark SC et al. Characterization of granulocyte-macrophage colony-stimulating factor receptor on the blast cells of acute myeloblastic leukemia. Blood 1990; 75: 59–66.
Hansen G, Hercus TR, McClure BJ, Stomski FC, Dottore M, Powell J et al. The structure of the GM-CSF receptor complex reveals a distinct mode of cytokine receptor activation. Cell 2008; 134: 496–507.
Suh HS, Kim MO, Lee SC . Inhibition of granulocyte-macrophage colony-stimulating factor signaling and microglial proliferation by anti-CD45RO: role of Hck tyrosine kinase and phosphatidylinositol 3-kinase/Akt. J Immunol 2005; 174: 2712–2719.
Notarangelo LD, Pessach I . Out of breath: GM-CSFRalpha mutations disrupt surfactant homeostasis. J Exp Med 2008; 205: 2693–2697.
Lukens JR, Barr MJ, Chaplin DD, Chi H, Kanneganti TD . Inflammasome-derived IL-1beta regulates the production of GM-CSF by CD4(+) T cells and gammadelta T cells. J Immunol 2012; 188: 3107–3115.
Sonderegger I, Iezzi G, Maier R, Schmitz N, Kurrer M, Kopf M . GM-CSF mediates autoimmunity by enhancing IL-6-dependent Th17 cell development and survival. J Exp Med 2008; 205: 2281–2294.
Atanga E, Dolder S, Dauwalder T, Wetterwald A, Hofstetter W . TNFalpha inhibits the development of osteoclasts through osteoblast-derived GM-CSF. Bone 2011; 49: 1090–1100.
Miyashita H, Katayama N, Fujieda A, Shibasaki T, Yamamura K, Sugimoto Y et al. IL-4 and IL-10 synergistically inhibit survival of human blood monocytes supported by GM-CSF. Int J Oncol 2005; 26: 731–735.
Dang PM, Elbim C, Marie JC, Chiandotto M, Gougerot-Pocidalo MA, El-Benna J . Anti-inflammatory effect of interleukin-10 on human neutrophil respiratory burst involves inhibition of GM-CSF-induced p47PHOX phosphorylation through a decrease in ERK1/2 activity. FASEB J 2006; 20: 1504–1506.
Kasper S, Kindler T, Sonnenschein S, Breitenbuecher F, Bohmer FD, Huber C et al. Cross-inhibition of interferon-induced signals by GM-CSF through a block in Stat1 activation. J Interferon Cytokine Res 2007; 27: 947–959.
Gabrusiewicz K, Ellert-Miklaszewska A, Lipko M, Sielska M, Frankowska M, Kaminska B . Characteristics of the alternative phenotype of microglia/macrophages and its modulation in experimental gliomas. PLoS ONE 2011; 6: e23902.
Nehme A, Edelman J . Dexamethasone inhibits high glucose-, TNF-alpha-, and IL-1beta-induced secretion of inflammatory and angiogenic mediators from retinal microvascular pericytes. Invest Ophthalmol Vis Sci 2008; 49: 2030–2038.
Pastore S, Fanales-Belasio E, Albanesi C, Chinni LM, Giannetti A, Girolomoni G . Granulocyte macrophage colony-stimulating factor is overproduced by keratinocytes in atopic dermatitis. Implications for sustained dendritic cell activation in the skin. J Clin Invest 1997; 99: 3009–3017.
Reynolds G, Gibbon JR, Pratt AG, Wood MJ, Coady D, Raftery G et al. Synovial CD4+ T-cell-derived GM-CSF supports the differentiation of an inflammatory dendritic cell population in rheumatoid arthritis. Ann Rheum Dis 2016; 75: 899–907.
Noster R, Riedel R, Mashreghi MF, Radbruch H, Harms L, Haftmann C et al. IL-17 and GM-CSF expression are antagonistically regulated by human T helper cells. Sci Transl Med 2014; 6 241ra80.
Zinkernagel RM . On natural and artificial vaccinations. Annu Rev Immunol 2003; 21: 515–546.
Vesely MD, Kershaw MH, Schreiber RD, Smyth MJ . Natural innate and adaptive immunity to cancer. Annu Rev Immunol 2011; 29: 235–271.
Zitvogel L, Tesniere A, Kroemer G . Cancer despite immunosurveillance: immunoselection and immunosubversion. Nat Rev Immunol 2006; 6: 715–727.
Dougan M, Dranoff G . Immune therapy for cancer. Annu Rev Immunol 2009; 27: 83–117.
Chu Y, Wang LX, Yang G, Ross HJ, Urba WJ, Prell R et al. Efficacy of GM-CSF-producing tumor vaccine after docetaxel chemotherapy in mice bearing established Lewis lung carcinoma. J Immunother 2006; 29: 367–380.
Mach N, Gillessen S, Wilson SB, Sheehan C, Mihm M, Dranoff G . Differences in dendritic cells stimulated in vivo by tumors engineered to secrete granulocyte-macrophage colony-stimulating factor or Flt3-ligand. Cancer Res 2000; 60: 3239–3246.
Qin Z, Noffz G, Mohaupt M, Blankenstein T . Interleukin-10 prevents dendritic cell accumulation and vaccination with granulocyte-macrophage colony-stimulating factor gene-modified tumor cells. J Immunol 1997; 159: 770–776.
Gillessen S, Naumov YN, Nieuwenhuis EE, Exley MA, Lee FS, Mach N et al. CD1d-restricted T cells regulate dendritic cell function and antitumor immunity in a granulocyte-macrophage colony-stimulating factor-dependent fashion. Proc Natl Acad Sci USA 2003; 100: 8874–8879.
Spitler LE, Grossbard ML, Ernstoff MS, Silver G, Jacobs M, Hayes FA et al. Adjuvant therapy of stage III and IV malignant melanoma using granulocyte-macrophage colony-stimulating factor. J Clin Oncol 2000; 18: 1614–1621.
Rini BI, Weinberg V, Bok R, Small EJ . Prostate-specific antigen kinetics as a measure of the biologic effect of granulocyte-macrophage colony-stimulating factor in patients with serologic progression of prostate cancer. J Clin Oncol 2003; 21: 99–105.
Anderson PM, Markovic SN, Sloan JA, Clawson ML, Wylam M, Arndt CA et al. Aerosol granulocyte macrophage-colony stimulating factor: a low toxicity, lung-specific biological therapy in patients with lung metastases. Clin Cancer Res 1999; 5: 2316–2323.
Yoshimura S, Bondeson J, Foxwell BM, Brennan FM, Feldmann M . Effective antigen presentation by dendritic cells is NF-kappaB dependent: coordinate regulation of MHC, co-stimulatory molecules and cytokines. Int Immunol 2001; 13: 675–683.
Daro E, Pulendran B, Brasel K, Teepe M, Pettit D, Lynch DH et al. Polyethylene glycol-modified GM-CSF expands CD11b(high)CD11c(high) but notCD11b(low)CD11c(high) murine dendritic cells in vivo: a comparative analysis with Flt3 ligand. J Immunol 2000; 165: 49–58.
Vremec D, Lieschke GJ, Dunn AR, Robb L, Metcalf D, Shortman K . The influence of granulocyte/macrophage colony-stimulating factor on dendritic cell levels in mouse lymphoid organs. Eur J Immunol 1997; 27: 40–44.
Zarei S, Schwenter F, Luy P, Aurrand-Lions M, Morel P, Kopf M et al. Role of GM-CSF signaling in cell-based tumor immunization. Blood 2009; 113: 6658–6668.
Hanahan D, Weinberg RA . The hallmarks of cancer. Cell 2000; 100: 57–70.
Banchereau J, Palucka AK . Dendritic cells as therapeutic vaccines against cancer. Nat Rev Immunol 2005; 5: 296–306.
Mach N, Dranoff G . Cytokine-secreting tumor cell vaccines. Curr Opin Immunol 2000; 12: 571–575.
Dranoff G, Jaffee E, Lazenby A, Golumbek P, Levitsky H, Brose K et al. Vaccination with irradiated tumor cells engineered to secrete murine granulocyte-macrophage colony-stimulating factor stimulates potent, specific, and long-lasting anti-tumor immunity. Proc Natl Acad Sci USA 1993; 90: 3539–3543.
Soiffer R, Lynch T, Mihm M, Jung K, Rhuda C, Schmollinger JC et al. Vaccination with irradiated autologous melanoma cells engineered to secrete human granulocyte-macrophage colony-stimulating factor generates potent antitumor immunity in patients with metastatic melanoma. Proc Natl Acad Sci USA 1998; 95: 13141–13146.
Salgia R, Lynch T, Skarin A, Lucca J, Lynch C, Jung K et al. Vaccination with irradiated autologous tumor cells engineered to secrete granulocyte-macrophage colony-stimulating factor augments antitumor immunity in some patients with metastatic non-small-cell lung carcinoma. J Clin Oncol 2003; 21: 624–630.
Soiffer R, Hodi FS, Haluska F, Jung K, Gillessen S, Singer S et al. Vaccination with irradiated, autologous melanoma cells engineered to secrete granulocyte-macrophage colony-stimulating factor by adenoviral-mediated gene transfer augments antitumor immunity in patients with metastatic melanoma. J Clin Oncol 2003; 21: 3343–3350.
Simons JW, Mikhak B, Chang JF, DeMarzo AM, Carducci MA, Lim M et al. Induction of immunity to prostate cancer antigens: results of a clinical trial of vaccination with irradiated autologous prostate tumor cells engineered to secrete granulocyte-macrophage colony-stimulating factor using ex vivo gene transfer. Cancer Res 1999; 59: 5160–5168.
Baldwin GC, Gasson JC, Kaufman SE, Quan SG, Williams RE, Avalos BR et al. Nonhematopoietic tumor cells express functional GM-CSF receptors. Blood 1989; 73: 1033–1037.
Braun B, Lange M, Oeckler R, Mueller MM . Expression of G-CSF and GM-CSF in human meningiomas correlates with increased tumor proliferation and vascularization. J Neurooncol 2004; 68: 131–140.
Mueller MM, Fusenig NE . Constitutive expression of G-CSF and GM-CSF in human skin carcinoma cells with functional consequence for tumor progression. Int J Cancer 1999; 83: 780–789.
Mueller MM, Peter W, Mappes M, Huelsen A, Steinbauer H, Boukamp P et al. Tumor progression of skin carcinoma cells in vivo promoted by clonal selection, mutagenesis, and autocrine growth regulation by granulocyte colony-stimulating factor and granulocyte-macrophage colony-stimulating factor. Am J Pathol 2001; 159: 1567–1579.
Obermueller E, Vosseler S, Fusenig NE, Mueller MM . Cooperative autocrine and paracrine functions of granulocyte colony-stimulating factor and granulocyte-macrophage colony-stimulating factor in the progression of skin carcinoma cells. Cancer Res 2004; 64: 7801–7812.
Mueller MM, Herold-Mende CC, Riede D, Lange M, Steiner HH, Fusenig NE . Autocrine growth regulation by granulocyte colony-stimulating factor and granulocyte macrophage colony-stimulating factor in human gliomas with tumor progression. Am J Pathol 1999; 155: 1557–1567.
Ninck S, Reisser C, Dyckhoff G, Helmke B, Bauer H, Herold-Mende C . Expression profiles of angiogenic growth factors in squamous cell carcinomas of the head and neck. Int J Cancer 2003; 106: 34–44.
Pei XH, Nakanishi Y, Takayama K, Bai F, Hara N . Granulocyte, granulocyte-macrophage, and macrophage colony-stimulating factors can stimulate the invasive capacity of human lung cancer cells. Br J Cancer 1999; 79: 40–46.
Hu Z, Tan W, Zhang L, Liang Z, Xu C, Su H et al. A novel immunotherapy for superficial bladder cancer by intravesical immobilization of GM-CSF. J Cell Mol Med 2010; 14: 1836–1844.
Izard JP, Gore JL, Mostaghel EA, Wright JL, Yu EY . Persistent, unexplained leukocytosis is a paraneoplastic syndrome associated with a poor prognosis in patients with urothelial carcinoma. Clin Genitourin Cancer 2015; 13: e253–e258.
Wetzler M, Estrov Z, Talpaz M, Markowitz A, Gutterman JU, Kurzrock R . Granulocyte-macrophage colony-stimulating factor as a cause of paraneoplastic leukaemoid reaction in advanced transitional cell carcinoma. J Intern Med 1993; 234: 417–420.
Sanchez-Carbayo M, Socci ND, Lozano J, Saint F, Cordon-Cardo C . Defining molecular profiles of poor outcome in patients with invasive bladder cancer using oligonucleotide microarrays. J Clin Oncol 2006; 24: 778–789.
Walid MS . Prognostic factors for long-term survival after glioblastoma. Perm J 2008; 12: 45–48.
Kohanbash G, McKaveney K, Sakaki M, Ueda R, Mintz AH, Amankulor N et al. GM-CSF promotes the immunosuppressive activity of glioma-infiltrating myeloid cells through interleukin-4 receptor-alpha. Cancer Res 2013; 73: 6413–6423.
Albulescu R, Codrici E, Popescu ID, Mihai S, Necula LG, Petrescu D et al. Cytokine patterns in brain tumour progression. Mediators Inflamm 2013; 2013: 979748.
Wang J, Yao L, Zhao S, Zhang X, Yin J, Zhang Y et al. Granulocyte-colony stimulating factor promotes proliferation, migration and invasion in glioma cells. Cancer Biol Ther 2012; 13: 389–400.
Karcher S, Steiner HH, Ahmadi R, Zoubaa S, Vasvari G, Bauer H et al. Different angiogenic phenotypes in primary and secondary glioblastomas. Int J Cancer 2006; 118: 2182–2189.
Jung KH, Chu K, Lee ST, Kim SJ, Sinn DI, Kim SU et al. Granulocyte colony-stimulating factor stimulates neurogenesis via vascular endothelial growth factor with STAT activation. Brain Res 2006; 1073-1074: 190–201.
Ohki Y, Heissig B, Sato Y, Akiyama H, Zhu Z, Hicklin DJ et al. Granulocyte colony-stimulating factor promotes neovascularization by releasing vascular endothelial growth factor from neutrophils. FASEB J 2005; 19: 2005–2007.
Curran CS, Evans MD, Bertics PJ . GM-CSF production by glioblastoma cells has a functional role in eosinophil survival, activation, and growth factor production for enhanced tumor cell proliferation. J Immunol 2011; 187: 1254–1263.
Kucerova L, Matuskova M, Hlubinova K, Altanerova V, Altaner C . Tumor cell behaviour modulation by mesenchymal stromal cells. Mol Cancer 2010; 9: 129.
Terzic J, Grivennikov S, Karin E, Karin M . Inflammation and colon cancer. Gastroenterology 2010; 138: 2101–14 e5.
Danese S, Malesci A, Vetrano S . Colitis-associated cancer: the dark side of inflammatory bowel disease. Gut 2011; 60: 1609–1610.
Verreck FA, de Boer T, Langenberg DM, Hoeve MA, Kramer M, Vaisberg E et al. Human IL-23-producing type 1 macrophages promote but IL-10-producing type 2 macrophages subvert immunity to (myco)bacteria. Proc Natl Acad Sci USA 2004; 101: 4560–4565.
Demirci U, Coskun U, Sancak B, Ozturk B, Bahar B, Benekli M et al. Serum granulocyte macrophage-colony stimulating factor: a tumor marker in colorectal carcinoma? Asian Pac J Cancer Prev 2009; 10: 1021–1024.
Tomita T, Fujii M, Tokumaru Y, Imanishi Y, Kanke M, Yamashita T et al. Granulocyte-macrophage colony-stimulating factor upregulates matrix metalloproteinase-2 (MMP-2) and membrane type-1 MMP (MT1-MMP) in human head and neck cancer cells. Cancer Lett 2000; 156: 83–91.
Shafiq M, Venkateshiah S . Hematologic abnormalities associated with lung carcinoma. Respir Care 2011; 56: 523–526.
Shalom G, Sion-Vardy N, Dudnik J, Ariad S . Leukemoid reaction in lung cancer patients. Isr Med Assoc J 2010; 12: 255–256.
Hauber HP . Paraneoplastic syndromes in lung cancer. Pneumologie 2011; 65: 347–358.
Bahar B, Acedil Ayc Iota B, Coskun U, Buyukberber S, Benekli M, Yildiz R . Granulocyte colony stimulating factor (G-CSF) and macrophage colony stimulating factor (M-CSF) as potential tumor markers in non small cell lung cancer diagnosis. Asian Pac J Cancer Prev 2010; 11: 709–712.
Mroczko B, Szmitkowski M . Hematopoietic cytokines as tumor markers. Clin Chem Lab Med 2004; 42: 1347–1354.
Levina V, Marrangoni AM, DeMarco R, Gorelik E, Lokshin AE . Drug-selected human lung cancer stem cells: cytokine network, tumorigenic and metastatic properties. PLoS ONE 2008; 3: e3077.
Kowanetz M, Wu X, Lee J, Tan M, Hagenbeek T, Qu X et al. Granulocyte-colony stimulating factor promotes lung metastasis through mobilization of Ly6G+Ly6C+ granulocytes. Proc Natl Acad Sci USA 2010; 107: 21248–21255.
Aliper AM, Frieden-Korovkina VP, Buzdin A, Roumiantsev SA, Zhavoronkov A . A role for G-CSF and GM-CSF in nonmyeloid cancers. Cancer Med 2014; 3: 737–746.
Ruff MR, Farrar WL, Pert CB . Interferon gamma and granulocyte/macrophage colony-stimulating factor inhibit growth and induce antigens characteristic of myeloid differentiation in small-cell lung cancer cell lines. Proc Natl Acad Sci USA 1986; 83: 6613–6617.
Yamashita Y, Nara N, Aoki N . Antiproliferative and differentiative effect of granulocyte-macrophage colony-stimulating factor on a variant human small cell lung cancer cell line. Cancer Res 1989; 49: 5334–5338.
Berdel WE, Danhauser-Riedl S, Steinhauser G, Winton EF . Various human hematopoietic growth factors (interleukin-3, GM-CSF, G-CSF) stimulate clonal growth of nonhematopoietic tumor cells. Blood 1989; 73: 80–83.
Dedhar S, Gaboury L, Galloway P, Eaves C . Human granulocyte-macrophage colony-stimulating factor is a growth factor active on a variety of cell types of nonhemopoietic origin. Proc Natl Acad Sci USA 1988; 85: 9253–9257.
Onetto N . Extra hematopoietic effect of colony-stimulating factors. Blood 1989; 74: 1446–1447.
Campos B, Wan F, Farhadi M, Ernst A, Zeppernick F, Tagscherer KE et al. Differentiation therapy exerts antitumor effects on stem-like glioma cells. Clin Cancer Res 2010; 16: 2715–2728.
Acknowledgements
This work was also supported by a grant (15182MFDS455) from Ministry of Food and Drug Safety.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no conflict of interest.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Hong, IS. Stimulatory versus suppressive effects of GM-CSF on tumor progression in multiple cancer types. Exp Mol Med 48, e242 (2016). https://doi.org/10.1038/emm.2016.64
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/emm.2016.64
This article is cited by
-
Mechanisms of chemotherapeutic resistance and the application of targeted nanoparticles for enhanced chemotherapy in colorectal cancer
Journal of Nanobiotechnology (2022)
-
Th17 cells and their related cytokines: vital players in progression of malignant pleural effusion
Cellular and Molecular Life Sciences (2022)
-
STAT5 is activated in macrophages by breast cancer cell-derived factors and regulates macrophage function in the tumor microenvironment
Breast Cancer Research (2021)
-
Roles of acyl-CoA synthetase long-chain family member 5 and colony stimulating factor 2 in inhibition of palmitic or stearic acids in lung cancer cell proliferation and metabolism
Cell Biology and Toxicology (2021)
-
Skeletal muscle healing by M1-like macrophages produced by transient expression of exogenous GM-CSF
Stem Cell Research & Therapy (2020)