Abstract
Tumor necrosis factor-α (TNF-α) and inflammatory cytokines released from activated macrophages in response to particulate debris greatly impact periprosthetic bone loss and consequent implant failure. In the present study, we found that a major polyphenolic component of green tea, (-)-epigallocatechin gallate (EGCG), inhibited Ti particle-induced TNF-α release in macrophages in vitro and calvarial osteolysis in vivo. The Ti stimulation of macrophages released TNF-α in a dose- and time-dependent manner, and EGCG substantially suppressed Ti particle-induced TNF-α release. Analysis of signaling pathway showed that EGCG inhibited the Ti-induced c-Jun N-terminus kinase (JNK) activation and inhibitory κB (IκB) degradation, and consequently the Ti-induced transcriptional activation of AP-1 and NF-κB. In a mouse calvarial osteolysis model, EGCG inhibited Ti particle-induced osteolysis in vivo by suppressing TNF-α expression and osteoclast formation. Therefore, EGCG may be a potential candidate compound for osteolysis prevention and treatment as well as aseptic loosening after total replacement arthroplasty.
Similar content being viewed by others
Introduction
Periprosthetic osteolysis and subsequent aseptic loosening by wear debris of orthopaedic implants are the most common causes of total joint arthroplasty (TJA) failure. Particulate debris stimulates macrophages, fibroblasts, giant cells, and osteoclasts in the granulomatous tissues. The debris also induces the production of inflammatory cytokines and peptide factors such as TNF-α, interleukin (IL)-1β, IL-6, IL-8, IL-10, IL-12p40, IL-11, macrophage chemoattractant protein-1, and granulocytemonocyte colony-stimulating factor, which are involved in osteolysis and bone loosening (Kim et al., 1993; Sabokbar and Rushton 1995; Al-Saffar et al., 1996; Xu et al., 1998; Merkel et al., 1999; Kaufman et al., 2008). The particle-induced release of TNF-α and IL-6 in macrophages depends on tyrosine and serine/threonine kinase activity leading to the activation of nuclear factor-κB (NF-κB) and NF-interleukin-6 (NF-IL-6), which are critical for the upregulation of proinflammatory cytokines (Nakashima et al., 1999). In addition, TNF-α augments the osteoblast expression of receptor activation of NF-κB ligand (RANKL) and macrophage-colony stimulating factor (M-CSF), which are essential factors involved in the expansion, commitment, and differentiation of osteoclast precursors into mature osteoclasts (Kimble et al., 1996; Hofbauer et al., 1999; Crotti et al., 2004; Liu et al., 2009). Anti-TNF-α antibody significantly inhibits bone resorption by supernatants from particle-stimulated macrophages (Algan et al., 1996). TNF receptor (TNFR) gene knockout mice show no evidence of osteolysis following expose to polymethylmethacrylate particles (Merkel et al., 1999). Therefore, controlling the synthesis of TNF-α and other inflammatory cytokines in the periprosthetic environment may be a potential target to prevent or reduce wear particle-induced osteolysis (Schwarz et al., 2000).
Much effort has been made to prevent or treat osteolysis through the improvement of prosthesis materials (Katti, 2004). Pharmacological studies have been performed to evaluate the effectiveness of drugs on osteolysis. In a canine model, bisphosphonates such as alendronate dramatically reduce osteolysis without affecting the inflammatory response (Shanbhag et al., 1994a, 1994b, 1997). Zoledronate and simvastatin decrease ultrahigh molecular weight polyethylene (UHMWPE) particle-induced osteolysis in a murine calvarial model (von Knoch et al., 2005a). Erythromycin, a 14-membered lactone ring macrolide antibiotic, also dramatically reduces UHMWPE particle-induced tissue inflammation in vivo and inhibits osteoclast formation and function in vitro (Ren et al., 2006). Exogenously-administered osteoprotegerin (OPG), a decoy receptor of RANKL, can prevent and treat polyethylene particle-induced osteolysis in a murine calvarial model (von Knoch et al., 2005b). Recent reports demonstrate that VIVD (a peptide inhibitor of a nuclear factor of activated T cells) and AM630 (a cannabinoid receptor-selective antagonist) suppress Ti particle-induced inflammatory cytokines and osteoclast formation in vitro (Liu et al., 2009). These studies suggest that drugs that inhibit osteoclast differentiation and inflammatory cytokine synthesis may have positive effects on particulate-induced osteolysis.
Many foods and beverages have received considerable attention because of their beneficial effects on human health. In particular, epidemiologic and experimental observations have confirmed a close relationship between green tea consumption and the prevention of cancer development and cardiovascular disease (Yang and Wang, 1993). These effects largely have been attributed to the most prevalent polyphenol contained in green tea, (-)-epigallocatechin (EGCG). EGCG induces the apoptotic cell death of osteoclasts and inhibits osteoclast formation and lipopolysaccharide (LPS)-induced TNF-α release in macrophages (Yang et al., 1998; Nakagawa et al., 2002; Yun et al., 2004). However, it remains to be investigated whether EGCG can inhibit particulate-induced TNF-α release in macrophages and in vivo osteolysis. In this study, we investigated the effects of EGCG on Ti particle-induced TNF-α release and the underlying molecular mechanism in vitro. We further examined the effects of EGCG on Ti particle-induced osteolysis in a mouse calvarial model.
Results
Effect of EGCG on Ti particle-induced TNF-α release
To determine the effective concentration of Ti particles that induces TNF-α release in RAW264.7 and J774 cells, 3 different concentrations of Ti particles were treated to treat both cell lines and TNF-α release was measured by ELISA. TNF-α release was substantially induced by Ti particles in a dose-dependent manner (Supplemental Data Figure S1A). TNF-α release was dependent on the time of stimulation in RAW264.7 and J774 cells (Supplemental Data Figure S1B).
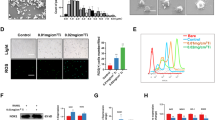
To evaluate the effect of EGCG on Ti particle-induced TNF-α release, RAW264.7 and J774 cells were treated with or without EGCG (50 or 100 µM) for 30 min prior to stimulation with 0.02% Ti for 24 h. EGCG substantially inhibited Ti particle-induced TNF-α release by 51.9% at 50 µM (308 ± 88.6 pg/ml) and by 80.5% at 100 µM (125.2 ± 3.3 pg/ml) compared to the control (640.8 ± 12.4 pg/ml) in RAW264.7 cells (Figure 1, upper panel). Similar results were obtained in J774 cells (Figure 1, lower panel). The results demonstrate that EGCG has a dose-dependent inhibitory effect on Ti particle-induced TNF-α release in macrophages.
Inhibitory effect of EGCG on Ti particle-induced TNF-α release. RAW264.7 and J774 cells were pretreated with or without the indicated amount of EGCG for 30 min and then stimulated with 0.02% Ti for 24 h. TNF-α release was analyzed by ELISA. Results are shown as the means ± SDs of 3 independent experiments. *P < 0.05.
Effect of EGCG on Ti particle-induced activation of JNK/AP-1 and NF-κB pathways
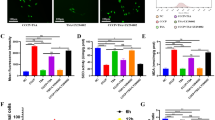
To define the molecular mechanism(s) by which EGCG inhibits Ti-induced TNF-α release, we first examined signaling pathways which are important in Ti particle-induced TNF-α release using pharmacological inhibitors of signaling molecules. Ti-induced TNF-α release in RAW264.7 and J774 cells was inhibited by MG132 and SP600125, but not by PD98059 or SB203580 (Supplemental Data Figure S2). These results imply that the NF-κB and JNK pathways are involved in Ti-induced TNF-α release in macrophages. To confirm these results, the activities of JNK were examined using antibodies specific for phosphorylated JNK. We also examined the protein level of IκBα, whose degradation releases and activates NF-κB. JNK phosphorylation was induced from 30 min after Ti stimulation, peaked at 1 h, and decreased thereafter. IκBα degradation by Ti particle stimulation was most prominent at 30 min (Figure 2A). Treatment with EGCG dramatically inhibited JNK activation (Figure 2B) and IκBα degradation in a dose-dependent manner (Figure 2C). These results suggest that EGCG inhibits signaling pathways leading to JNK and NF-κB activation.
Effect of Ti particles on the activation of signaling molecules and their inhibition by EGCG. (A) RAW264.7 cells (2 × 106 cells/dish) were incubated with or without 0.02% Ti particles for 30 min or 1, 3, or 18 h. Cell lysates were prepared and Western blotting was performed using anti-phospho-JNK and anti-IκBα antibodies. Results are representative of 3 independent experiments. (B and C) RAW264.7 and J774 cells (2 × 106 cells/dish) were pretreated with EGCG (25, 50, or 100 µM) for 30 min and stimulated with 0.02% Ti particles for an additional 1 h. Equal amounts of cell lysates were separated by 10% SDS-PAGE and analyzed by Western blotting using anti-phospho-JNK (B) and anti-IκBα antibodies (C). Results are representative of 3 independent experiments.
Activated JNK phosphorylates c-Jun and subsequently activates the AP-1 transcription factor (Cano and Mahadevan, 1995; David et al., 2002). To determine the effects of EGCG on AP-1 and NF-κB activations, RAW 264.7 and J774 cells were treated with Ti particles alone or with EGCG (50 µM) for 15, 30, and 60 min. As expected, EMSA revealed strong activations of AP-1 and NF-κB at 15 or 30 min after Ti stimulation. The activations of AP-1 and NF-κB were inhibited substantially by pretreatment with EGCG in a dose-dependent manner (Figures 3A and 3B). Taken together, these data suggest that the inhibitory effect of EGCG on Ti-induced TNF-α release is mediated in part by the downregulation of the JNK/AP-1 and NF-κB pathways.
Effect of EGCG on the activation of AP-1 and NF-κB in Ti particle-stimulated macrophages. RAW264.7 and J774 cells (2 × 106 cells/dish) were pretreated with or without EGCG (50 µM) for 30 min and stimulated with 0.02% Ti particles for an additional 15, 30, or 60 min. Nuclear extracts were harvested at the indicated time points. Transcriptional activities of AP-1 (A) and NF-κB (B) were analyzed by EMSA. Results are representatives of 3 independent experiments.
Inhibitory effect of EGCG on Ti particle-induced osteolysis in a mouse calvaria model in vivo
Having established that EGCG inhibits Ti particle-induced TNF-α release through the downregulation of the JNK/AP-1 and NF-κB pathways in vitro, we next tested whether EGCG prevents or suppresses Ti-induced osteolysis in vivo. Injection of Ti particles into the mouse calvaria dramatically induced osteolysis (Figure 4Ab) compared to uninjected cavaria (Figure 4Aa), while administration of EGCG at 1 mg/kg/d (Figure 4Ac) or 2 mg/kg/d (Figure 4Ad) suppressed osteolysis. Histomorphometric analysis revealed that the average bone area of Ti particle-implanted mice (0.142 mm2) was 41% less than that of sham-treated mice (0.243 mm2). The bone area of Ti-treated mice was increased by treatment with EGCG at 1 or 2 mg/kg/d to 0.207 and 0.24 mm2, respectively (Figure 4B).
Representative photographs of calvarial histology stained with H&E. (A) Mouse calvaria were left untreated (a) or were implanted with Ti particles (b, c, and d). Mice injected with Ti particles were fed with 1 mg/kg/d (c) or 2 mg/kg/d (d) of EGCG. Seven days after implantation, calvaria were sectioned and stained with H&E. Photographs were taken under a light microscope at 40× magnification. (B) Bone area was measured using a digitalized image analyzer (IMT i-Solution; Daejeon, Korea). *P < 0.05.
To examine whether Ti particles induce osteoclast formation in the calvaria and whether EGCG suppresses osteolysis, TRAP staining was performed. Compared to control (Figure 5Aa), a dramatic increase in osteoclasts was observed in the granulomatous tissue of Ti particle-implanted cavarium (Figure 5Ab). Treatment with EGCG at 1 mg/kg/d (Figure 5Ac) and 2 mg/kg/d (Figure 5Ad) markedly decreased osteoclasts in a dose-dependent manner, with observed reductions in osteoclast number of up to 73% (Figure 5B).
Representative photographs of TRAP and TNF-α staining. Mouse calvarial sections prepared as in Fig. 4 were stained with TRAP (A). TRAP-positive osteoclasts were counted using IMT i-Solution (B). Mouse calvarial sections were immunostained with anti-TNF-α antibody (C). **P < 0.01.
To examine whether TNF-α is also decreased by EGCG, immunohistochemical staining was performed. Intensified staining of TNF-α was found in Ti-implanted calvarium (Figure 5Cb) compared to the sham control (Figure 5Ca). EGCG significantly reduced TNF-α in a dose-dependent manner (Figures 5Cc and 5Cd). Taken together, these data demonstrate that EGCG treatment reduces Ti particle-induced TNF-α production and osteoclast formation, and significantly improves osteolytic responses.
Discussion
The pathogenesis of osteolysis and aseptic loosening after TJA involves the activation of macrophages by particulate debris and the release of various inflammatory cytokines (Kim et al., 1993; Sabokbar and Rushton, 1995; Al-Saffar et al., 1996; Xu et al., 1996, 1998).
In particular, TNF-α expression has been studied extensively in response to various stimuli. Our results demonstrate that Ti particles dose- and time-dependently induce TNF-α release in RAW264.7 and J774 macrophage cell lines (Supplemental Data Figure S1). Consistent with a previous report (Schwarz et al., 2000), TNF-α release was detected as early as 30 min after Ti stimulation. NF-κB is a well-studied transcription factor mediating TNF-α induction (Shakhov et al., 1990; Kuprash et al., 1999; Schwarz et al., 2000). AP-1 or other transcription factors may also mediate TNF-α expression (Diaz and Lopez-Berestein, 2000; Kiemer et al., 2002). Interestingly, Soloviev et al (Soloviev et al., 2002) showed that the Ti particle-induced stimulation of ANA-1 murine macrophages induced NF-κB activation, which was dependent on p105 degradation but independent of classical IκBα degradation. The DNA-binding activities of other transcription factors, including AP-1, were unchanged following exposure to Ti particles. Therefore, the authors concluded that NF-kB activation is selective and is one of the primary events that follow the exposure of ANA-1 cells to Ti particles (Soloviev et al., 2002). In this study, Ti stimulation of RAW264.7 and J774 macrophages induced the degradation of IκBα and the activation of signaling molecules such as JNK (Figure 2A). Consistent with these results, pharmacological inhibitors for JNK and IkBα degradation substantially reduced TNF-α release in response to Ti particles (Supplemental Data Figure S2). Although the pharmacological inhibitors are not specific for the signaling molecules, these results suggest that, the JNK/AP-1 and NF-kB pathway also mediates Ti particle-induced TNF-α expression in macrophages. As expected, NF-κB and AP-1 were activated rapidly by Ti particles (Figure 3), clearly demonstrating that the JNK/AP-1 and IκB/NF-κB pathways are activated and involved in TNF-α production in macrophages. We have no direct evidence demonstrating the differential involvement of AP-1 in the Ti-induced TNF-α production, but it may be due to the different macrophage lines used. Various macrophage populations such as alveolar macrophages, P388D and IC21 are known to respond differently to Ti particles (Glant and Jacobs, 1994). Moreover, different amounts and sizes of Ti particles also may cause varying levels of responses, such as AP-1 activation and IκB degradation.
Since Ti particles stimulate TNF-α release through the activation of the JNK/AP-1 and IκB/NF-κB pathways in vitro, we looked for a compound that potentially could inhibit the activation of these transcription factors. Plant-derived flavonoids have attracted considerable recent attention because of their physiological and pharmacological properties such as antioxidative, antibacterial, antimutagenic, and antitumorigenic activities (Morel et al., 1993; Jankun et al., 1997; Harborne and Williams, 2000; Anderson et al., 2001). EGCG dose-dependently inhibited Ti particle-induced TNF-α release in RAW264.7 and J774 cells (Figures 1A and 1B). The results suggest that EGCG has the potential to suppress the activation of macrophages stimulated with wear debris, including Ti particles.
Since EGCG inhibits Ti particle-induced TNF-α release, it may also suppress Ti-induced osteolysis in vivo. Consistently, EGCG significantly suppressed Ti particle-induced osteolysis in the mouse calvarial model (Figure 4). Osteolytic suppression was accompanied by a decrease in TNF-α production and osteoclast numbers (Figure 5). These results strongly suggest that EGCG inhibits Ti-induced osteolysis in vivo through suppression of TNF-α production and osteoclast numbers.
In conclusion, we have demonstrated that EGCG inhibits Ti particle-induced TNF-α release through downregulation of the JNK/AP-1 and NF-κB pathways in vitro and osteolysis in vivo, and therefore may be a potential candidate compound for the prevention and/or treatment of osteolysis and loosening after TJA.
Methods
Materials
Dulbecco's modified Eagle medium (DMEM), L-glutamine, penicillin, streptomycin, and fetal bovine serum (FBS) were from Gibco Invitrogen (Rockville, MD). Anti-phospho-JNK and anti-IκBα antibodies were from Cell Signaling Technology (Beverly, MA). An enzyme-linked immunosorbent assay (ELISA) kit for murine TNF-α was obtained from BioSource International (Camarillo, CA). (-)-Epigallocatechin gallate (EGCG) was from Sigma (St. Louis, MO). A nitrocellulose membrane and an enhanced chemiluminescence (ECL) kit were purchased from Amersham Biosciences Corp. (Piscataway, NJ). Unless otherwise noted, all other chemicals were obtained from Sigma.
Cell culture
The RAW264.7 and J774 murine macrophage cell lines (American Type Culture Collection, Manassas, VA) were cultured in DMEM supplemented with 10% FBS and 1% penicillin/streptomycin in 5% CO2 humidified air at 37℃. RAW264.7 and J774 cells were grown to 60-70% confluency, counted, and cultured in 96-well tissue culture plates (1 × 104 cells/well) or 60-mm tissue culture dishes (2 × 106 cells/dish) (Nunc; Roskilde, Denmark). Cells were pretreated with the indicated concentrations EGCG for 30 min or 1 h and stimulated with Ti particles for the time indicated. Supernatants or cells were collected for further analysis.
Preparation of Ti particles
Commercially pure Ti particles (1-3 µm) (Cerac, Milwaukee, WI) were sterilized by incubation in 25% nitric acid and then incubated in a mixture of 95% ethanol and 0.1 N NaOH for 18-20 h, as previously described (Ragab et al., 1999). The Ti particles were stored at a concentration of 25% Ti (0.25 mg/ml).
ELISA
RAW264.7 and J774 cells at 1 × 104 cells/well were exposed to Ti particles with or without EGCG. Culture medium was collected at the indicated times. The TNF-α levels were measured by ELISA according to the manufacturer's instructions (BioSource). Briefly, serial dilutions of 100 µl of RAW264.7 or J774 cell culture medium were added to the wells of a microtiter plate coated with mouse monoclonal antibody specific for murine TNF-α. Plates were incubated for 2 h at room temperature (RT), washed, and treated with diluted horseradish peroxidase (HP)-conjugated polyclonal rabbit antibody (100 µl) against the specific antigen. Plates with the secondary antibody were incubated for 2 h at RT and washed. The color reaction was developed by adding 100 µl of hydrogen peroxide and tetramethyl benzidine. The enzyme reaction was stopped by adding 1 M phosphoric acid. The absorbance (450 nm/650 nm) was measured in an ELISA plate reader (Biorad 550; Hercules, CA).
Western blotting
Cell lysates were prepared using an extraction buffer composed of 50 mM Tris, pH 7.4, 1% NP-40, 150 mM NaCl, 1 mM EDTA, 1 mM PMSF, 1 mM Na3VO4, 1 mM NaF, 1 µg/ml pepstatin, and 1 µg/ml aprotinin. Protein concentrations were determined by BCA protein assay (Pierce, Rockford, IL). Cell lysates (20 µg) were electrophoresed in 10% SDS-polyacrylamide gels, transferred to nitrocellulose membranes, and blocked with 5% nonfat dried milk in Trisbuffered saline. The nitrocellulose membranes were reacted overnight at 4℃ with antibodies specific for phosphorylated ERK, JNK, or IκBα (1:1000 dilution). Blots were incubated with a HP-conjugated secondary antibody (1:5000 dilution) for 1 h. Proteins were detected by ECL.
Electrophoretic mobility shift assay (EMSA)
Nuclear extracts were prepared as described previously (Park et al., 2004). Protein concentrations were measured with a BCA kit. A gel shift assay was performed by incubating 7 µg of the nuclear extract with 1 µg of poly d (I-C) with 50,000-100,000 cpm of the radiolabeled AP-1 or NF-κB oligonucleotides in 25 µl of binding buffer (10 mM Tris-HCl, pH 7.5, 100 mM NaCl, 1 mM DTT, and 4% glycerol) at RT for 30 min according to the manufacturer's instructions (Promega). The reaction product was subjected to 5% native polyacrylamide gel electrophoresis (PAGE) in 0.5 × TBE buffer (50 mM Tris-HCl, pH 8.5, 50 mM borate, and 1 mM EDTA). The gel was dried in a vacuum for 60 min and exposed to X-ray film.
Mouse calvarial osteolysis model and staining
The mouse calvarial model of Ti particles was described previously (von Knoch et al., 2004). Briefly, 12 healthy 8-week-old male C57BL/6 mice were equally randomized to 4 groups. Group I mice underwent sham surgery only. In groups II, III, and IV, mice were injected with Ti particles. In groups III and IV, mice were fed with a daily dose of 1 or 2 mg of EGCG per kg of body weight, respectively. EGCG was fed from the third day before the operation until sacrifice.
To inject Ti particles, mice were anesthetized, the cranial periosteum was elevated off from the calvarium by sharp dissection, and 30 mg of Ti particles were placed directly on the bone surface as described. Seven days after the operation, mice were sacrificed and the calvaria were excised, fixed, and decalcified in EDTA. Histological sections of calvaria were stained with hematoxylin-eosin (H&E) or tartrate-resistant acid phosphatase (TRAP), according to the manufacturers' instructions.
To examine TNF-α levels in calvarium, sections were blocked with 1.5% normal goat serum for 1 h and incubated with goat anti-mouse TNF-α antibodies (Santa Cruz Biotechnology, Santa Cruz, CA) at 48℃ overnight in a humidified chamber. After washing, sections were incubated for 30 min with biotin-conjugated secondary antibody (rabbit anti-goat IgG) followed by an additional 30 min with avidin-biotin enzyme reagent. The colorimetric reaction was conducted using 3.3'-diaminobezidine tetrahydrochloride. For the negative control, the primary antibody was omitted.
Statistics
Statistical analysis was performed with SPSS 11.0. Data were analyzed by one-way analysis of variance (ANOVA). A P-value < 0.05 was considered significant.
Supplemental Data
Supplemental data include two figures and can be found with this article online.
Abbreviations
- EGCG:
-
(-)-epigallocatechin gallate
- M-CSF:
-
macrophage-colony stimulating factor
- RANKL:
-
receptor activator of NF-κB ligand
References
Algan SM, Purdon M, Horowitz SM . Role of tumor necrosis factor alpha in particulate-induced bone resorption . J Orthop Res 1996 ; 14 : 30 - 35
Al-Saffar N, Khwaja HA, Kadoya Y, Revell PA . Assessment of the role of GM-CSF in the cellular transformation and the development of erosive lesions around orthopaedic implants . Am J Clin Pathol 1996 ; 105 : 628 - 639
Anderson RF, Fisher LJ, Hara Y, Harris T, Mak WB, Melton LD, Packer JE . Green tea catechins partially protect DNA from (.)OH radical-induced strand breaks and base damage through fast chemical repair of DNA radicals . Carcinogenesis 2001 ; 22 : 1189 - 1193
Cano E, Mahadevan LC . Parallel signal processing among mammalian MAPKs . Trends Biochem Sci 1995 ; 20 : 117 - 122
Crotti TN, Smith MD, Findlay DM, Zreiqat H, Ahern MJ, Weedon H, Hatzinikolous G, Capone M, Holding C, Haynes DR . Factors regulating osteoclast formation in human tissues adjacent to peri-implant bone loss: expression of receptor activator NFkappaB, RANK ligand and osteoprotegerin . Biomaterials 2004 ; 25 : 565 - 573
David JP, Sabapathy K, Hoffmann O, Idarraga MH, Wagner EF . JNK1 modulates osteoclastogenesis through both c-Jun phosphorylation-dependent and -independent mechanisms . J Cell Sci 2002 ; 115 : 4317 - 4325
Diaz B, Lopez-Berestein G . A distinct element involved in lipopolysaccharide activation of the tumor necrosis factor-alpha promoter in monocytes . J Interferon Cytokine Res 2000 ; 20 : 741 - 748
Geng DC, Xu YZ, Yang HL, Zhu XS, Zhu GM, Wang XB . Inhibition of titanium particle-induced inflammatory osteolysis through inactivation of cannabinoid receptor 2 by AM630 . J Biomed Mater Res A 2010 ; 95 : 321 - 326
Glant TT, Jacobs JJ . Response of three murine macrophage populations to particulate debris: bone resorption in organ cultures . J Orthop Res 1994 ; 12 : 720 - 731
Harborne JB, Williams CA . Advances in flavonoid research since 1992 . Phytochemistry 2000 ; 55 : 481 - 504
Hofbauer LC, Lacey DL, Dunstan CR, Spelsberg TC, Riggs BL, Khosla S . Interleukin-1beta and tumor necrosis factor-alpha, but not interleukin-6, stimulate osteoprotegerin ligand gene expression in human osteoblastic cells . Bone 1999 ; 25 : 255 - 259
Jankun J, Selman SH, Swiercz R, Skrzypczak-Jankun E . Why drinking green tea could prevent cancer . Nature 1997 ; 387 : 561 -
Katti KS . Biomaterials in total joint replacement . Colloids Surf B Biointerfaces 2004 ; 39 : 133 - 142
Kaufman AM, Alabre CI, Rubash HE, Shanbhag AS . Human macrophage response to UHMWPE, TiAlV, CoCr, and alumina particles: analysis of multiple cytokines using protein arrays . J Biomed Mater Res A 2008 ; 84 : 464 - 474
Kiemer AK, Muller C, Vollmar AM . Inhibition of LPS-induced nitric oxide and TNF-alpha production by alpha-lipoic acid in rat Kupffer cells and in RAW 264.7 murine macrophages . Immunol Cell Biol 2002 ; 80 : 550 - 557
Kim KJ, Rubash HE, Wilson SC, D'Antonio JA, McClain EJ . A histologic and biochemical comparison of the interface tissues in cementless and cemented hip prostheses . Clin Orthop Relat Res 1993 : 142 - 152
Kimble RB, Srivastava S, Ross FP, Matayoshi A, Pacifici R . Estrogen deficiency increases the ability of stromal cells to support murine osteoclastogenesis via an interleukin-1and tumor necrosis factor-mediated stimulation of macrophage colony-stimulating factor production . J Biol Chem 1996 ; 271 : 28890 - 28897
Kuprash DV, Udalova IA, Turetskaya RL, Kwiatkowski D, Rice NR, Nedospasov SA . Similarities and differences between human and murine TNF promoters in their response to lipopolysaccharide . J Immunol 1999 ; 162 : 4045 - 4052
Liu F, Zhu Z, Mao Y, Liu M, Tang T, Qiu S . Inhibition of titanium particle-induced osteoclastogenesis through inactivation of NFATc1 by VIVIT peptide . Biomaterials 2009 ; 30 : 1756 - 1762
Merkel KD, Erdmann JM, McHugh KP, Abu-Amer Y, Ross FP, Teitelbaum SL . Tumor necrosis factor-alpha mediates orthopedic implant osteolysis . Am J Pathol 1999 ; 154 : 203 - 210
Morel I, Lescoat G, Cogrel P, Sergent O, Pasdeloup N, Brissot P, Cillard P, Cillard J . Antioxidant and iron-chelating activities of the flavonoids catechin, quercetin and diosmetin on iron-loaded rat hepatocyte cultures . Biochem Pharmacol 1993 ; 45 : 13 - 19
Nakagawa H, Wachi M, Woo JT, Kato M, Kasai S, Takahashi F, Lee IS, Nagai K . Fenton reaction is primarily involved in a mechanism of (-)-epigallocatechin-3-gallate to induce osteoclastic cell death . Biochem Biophys Res Commun 2002 ; 292 : 94 - 101
Nakashima Y, Sun DH, Trindade MC, Maloney WJ, Goodman SB, Schurman DJ, Smith RL . Signaling pathways for tumor necrosis factor-alpha and interleukin-6 expression in human macrophages exposed to titanium-alloy particulate debris in vitro . J Bone Joint Surg Am 1999 ; 81 : 603 - 615
Park EK, Kim MS, Lee SH, Kim KH, Park JY, Kim TH, Lee IS, Woo JT, Jung JC, Shin HI, Choi JY, Kim SY . Furosin, an ellagitannin, suppresses RANKL-induced osteoclast differentiation and function through inhibition of MAP kinase activation and actin ring formation . Biochem Biophys Res Commun 2004 ; 325 : 1472 - 1480
Ragab AA, Van De Motter R, Lavish SA, Goldberg VM, Ninomiya JT, Carlin CR, Greenfield EM . Measurement and removal of adherent endotoxin from titanium particles and implant surfaces . J Orthop Res 1999 ; 17 : 803 - 809
Ren W, Wu B, Peng X, Mayton L, Yu D, Ren J, Chen BD, Wooley PH . Erythromycin inhibits wear debris-induced inflammatory osteolysis in a murine model . J Orthop Res 2006 ; 24 : 280 - 290
Sabokbar A, Rushton N . Role of inflammatory mediators and adhesion molecules in the pathogenesis of aseptic loosening in total hip arthroplasties . J Arthroplasty 1995 ; 10 : 810 - 816
Schwarz EM, Lu AP, Goater JJ, Benz EB, Kollias G, Rosier RN, Puzas JE, O'Keefe RJ . Tumor necrosis factor-alpha/nuclear transcription factor-kappaB signaling in periprosthetic osteolysis . J Orthop Res 2000 ; 18 : 472 - 480
Shakhov AN, Collart MA, Vassalli P, Nedospasov SA, Jongeneel CV . Kappa B-type enhancers are involved in lipopolysaccharide-mediated transcriptional activation of the tumor necrosis factor alpha gene in primary macrophages . J Exp Med 1990 ; 171 : 35 - 47
Shanbhag AS, Jacobs JJ, Black J, Galante JO, Glant TT . Macrophage/particle interactions: effect of size, composition and surface area . J Biomed Mater Res 1994a ; 28 : 81 - 90
Shanbhag AS, Jacobs JJ, Glant TT, Gilbert JL, Black J, Galante JO . Composition and morphology of wear debris in failed uncemented total hip replacement . J Bone Joint Surg Br 1994b ; 76 : 60 - 67
Shanbhag AS, Hasselman CT, Rubash HE . The John Charnley Award. Inhibition of wear debris mediated osteolysis in a canine total hip arthroplasty model . Clin Orthop Relat Res 1997 : 33 - 43
Soloviev A, Schwarz EM, Kuprash DV, Nedospasov SA, Puzas JE, Rosier RN, O'Keefe RJ . The role of p105 protein in NFkappaB activation in ANA-1 murine macrophages following stimulation with titanium particles . J Orthop Res 2002 ; 20 : 714 - 722
von Knoch M, Jewison DE, Sibonga JD, Sprecher C, Morrey BF, Loer F, Berry DJ, Scully SP . The effectiveness of polyethylene versus titanium particles in inducing osteolysis in vivo . J Orthop Res 2004 ; 22 : 237 - 243
von Knoch F, Wedemeyer C, Heckelei A, Sprecher C, Saxler G, Hilken G, Henschke F, von Knoch M, Bereiter H, Loer F, von Knoch M . [A comparison of the antiresorptive effects of bisphosphonates and statins on polyethylene particle-induced osteolysis] . Biomed Tech (Berl) 2005a ; 50 : 195 - 200
von Knoch F, Heckelei A, Wedemeyer C, Saxler G, Hilken G, Brankamp J, Sterner T, Landgraeber S, Henschke F, Loer F, von Knoch M . Suppression of polyethylene particle-induced osteolysis by exogenous osteoprotegerin . J Biomed Mater Res A 2005b ; 75 : 288 - 294
Xu JW, Konttinen YT, Lassus J, Natah S, Ceponis A, Solovieva S, Aspenberg P, Santavirta S . Tumor necrosis factor-alpha (TNF-alpha) in loosening of total hip replacement (THR) . Clin Exp Rheumatol 1996 ; 14 : 643 - 648
Xu JW, Li TF, Partsch G, Ceponis A, Santavirta S, Konttinen YT . Interleukin-11 (IL-11) in aseptic loosening of total hip replacement (THR) . Scand J Rheumatol 1998 ; 27 : 363 - 367
Yang CS, Wang ZY . Tea and cancer . J Natl Cancer Inst 1993 ; 85 : 1038 - 1049
Yang F, de Villiers WJ, McClain CJ, Varilek GW . Green tea polyphenols block endotoxin-induced tumor necrosis factor-production and lethality in a murine model . J Nutr 1998 ; 128 : 2334 - 2340
Yun JH, Pang EK, Kim CS, Yoo YJ, Cho KS, Chai JK, Kim CK, Choi SH . Inhibitory effects of green tea polyphenol (-)-epigallocatechin gallate on the expression of matrix metalloproteinase-9 and on the formation of osteoclasts . J Periodontal Res 2004 ; 39 : 300 - 307
Acknowledgements
This work was supported by a grant from the Ministry of Health and Welfare of Korea [A010252 and A080840] and by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology [2010-0029494].
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplementary Information accompanies the paper on the Experimental & Molecular Medicine website
Supplementary information
Rights and permissions
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Jin, S., Park, JY., Hong, JM. et al. Inhibitory effect of (-)-epigallocatechin gallate on titanium particle-induced TNF-α release and in vivo osteolysis. Exp Mol Med 43, 411–418 (2011). https://doi.org/10.3858/emm.2011.43.7.045
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3858/emm.2011.43.7.045