Abstract
An association between inflammatory processes and the pathogenesis of insulin resistance has been increasingly suggested. The IκB kinase-β (IKK-β)/ nuclear factor-κB (NF-κB) pathway is a molecular mediator of insulin resistance. S-Adenosyl-L-methionine (SAM) has both antioxidative and anti-inflammatory properties. We investigated the effects of SAM on the glucose transport and insulin signaling impaired by the tumor necrosis factor α (TNFα) in 3T3-L1 adipocytes. SAM partially reversed the basal and insulin stimulated glucose transport, which was impaired by TNFα. The TNFα-induced suppression of the tyrosine phosphorylation of the insulin receptor substrate-1 (IRS-1) and Akt in 3T3-L1 adipocytes was also reversed by SAM. In addition, SAM significantly attenuated the TNFα-induced degradation of IκB-α and NF-κB activation. Interestingly, SAM directly inhibited the kinase activity of IKK-β in vitro. These results suggest that SAM can alleviate TNFα mediated-insulin resistance by inhibiting the IKK-β/NF-κB pathway and thus can have a beneficial role in the treatment of type 2 diabetes mellitus.
Similar content being viewed by others
Introduction
Mounting evidence suggests that inflammatory processes are related to the pathogenesis of insulin resistance. The IκB kinase-β (IKK-β)/NF-κB pathway is a molecular mediator of insulin resistance (Shoelson et al., 2003). Shoelson et al. studied the glucose lowering effects of salicylates and found that activation of IKK-β/NF-κB pathway causes insulin resistance, while inhibition of the pathway can reverse insulin resistance. Various inflammatory stimuli activate IKK-β and anti-inflammatory agents with IKK-β-inhibiting properties have been suggested to improve insulin resistance (Shoelson et al., 2006). Insulin signaling pathway has been well studied. When insulin binds to insulin receptor (IR), IR activates insulin receptor substrate (IRS) through specific tyrosine phosphorylation, which in turn induces a serial activation of PI3K, Akt and glucose transporter (GLUT) translocation to the plasma membrane. Finally, glucose uptake through GLUT is increased (Schinner et al., 2005). Inflammatory cytokines such as tumor necrosis factor α (TNFα activates several kinases including MAPK, JNK and IKKβ, which phosphorylate serine residue on IR or IRS-1. The serine phosphorylation inhibits activation of IR or IRS-1 and results in insulin resistance (Aguirre et al., 2000; Gao et al., 2002). IKKβ is also indirectly involved in insulin resistance by activating NF-κB which stimulates other inflammatory mediators (Baud and Karin, 2001).
S-Adenosyl-L-methionine (SAM) is a well-known methyl donor that is vital for many reactions catalyzed by methyltransferases (Chiang et al., 1996; Cheng and Blumenthal, 1999). SAM is also known to be as effective as celecoxib or non-steroidal anti-inflammatory drugs in the management of osteoarthritis (di Padova, 1987; Najm et al., 2004). Many possible mechanisms of action of SAM have been suggested in the treatment of osteoarthritis, and the reduction of inflammatory mediators is one of them (Hosea Blewett, 2008). SAM also serves as a precursor molecule in the transsulfuration pathway, which leads to the synthesis of glutathione (GSH). Glutathione plays an important role in the natural defense mechanism against oxidative stress. SAM can restore the mitochondrial glutathione concentration, decrease the concentration of TNFα, and increase the expression of interleukin 10 (IL-10) (Purohit et al., 2007). These effects may explain its protective role in alcoholic liver disease and acetaminophen toxicity. If SAM has an anti-inflammatory effect through suppression of the IKK-β/NF-κB pathway, it may be an effective treatment modality for type 2 diabetes mellitus as in the case of salicylates. We showed in our previous study that the treatment with SAM improved the whole body insulin sensitivity in Otsuka Long Evans Tokushima Fatty (OLETF) rats (Jin et al., 2007).
The objective of this study was to investigate the effect of SAM on inflammatory signaling and insulin resistance. In this study, we demonstrate that SAM could improve the TNFα-induced impairment of the glucose transport and insulin-signaling pathway, and could directly inhibit IKK-β activity.
Results
SAM improved glucose transport impaired by TNFα
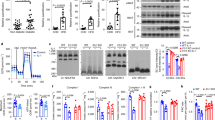
To investigate the effect of SAM on insulin resistance, we tested whether SAM could reverse the impaired glucose transport which was induced by TNFα in 3T3-L1 adipocytes. Treatment with TNFα (50 ng/ml) for 72 h reduced the basal and insulin-stimulated glucose transport by 61.5% (P < 0.001) and 77.2% (P < 0.001), respectively when compared to untreated cells (Figure 1). Pretreatment with 1 mM SAM increased the basal and insulin stimulated glucose transport by 44.7% (P < 0.001) and 101.0% (P < 0.001), respectively when compared to TNFα treated cells. However, SAM treatment restored the impaired glucose transport to about half of the control levels. In the absence of TNFα, treatment of SAM did not affect glucose transport either in the basal or insulin-stimulated level. These results suggest that SAM specifically ameliorates glucose transport impaired by TNFα.
Effects of SAM on glucose transport in TNFα treated 3T3-L1 adipocytes. Cells were treated with SAM (1 mM) and TNFα (50 ng/ml) for 72 h and incubated with or without insulin (100 nM) for 30 min. Glucose transport levels seen in the cells not treated with SAM, TNFα or insulin (control) was expressed as 100 and the others were expressed as its relative values. *P < 0.001 compared to the control, †P < 0.001 compared to the level of cells treated with insulin and not TNFα, **P < 0.001 compared to the level of cells treated with TNFα and not but SAM. ‡P < 0.001 compared to cells treated with insulin and TNFα.
SAM enhances insulin signaling impaired by TNFα
To elucidate the molecular mechanisms underlying the glucose transport improved by SAM, we evaluated the effect of SAM on phosphorylation of IRS-1 and Akt, two important factors in the insulin signaling pathway, after TNFα treatment in 3T3-L1 adipocytes. Treatment with 50 ng/ml of TNFα for 18 h reduced the IRS-1 tyrosine phosphorylation at position Y612 and the Akt phosphorylation at position Ser473. Consistent with the result of glucose transport in Figure 1, pretreatment of cells with 0.5 mM or 1 mM SAM partially reversed the impaired phosphorylation of IRS-1 and Akt (Figure 2).
Effects of SAM on phosphorylations of IRS-1 and Akt in 3T3-L1 adipocytes treated with TNFα. (A) Adipocytes were treated with SAM and TNFα (50 ng/ml) for 18 h, and then treated with insulin for 30 min before harvesting. Cell lysates were subjected to immunoblot analysis. (B) The band intensities of phopho-IRS-1 (pIRS-1) or pAkt were normalized by their total IRS-1 or Akt, respectively. *P < 0.01 compared to the value of control cells treated with insulin (n = 3). **P < 0.01 compared to the value of cells treated with insulin and TNF-α (n = 3).
SAM reduces DNA binding activity of NF-κB induced by TNFα
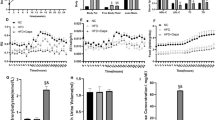
Since activation of IKK-β/NF-κB is involved in the TNFα-induced insulin resistance, we investigated whether SAM has an inhibitory effect on the IKK-β/NF-κB pathway. The effect of SAM on the DNA binding activity of NF-κB was investigated in 3T3-L1 adipocytes. The DNA binding activity of NF-κB was dramatically increased by TNFα (Figure 3A). Two specific bands were observed, the upper band was super-shifted by the addition of antibody against p65 (lane 8), suggesting that the upper band was p65/p50 complex and the lower band was p50/p50 homodimer (Udalova et al., 2000; Daosukho et al., 2002). SAM efficiently decreased the TNFα-induced DNA binding activity of NF-κB. The inhibitory effect of SAM on NF-κB activity was also confirmed in HepG2 cells (human hepatoma cell line) and L6 cells (rat skeletal muscle cell line) (data not shown). In addition, TNFα-induced translocation of p65 into nucleus was confirmed by Western blot analysis, and treatment of SAM significantly inhibited p65 translocation (Figure 3B). Total amount of p65 in the cell lysates was not changed by TNFα or SAM. To elucidate how SAM inhibits the activation of NF-κB in the presence of TNFα, the effect of SAM on the degradation of IκB-α was investigated by Western-blot analysis. The TNFα induced degradation of IκB-α in 3T3-L1 adipocytes and treatment with SAM partially inhibited this degradation (Figure 3B).
Effects of SAM on TNFα-induced NF-κB activation in adipocytes. (A) After adipocytes were treated with SAM (1 mM) for the indicated days, TNFα (50 ng/ml) were treated for 18 h. Nuclear proteins were prepared and NF-κB binding activity was determined by EMSA. Lane 1 shows free probe (FP) alone. Anti-p65 antibody was incubated with the nuclear extract before the addition of the probe (lane 8). (B) After treatment of cells with TNFα or SAM, nuclear extracts and cell lysates were subjected to immunoblot analysis with anti-p65, anti-IkBα or γ-tubulin. (C) The band intensity of p65 in the nuclear extract of the cells treated with TNFα was expressed as 100 and the others were expressed as its relative values (upper panel) (n = 3). *P < 0.01 compared to the value of the cells treated with TNFα. **P < 0.05 compared to the value of the cells treated with TNFα. The band intensities of p65 in the cell lysates were normalized by those of γ-tubulin. The value of the control cells was expressed as 100 and the others were expressed as its relative values (lower panel) (n = 4). *P < 0.01 compared to the value of the control cells. **P < 0.01 compared to the value of cells treated with TNFα.
SAM directly affects the kinase activity of IKK-β
As the results in Figure 3 suggest that SAM has an inhibitory effect on NF-κB activation at the upstream of Iκβ degradation step, we tested whether SAM could directly inhibit the kinase activity of IKK-β by using an in vitro IKK-β activity assay. As shown in Figure 4, SAM significantly inhibited the kinase activity of IKK-β compared to the controls in a dose-dependent manner. The inhibitory effect of 1 mM SAM was comparable to that of 10 µM staurosporine, a strong kinase inhibitor (Seo and Seo, 2009). This result clearly showed that SAM could act as an inhibitor of IKK-β.
SAM inhibited IKK-β kinase activity in a dose-dependent manner. IKK-β and different concentrations of SAM had been preincubated for 10 min and the kinase reaction was allowed to continue for 30 min. IKK-β kinase activity was also measured in the presence of 10 µM staurosporine (Stau), a strong kinase inhibitor. *P = 0.014 by Kruskal Wallace test, *P < 0.001 by ANOVA with posthoc test (n = 3).
Discussion
In the present study, we showed that SAM inhibited the activity of IKK-β and the degradation of IκB and the NF-κB binding activity induced by TNFα. SAM improved the glucose transport that was impaired by TNFα. TNFα treatment decreased glucose transport both in the absence and in the presence of insulin, and SAM partially recovered the effect of TNFα regardless of the presence of insulin (Figure 1). Although it has not been well investigated how TNFα decreases the basal level of glucose transport, it may be explained by the significant reduction of total IRS level after the treatment of TNFα shown in Figure 2. SAM also increased the level of total IRS in the presence of TNFα (Figure 2), and similarly increased the basal level of glucose transport (Figure 1). Definitely, further studies are required to clarify this possibility. Anyhow, the results in this study suggest that SAM could improve insulin sensitivity, which has been reported in a previous animal study (Jin et al., 2007).
Various inflammatory stimuli including interleukin-1β, TNFα, IL-6 and interferon-β activate IKK. Activated IKK catalyzes the phosphorylation of IκB which leads to the degradation of IκB and allows the translocation of NF-κB to the nucleus. Activated IKK-β and/or NF-κB activation is known to play a central role in the pathogenesis of insulin resistance (Evans et al., 2002). Suppression of IKK-β/NF-κB activation could be a therapeutic approach in type 2 diabetes mellitus. Some anti-inflammatory drugs, e.g. salicylate, aspirin and sulfasalazine, have an inhibitory effect on the IKK-β/NF-κB pathway. Their insulin sensitizing and glucose lowering effect has been shown in animal and human studies (Kim et al., 2001; Hundal et al., 2002; Goldfine et al., 2008). Although it is possible that most of the anti-inflammatory drugs have an inhibitory effect on IKK-β/NF-κB pathway, few studies have explored this hypothesis (De Bosscher et al., 1997; Yin et al., 1998; Yamamoto et al., 1999). SAM, which was tested in our study, plays not only a role as intracellular antioxidant but also as therapeutic agent in osteoarthritis. Several possible mechanisms of SAM in the treatment of osteoarthritis were proposed: 1) the reduction of inflammatory mediators such as TNFα and nitric oxide (NO), 2) an increase of proteoglycan synthesis and glutathione and 3) DNA methylation (Hosea Blewett, 2008). There is increasing evidence indicating that the modulation of the inflammatory response could be an explanation for the beneficial effects of SAM. It has been reported that SAM inhibits the LPS-induced expression of TNFα, the TNFα promoter activity and the expression of the inducible NO synthase (iNOS) in vivo. In addition, it stimulates the expression of interleukin-10 in macrophages (Watson et al., 1999; Veal et al., 2004; Ara et al., 2008). Based on these data, we analogized that SAM would have an anti-inflammatory effect through inhibition of the IKK-β/NF-κB pathway and possibly has a beneficial role in the treatment of type 2 diabetes mellitus.
The role TNFα in the pathogenesis of insulin resistance is well established. An increase in the expression of TNFα has been postulated to induce insulin resistance. In addition, direct exposure of healthy individuals, animals or cells to TNFα also induces insulin resistance (Sethi and Hotamisligil, 1999; Cawthorn and Sethi, 2008). It is interesting to note that SAM improved the glucose transport and insulin signaling that was impaired by TNFα although its effect was not completely reversed in this study. We showed in the present study that SAM could have an insulin sensitizing effect by inhibiting the activation of IKK-β, degradation of IκB, and activation of NF-κB.
Besides the anti-inflammatory effect of SAM, it also acts as an antioxidant. It plays an antioxidative role as a precursor of glutathione, which is an important intracellular antioxidant. Supplementation of SAM could increase the intracellular glutathione concentration (Garcia-Roman et al., 2008). We also observed that SAM could increase the intracellular glutathione concentration after intracellular depletion of glutathione by high concentrations of glucose (data not shown). In addition to its role as a precursor of glutathione, SAM itself has several characteristics as a good antioxidant. It has been reported that SAM can scavenge hydroxyl radicals more effectively than glutathione (Evans et al., 1997) and that it does not stimulate the formation of hydroxyl radicals. SAM has also been reported to have little pro-oxidative effect in a variety of lipid-peroxidation systems. NF-κB is the common denominator of oxidative stress activated pathway and inflammation (Kim et al., 2008). The inhibitory role of SAM on TNFα-induced activation of NF-κB shown in this study could be attributed to its antioxidative properties and its inhibitory effect on the activity of IKK-β.
In the present study, SAM reversed the impairment of the insulin-induced tyrosine phosphorylation of IRS-1 and AKT caused by TNFα. Indeed, SAM appears to intersect the early events in the insulin-signaling pathway. Since SAM acts as a major methyl donor in vivo, it may affect the methylation of a specific DNA sequence or protein involved in the insulin-signaling pathway. However, we did not observe any differences in the methylation status of genomic DNA after SAM treatment in a previous animal study (Jin et al., 2007).
When 400-1,000 mg of SAM was administered orally to healthy volunteers, the plasma concentrations were in the range of 0.5-1 mg/L (1.25-2.5 µM) (Friedel et al., 1989; Evans et al., 1997). The tissue concentration of SAM is reported to be about 40 times higher than in the plasma in humans (Becker et al., 2003). In this study, cells were treated with 0.5 mM or 1 mM SAM, which would be 2.5-5 times higher than its usual therapeutic dose. Therefore, in order to observe the insulin sensitizing effect of SAM in type 2 diabetic patients, much higher dose of SAM would need to be administered.
In conclusion, SAM inhibited the activity of IKK-β and the degradation of IkB and the NF-κB binding activity induced by TNFα. SAM reversed the impaired insulin signaling and glucose transport in a TNFα-induced model of insulin resistance. To the best of our knowledge, this is the first report on the beneficial metabolic effect of SAM in a TNFα-induced insulin resistance model. Our results suggest that SAM could have a beneficial role in the treatment of type 2 diabetes mellitus. However, further clinical studies are required to confirm these observations.
Methods
Cell culture and differentiation
3T3-L1 preadipocytes were maintained in DMEM supplemented with 10% fetal calf serum (Invitrogen, Carlsbad, CA). Two days after the cells became confluent, their differentiation was induced by adding an adipogenic differentiation mixture [0.25 µM dexamethasone, 0.5 mM 3-isobutyl-α-methylxanthine, and 5 µg/ml insulin] to the DMEM (supplemented with 10% FBS) for two days. The medium was then replaced with 10% FBS DMEM containing 1 µg/ml insulin for two days and the cells were then incubated with 10% FBS DMEM for up to six days. The medium was changed every other day.
Glucose-transport assay
3T3-L1 cells were cultured in 12-well cluster plates and induced to differentiate as described above. The adipocytes were treated with 1 mM SAM (Yooyoung Pharm, Seoul, Korea) and 50 ng/mL TNFα (R&D Systems, Minneapolis, MN) for 72 h and then incubated with or without insulin (100 nM) for 30 min (Rotter et al., 2003; Burgermeister et al., 2006). The cells were then placed in a transport buffer consisting of 20 mmol/L HEPES (pH 7.4), 140 mmol/L NaCl, 5 mmol/L KCl, 2.5 mmol/L MgCl2, 1 mmol/L CaCl2 and 0.1% BSA (wt/vol). Next, insulin (100 nmol/L) was added for 30 min, followed by treatment with 10 µmol/L 2-deoxyglucose (1.0 µCi/ml) for 15 min at 37℃. The reactions were stopped by aspirating the media and the cells were washed thoroughly with cold PBS. The cells were lysed in 0.5N NaOH and the radioactivity was determined by liquid scintillation counting.
Western-blot analysis
The level of degradation of IκB-α and the level of phosphorylation of IKK-β, IRS-1 and Akt were determined by Western blot analysis. Differentiated 3T3-L1 adipocytes were treated with SAM, TNFα or insulin as indicated. Cellular proteins (20 µg) were separated by SDS-polyacrylamide gel electrophoresis (SDS-PAGE; 8% gel) and then transferred onto a nitrocellulose membrane. The membrane was blocked in 5% skim milk and incubated with specific primary antibodies in 0.1% Tween20/TBS. The primary antibodies used were as follows: IκB-α, pAkt (ser473) and Akt (Cell Signaling, Danvers, MA), IRS-1, γ-tubulin (Sigma-Aldrich, St. Louis, MO), p65, GAPDH (Santa Cruz biotechnology, Santa Cruz, CA) and phosphorylated IRS-1 (Y612) (Biosource, Camarillo, CA). Bound primary antibodies were detected by incubation of the membranes with a secondary horseradish peroxidase (HRP)-conjugated IgG antibody and visualized by ECL (Pierce).
Electrophoretic mobility shift assay
The activity of NF-κB was determined by an electrophoretic mobility shift assay (EMSA) as previously described (Skulachev, 1998). Nuclear proteins were isolated from adipocytes as described with minor modifications (Dignam et al., 1983). The following double-stranded oligonucleotide sequence containing the NF-κB binding sequence was used as a probe: forward 5'-GTTAGTTGAGGGGACTTTCCCAGGC-3' (Staiger et al., 2006). The oligonucleotide and its complementary strand were annealed and labeled with α-32P dATP using Klenow DNA polymerase (Ambion, Austin, TX). The labeled probe (35,000 cpm) was incubated with nuclear proteins (5 µg) at room temperature for 15 min in 10 mmol/L HEPES (pH 7.9) containing 50 mmol/L KCl, 0.1 mmol/L EDTA, 0.25 mmol/L DTT, 0.1 mg/mL poly (dIdC), 0.01% Nonidet P40 and 10% glycerol. The reaction mixtures were separated on a 5% polyacrylamide gel in 0.5× Tris-borate-EDTA (TBE) buffer. The DNA-protein complexes were visualized by autoradiography.
IKK-β kinase activity
The kinase activity of IKK-β was determined by a colorimetric enzyme-linked immunosorbent assay (ELISA; HTScan® IKKβ Kinase Assay Kit, Cell Signaling Technology Inc., Danvers, MA) according to the manufacturer's instructions. Briefly, the kinase reaction was initiated by addition of the substrate and ATP to the mixture of IKK-β and SAM that had been pre-incubated for 10 min. A biotinylated peptide (Biotin-IκB-α) that contained the residues around the position of Ser-32 of IκB-α served as substrate. The final reaction mixture contained 50 ng IKK-β kinase protein, 1.5 µM Biotin-IκB-α, 200 µM ATP, 25 mM Tris-HCl (pH 7.5), 10 mM MgCl2, 5 mM β-glycerophosphate, 0.1 mM Na2VO4, 2 mM DTT and different concentrations of SAM. Distilled water (DW) was used as control. The kinase reaction was allowed to continue for 30 min and was then quenched by addition of EDTA (50 mM, pH 8). The mixtures were incubated in a 96-well streptavidin-coated plate (Pierce Biotechnology Inc., Rockford, IL) to immobilize the substrate peptide. The mixtures were then treated with a phospho-IκB-α (Ser32/36) mouse monoclonal antibody followed by addition of a HRP-labeled secondary antibody to detect phosphorylation of the IKK-β substrate peptide. The absorbance was read at 450 nm with a microtiter plate reader.
Statistical analysis
Results are reported as mean ± standard error mean (SEM). Analysis of variance (ANOVA) with post hoc testing and Wilcoxon signed-rank tests were used as appropriate. A P value of less than 0.05 was considered statistically significant.
Abbreviations
- DW:
-
distilled water
- IKK-β:
-
IκB kinase-β
- IRS-1:
-
insulin receptor substrate-1
- NO:
-
nitric oxide
- SAM:
-
S-Adenosyl-L-methionine
References
Aguirre V, Uchida T, Yenush L, Davis R, White MF . The c-Jun NH(2)-terminal kinase promotes insulin resistance during association with insulin receptor substrate-1 and phosphorylation of Ser(307) . J Biol Chem 2000 ; 275 : 9047 - 9054
Ara AI, Xia M, Ramani K, Mato JM, Lu SC . S-adenosylmethionine inhibits lipopolysaccharide-induced gene expression via modulation of histone methylation . Hepatology 2008 ; 47 : 1655 - 1666
Baud V, Karin M . Signal transduction by tumor necrosis factor and its relatives . Trends Cell Biol 2001 ; 11 : 372 - 377
Becker A, Henry RM, Kostense PJ, Jakobs C, Teerlink T, Zweegman S, Dekker JM, Nijpels G, Heine RJ, Bouter LM, Smulders YM, Stehouwer CD . Plasma homocysteine and S-adenosylmethionine in erythrocytes as determinants of carotid intima-media thickness: different effects in diabetic and non-diabetic individuals. The Hoorn Study . Atherosclerosis 2003 ; 169 : 323 - 330
Burgermeister E, Schnoebelen A, Flament A, Benz J, Stihle M, Gsell B, Rufer A, Ruf A, Kuhn B, Marki HP, Mizrahi J, Sebokova E, Niesor E, Meyer M . A novel partial agonist of peroxisome proliferator-activated receptor-gamma (PPARgamma) recruits PPARgamma-coactivator-1alpha, prevents triglyceride accumulation, and potentiates insulin signaling in vitro . Mol Endocrinol 2006 ; 20 : 809 - 830
Cawthorn WP, Sethi JK . TNF-alpha and adipocyte biology . FEBS Lett 2008 ; 582 : 117 - 131
Cheng X, Blumenthal RM . S-Adenosylmethionine-dependent methyltransferases: structures and functions . World Scientific Publication Co 1999
Chiang PK, Gordon RK, Tal J . S-Adenosylmethionine and methylation . FASEB J 1996 ; 10 : 471 - 480
Daosukho C, Kiningham K, Kasarskis EJ, Ittarat W, St Clair DK . Tamoxifen enhancement of TNF-alpha induced MnSOD expression: modulation of NF-kappaB dimerization . Oncogene 2002 ; 21 : 3603 - 3610
De Bosscher K, Schmitz ML, Vanden Berghe W, Plaisance S, Fiers W, Haegeman G . Glucocorticoid-mediated repression of nuclear factor-kappaB-dependent transcription involves direct interference with transactivation . Proc Natl Acad Sci USA 1997 ; 94 : 13504 - 13509
di Padova C . S-adenosylmethionine in the treatment of osteoarthritis. Review of the clinical studies . Am J Med 1987 ; 83 : 60 - 65
Dignam JD, Lebovitz RM, Roeder RG . Accurate transcription initiation by RNA polymerase II in a soluble extract from isolated mammalian nuclei . Nucleic Acids Res 1983 ; 11 : 1475 - 1489
Evans JL, Goldfine ID, Maddux BA, Grodsky GM . Oxidative stress and stress-activated signaling pathways: a unifying hypothesis of type 2 diabetes . Endocr Rev 2002 ; 23 : 599 - 622
Evans PJ, Whiteman M, Tredger JM, Halliwell B . Antioxidant properties of S-adenosyl-L-methionine: a proposed addition to organ storage fluids . Free Rad Biol Med 1997 ; 23 : 1002 - 1008
Friedel HA, Goa KL, Benfield P . S-adenosyl-L-methionine. A review of its pharmacological properties and therapeutic potential in liver dysfunction and affective disorders in relation to its physiological role in cell metabolism . Drugs 1989 ; 38 : 389 - 416
Gao Z, Hwang D, Bataille F, Lefevre M, York D, Quon MJ, Ye J . Serine phosphorylation of insulin receptor substrate 1 by inhibitor kappa B kinase complex . J Biol Chem 2002 ; 277 : 48115 - 48121
Garcia-Roman R, Salazar-Gonzalez D, Rosas S, Arellanes-Robledo J, Beltran-Ramirez O, Fattel-Fazenda S, Villa-Trevino S . The differential NF-κB modulation by S-Adenosyl-L-methionine, N-acetylcysteine and quercetin on the promotion stage of chemical hepatocarcinogenesis . Free Radic Res 2008 ; 42 : 331 - 343
Goldfine AB, Silver R, Aldhahi W, Cai D, Tatro E, Lee J, Shoelson SE . Use of Salsalate to Target Inflammation in the Treatment of Insulin Resistance and Type 2 Diabetes . Clin Transl Sci 2008 ; 1 : 36 - 43
Hosea Blewett HJ . Exploring the mechanisms behind S-adenosylmethionine (SAMe) in the treatment of osteoarthritis . Crit Rev Food Sci Nutr 2008 ; 48 : 458 - 463
Hundal RS, Petersen KF, Mayerson AB, Randhawa PS, Inzucchi S, Shoelson SE, Shulman GI . Mechanism by which high-dose aspirin improves glucose metabolism in type 2 diabetes . J Clin Invest 2002 ; 109 : 1321 - 1326
Jin CJ, Park HK, Cho YM, Pak YK, Lee KU, Kim MS, Friso S, Choi SW, Park KS, Lee HK . S-adenosyl-L-methionine increases skeletal muscle mitochondrial DNA density and whole body insulin sensitivity in OLETF rats . J Nutr 2007 ; 137 : 339 - 344
Kim H, Hwang JS, Woo CH, Kim EY, Kim TH, Cho KJ, Seo JM, Lee SS, Kim JH . TNF-α-induced up-regulation of intracellular adhesion molecule-1 is regulated by a Rac-ROS-dependent cascade in human airway epithelial cells . Exp Mol Med 2008 ; 40 : 167 - 175
Kim JK, Kim YJ, Fillmore JJ, Chen Y, Moore I, Lee J, Yuan M, Li ZW, Karin M, Perret P, Shoelson SE, Shulman GI . Prevention of fat-induced insulin resistance by salicylate . J Clin Invest 2001 ; 108 : 437 - 446
Najm WI, Reinsch S, Hoehler F, Tobis JS, Harvey PW . S-adenosyl methionine (SAMe) versus celecoxib the for treatment of osteoarthritis symptoms: a double-blind cross-over trial. [ISRCTN36233495] . BMC Musculoskelet Disord 2004 ; 5 : 6 -
Purohit V, Abdelmalek MF, Barve S, Benevenga NJ, Halsted CH, Kaplowitz N, Kharbanda KK, Liu QY, Lu SC, McClain CJ, Swanson C, Zakhari S . Role of S-adenosylmethionine, folate, and betaine in the treatment of alcoholic liver disease: summary of a symposium . Am J Clin Nutr 2007 ; 86 : 14 - 24
Rotter V, Nagaev I, Smith U . Interleukin-6 (IL-6) induces insulin resistance in 3T3-L1 adipocytes and is, like IL-8 and tumor necrosis factor-alpha, overexpressed in human fat cells from insulin-resistant subjects . J Biol Chem 2003 ; 278 : 45777 - 45784
Schinner S, Scherbaum WA, Bornstein SR, Barthel A . Molecular mechanisms of insulin resistance . Diabet Med 2005 ; 22 : 674 - 682
Seo SR, Seo JT . Calcium overload is essential for the acceleration of staurosporine-induced cell death following neuronal differentiation in PC12 cells . Exp Mol Med 2009 ; 41 : 269 - 276
Sethi JK, Hotamisligil GS . The role of TNF alpha in adipocyte metabolism . Semin Cell Dev Biol 1999 ; 10 : 19 - 29
Shoelson SE, Lee J, Yuan M . Inflammation and the IKK beta/I kappa B/NF-kappa B axis in obesity- and diet-induced insulin resistance . Int J Obes Relat Metab Disord 2003 ; 27 : S49 - S52
Shoelson SE, Lee J, Goldfine AB . Inflammation and insulin resistance . J Clin Invest 2006 ; 116 : 1793 - 1801
Skulachev VP . Uncoupling: new approaches to an old problem of bioenergetics . Biochim Biophys Acta 1998 ; 1363 : 100 - 124
Staiger K, Staiger H, Weigert C, Haas C, Haring HU, Kellerer M . Saturated, but not unsaturated, fatty acids induce apoptosis of human coronary artery endothelial cells via nuclear factor-kappaB activation . Diabetes 2006 ; 55 : 3121 - 3126
Udalova IA, Richardson A, Denys A, Smith C, Ackerman H, Foxwell B, Kwiatkowski D . Functional consequences of a polymorphism affecting NF-kappaB p50-p50 binding to the TNF promoter region . Mol Cell Biol 2000 ; 20 : 9113 - 9119
Veal N, Hsieh CL, Xiong S, Mato JM, Lu S, Tsukamoto H . Inhibition of lipopolysaccharide-stimulated TNF-alpha promoter activity by S-adenosylmethionine and 5'-methylthioadenosine . Am J Physiol Gastrointest Liver Physiol 2004 ; 287 : G352 - G362
Watson WH, Zhao Y, Chawla RK . S-adenosylmethionine attenuates the lipopolysaccharide-induced expression of the gene for tumour necrosis factor alpha . Biochem J 1999 ; 342 : 21 - 25
Yamamoto Y, Yin MJ, Lin KM, Gaynor RB . Sulindac inhibits activation of the NF-kappaB pathway . J Biol Chem 1999 ; 274 : 27307 - 27314
Yin MJ, Yamamoto Y, Gaynor RB . The anti-inflammatory agents aspirin and salicylate inhibit the activity of IκB kinase β . Nature 1998 ; 396 : 77 - 80
Acknowledgements
This study was supported by grant No. 03-2003-009 from the Seoul National University Hospital Research Fund and by grant No. 03-2005-5 from the Seoul National University Boramae Medical Center Research Fund.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Moon, M., Kim, M., Chung, S. et al. S-Adenosyl-L-methionine ameliorates TNFα-induced insulin resistance in 3T3-L1 adipocytes. Exp Mol Med 42, 345–352 (2010). https://doi.org/10.3858/emm.2010.42.5.036
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3858/emm.2010.42.5.036
Keywords
This article is cited by
-
Oral administration of Blautia wexlerae ameliorates obesity and type 2 diabetes via metabolic remodeling of the gut microbiota
Nature Communications (2022)
-
β-Arrestins: multifunctional signaling adaptors in type 2 diabetes
Molecular Biology Reports (2011)
-
Apelin stimulates glucose uptake through the PI3K/Akt pathway and improves insulin resistance in 3T3-L1 adipocytes
Molecular and Cellular Biochemistry (2011)