Abstract
Myotonic dystrophy is an autosomal dominant, multisystem disorder that is characterized by myotonic myopathy. The symptoms and severity of myotonic dystrophy type l (DM1) ranges from severe and congenital forms, which frequently result in death because of respiratory deficiency, through to late-onset baldness and cataract. In adult patients, cardiac conduction abnormalities may occur and cause a shorter life span. In subsequent generations, the symptoms in DM1 may present at an earlier age and have a more severe course (anticipation). In myotonic dystrophy type 2 (DM2), no anticipation is described, but cardiac conduction abnormalities as in DM1 are observed and patients with DM2 additionally have muscle pain and stiffness. Both DM1 and DM2 are caused by unstable DNA repeats in untranslated regions of different genes: A (CTG)n repeat in the 3'-UTR of the DMPK gene and a (CCTG)n repeat in intron 1 of the CNBP (formerly ZNF9) gene, respectively. The length of the (CTG)n repeat expansion in DM1 correlates with disease severity and age of onset. Nevertheless, these repeat sizes have limited predictive values on individual bases. Because of the disease characteristics in DM1 and DM2, appropriate molecular testing and reporting is very important for the optimal counseling in myotonic dystrophy. Here, we describe best practice guidelines for clinical molecular genetic analysis and reporting in DM1 and DM2, including presymptomatic and prenatal testing.
Similar content being viewed by others
Myotonic dystrophy types 1 and 2
Myotonic dystrophy (DM) is an autosomal dominant, multisystem disorder that is characterized by myotonic myopathy with associated abnormalities in other organs.1 DM is clinically heterogeneous, and at least two types can be distinguished: DM type 1 (DM1; Steinert disease), which can be subdivided into four clinical subgroups, and DM type 2 (DM2; proximal myotonic myopathy (PROMM), or Ricker syndrome). DM1 is the most common form of muscular dystrophy in adults with an estimated incidence of 1:8000.2 The incidence for DM2 seems to be much lower than for DM1 and appears to be population dependent, reaching a higher incidence in Germany and Finland3 with an incidence similar to that of DM1. Despite the comparable core diagnostic features and multi-organ involvement, there are specific clinical features of DM1 and DM2, which enable them to be distinguished.
Myotonic dystrophy type 1
There is a wide range of symptoms in DM1, ranging from mild features, such as baldness and cataracts, to very severe features including muscle weakness and cardiac conduction defects. Many of the severely affected patients die of cardiac failure or respiratory distress. The cardiac involvement in DM1 may range between asymptomatic ECG abnormalities to sudden cardiac death.4
From a clinical perspective DM1 may be classified into four different subtypes: (i) mild; (ii) classical; (iii) juvenile and (iv) congenital types: (i) mild DM1: mildly symptomatic patients may have premature cataracts and baldness as the sole clinical features. A late-onset myopathy may develop and myotonia may only be detectable by electromyography. Cardiac conduction abnormalities may arise and result in a shorter life span. (ii) Classical or adult-onset DM1: The age of onset is typically in the second or third decade of life. The most frequent symptoms are distal weakness, involving the long finger flexors of the arms and the dorsiflexors of the legs, leading to symptoms relating to the strength of hand grasping and an increased incidence of stumbling. In addition patients may have cataracts, baldness and cardiac conduction abnormalities as in minimal DM1. Clinical myotonia, gastro-intestinal symptoms and fatigue may also occur. Apathy, lack of initiative, daytime sleepiness and experience fatigue can be distressing to DM1-patients, their partners and other family members. These features have a significant impact on quality of life. (iii) Juvenile DM1: this form resembles the classical form of myotonic dystrophy. However, it is more clearly associated with cognitive and behavioral abnormalities, for example, difficulties in learning and socialization at school. Muscle involvement may be minimal in the juvenile presentation. (iv) Congenital DM1. Polyhydramnios and poor fetal movements precede the birth of an infant with congenital DM1. The affected parent is nearly always the mother and congenital DM1 occurs in a quarter of offspring of affected DM1 mothers.2 The infant is typically a floppy baby with failure to thrive and severe respiratory distress. Facial and jaw muscles are weak and produce a tented upper lip with difficulties in feeding and suckling. Clubfeet and joint contractures are also common. Myotonia is typically absent clinically, and may be difficult to detect initially even on EMG. Mental retardation and developmental delay are common. There is a high mortality rate in the perinatal period. Muscle biopsies of patients with congenital DM1 may reveal only variability in fiber size and centralization of nuclei. However, none of the characteristics found in muscle biopsies of patients with classical or adult-onset DM1 myotonic dystrophy are present. Therefore, in order to confirm a clinical suspicion of congenital DM1, the diagnosis can only be established by DNA analysis.1, 2
Myotonic dystrophy type 2
The DM2 phenotype has some similarities to that of DM1 (myotonia, cataract and muscle weakness), but also some differences. The typical clinical presentation of DM2 is that of predominantly proximal muscle weakness, with muscle pain but no atrophy (PROMM phenotype).5 Myotonia is variable (both clinically as well as on EMG). However, in its mildest form, DM2 can be difficult to recognize. Conversely, DM2 may also present with severe proximal muscle involvement with remarkable muscle wasting without clinical myotonia.6, 7 The mean age of onset for DM2 is in the third and fourth decades of life. Unlike DM1, no congenital form of DM2 has been described.
In summary, the clinical challenge in DM1 lies in supplying optimal care for this multisystem disease, whereas in DM2 it is also a diagnostic challenge to recognize the disease. Detection of a repeat expansion in the DMPK gene and the CNBP gene is fast, inexpensive and reliable. Both the clinical sensitivity and the clinical specificity are >99% (Eurogentest clinical utility gene cards: http://www.eurogentest.org).
Molecular genetic defects in myotonic dystrophy types 1 and 2
DM1 (MIM #160900) is caused by a (CTG)n repeat expansion in the 3'-untranslated region of the DMPK gene located within chromosome band 19q13.3.8, 9, 10 DM2 (MIM #602668) is caused by a large (CCTG)n repeat expansion in intron l of the CNBP (formerly known as ZNF9) gene, at chromosome 3q21.11, 12, 13
The number of CTG-repeat units in stable nonpathogenic DMPK alleles ranges from 5 to 35.1, 14 (In previous publications, we have used 37 as limit of the largest normal allele size. Because of the use of 35 by other bodies, such as ‘The International Myotonic Dystrophy Consortium (IDMC),1 this was adapted for congruity. As repeats of 36 and 37 have not been detected in a large group of individuals,14 this adaptation is not anticipated to have any consequences for past or future reporting). DMPK alleles containing over 35 CTG-repeat units demonstrate a length-dependent risk of instability on transmission. Alleles containing a CTG-repeat with a length of 51–150 may be either asymptomatic or may give rise to minimal or classical DM1. A more severe DM1 phenotype is associated with DMPK alleles with sizes >150 CTG-repeat units. An overview of the general phenotype–genotype correlation1, 14, 15, 16 is provided in Table 1.
Thus, the severity of the disease and the age of onset is correlated with the number of CTG-repeat units within the expanded DNA sequence, although the individual variability is high.15, 16, 17, 18, 19 An increasing number of CTG-repeat units within the DMPK gene in successive generations in a family is associated with an earlier onset of the disorder. This phenomenon is termed ‘anticipation’.20 For DM1 parental sex has an important role in determining the level of intergenerational instability once the repeat size exceeds 150 repeats.15 Congenital DM1 is almost exclusively transmitted by the mother, a phenomenon that is not well understood.
In less than 5% of DM1 patients, the otherwise pure CTG-repeat tract is interrupted by GGC, CCG or CTC repeats.21, 22 Despite the reported unusual disease characteristics in some of these patients, the clinical relevance of such interruption remains to be established.
The CNBP gene harbors a complex-repeat tract (TG)n(TCTG)n(CCTG)n, and expansions of the CCTG-repeat array within this complex repeat are the cause of DM2.11 Nonpathogenic alleles contain to 26 CCTG-repeat units. In DM2 patients, the range of repeat units is extremely wide, ranging from about 75 to over 11 000 units, with a mean of 5000.11 Unlike in DM1, no significant association between the age of onset and the number of CCTG-repeat unit has been detected in DM2.23 Likewise, anticipation has not been observed in DM2 families.
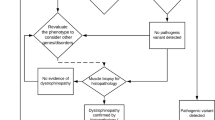
Molecular diagnostic analysis in DM1 and DM2
The first step in the molecular analysis of DM1 or DM2 is to analyze whether an individual has two alleles with a low number of repeats that can easily be detected by conventional PCR and fragment-length analysis. If only one allele size is detected, subsequent techniques such as repeat-primed PCR and or Southern blotting of genomic DNA or long-range PCR products are used to detect possible repeat expansions. PCR primers used by different laboratories are listed in Table 2.
In prenatal DM1 testing, analysis of DNA from the mother is also required to exclude maternal contamination in the fetal samples. The combination of these data allows interpretation and reporting. The delivery of diagnostic molecular genetics services for pre/post-natal DM requires in-house validation, on-going quality control processes and the supervision by a clinical molecular geneticist.
Molecular diagnostic tests in DM1
PCR and fragment-length analysis
In the lower range of DM1 expansions, the best analytical method is PCR. Alleles containing between 5 and 100 to 125 CTG-repeat units can be detected and characterized using synthetic fluorescently-labeled primers flanking the CTG-repeat region, followed by direct analysis of the length of the amplified products by capillary electrophoresis.
Triplet-repeat Primed (TP)-PCR
The TP-PCR is a PCR with three primers, where one lies outside the repeat, one within the repeat (which is added in limiting amounts) that also has a sequence-tail complementary to the third, universal, and fluorescently-labeled primer.24 This will result in a mixture of PCR fragments of different sizes that can be analyzed by capillary electrophoresis. Although expansions in all size ranges can be detected by TP-PCR, no reliable information about the length of the expanded repeat will be obtained because of extinction of the signal in the higher size region. In DM1 expansion analysis (see Figure 1a for an example), the TP-PCR has proven to be an accurate technique. Nevertheless, the presence of rare interruptions of the otherwise pure CTG repeat21, 22 may result in aberrant patterns or failure to detect expansions with TP-PCR.25 Therefore, to exclude false-negative reports, an additional test (such as a TP-PCR at the other repeat end or Southern blotting) is strongly recommended.
Molecular diagnostic tests in myotonic dystrophy type 1. (a) Fragment-length analysis of TP-PCR products of the CTG repeat in the DMPK gene. Fluorescently-labeled PCR products of a healthy individual having two normal alleles (5 and 13 CTG repeats; top panel) and an affected individual (bottom panel) with one normal allele (5 repeats) and one expanded allele were separated by capillary electrophoresis. (b) Southern blotting of long-range PCR-products of the CTG repeat in the DMPK gene. Long-range PCR fragments were subjected to to agarose gel electophoresis and capillary transfer to a nylon membrane. Subsequently, the membrane was hybridized with a labeled (CAG)5 probe. Visualized repeats were from a healthy control (lane 2), a patient with a heterozygous expansion in the size range of 51–150 repeats (lane 1), and a patient with a heterozygous expansion in the size range over 150 repeats (lane 3). (c) Southern blotting of genomic DNA probed for the DMPK gene. Genomic DNA samples were fragmented using BglI and subjected to electophoresis and capillary blotting to a nylon membrane. Subsequently, the membrane was hybridized with a 32P-labeled probe for the DMPK gene. Visualized fragments were from a healthy control (1), and patients with one normal and one expanded allele (2 and 3). Estimation of the lengths of the repeat expansions can be done using the size markers (M). Normal sized alleles and expansions are indicated.
Southern blotting of long-range PCR-products
This Southern blot method (for an example, see Figure 1b) requires precise PCR conditions to ensure the amplification of larger fragments, monitored by using appropriate positive and negative (healthy) controls, followed by probing with an end-labeled (CTG)x (where x is 5 or more) probe.26 On these Southern blots, expanded DMPK alleles will appear in virtually all cases as smears or multiple fragments, which is (in part) owing to somatic heterogeneity. Also, as with the TP-PCR, interrupted repeats may remain undetectable under certain conditions. Therefore, conditions that ensure the amplification of CG-rich and long templates is recommended.
Southern blotting of genomic DNA
Southern blotting of genomic DNA (for an example, see Figure 1c), digested with an appropriate restriction enzyme (EcoRI, BamHI, NcoI or BglI), has been the gold standard for the detection of DMPK alleles containing 100 CTG-repeat units and over since the identification of the DMPK gene.8 Several different probes have been reported for hybridization.8, 9, 27, 28 However, this procedure is time-consuming and has been replaced by TP-PCR and/or by Southern blotting of long-range PCR-products in most diagnostic centers.
Indirect DNA analysis
Though direct detection of expansions in the DMPK gene is the most straightforward technique, indirect DNA analysis is useful in some cases where confirmation of result is required by a separate technique. Polymorphic markers surrounding the DMPK gene that can be used for such an indirect analysis are outlined in Table 3.
Molecular diagnostic tests in DM2
Owing to the extreme expansion sizes (to >40 kb) that may be present in DM2 patients,11 it is important to ensure isolation of high molecular weight DNA from peripheral blood.
PCR and fragment-length analysis
In the lower range of DM2 repeat sizes, the best analytical method is PCR. Such alleles can be detected and characterized using synthetic, fluorescently-labeled primers flanking the complex-repeat region (also known as the CL3N58 repeat marker), followed by direct sizing of the amplified products by capillary electrophoresis. Because of the variability of the different units of this complex (TG)n(TCTG)n(CCTG)n, repeat, the precise length of the pathogenic CCTG unit within this repeat can only be determined by sequencing. However, as the difference between normal alleles (repeats to 26 CCTG units) and disease-associated alleles (75 units and more, with a mean of 5000) almost always is evident, exact sizing is not routinely performed.
Quadruplet-repeat primed (QP)-PCR
This technique, is similar to the TP-PCR used for DM1, but now with a primer in the tetra-nucleotide repeat;29 see Figure 2a for an example). In theory, the QP-PCR will detect expansions independent of their lengths. Although expansions in all size ranges can in theory be detected by QP-PCR, no reliable information about the length of the expanded repeat will be obtained because of extinction of the signal in the higher size region.
Molecular diagnostic tests in myotonic dystrophy type 2. (a) Fragment-length analysis of QP-PCR products of the CCTG repeat in the CNBP gene. Fluorescently-labeled PCR products of a healthy individual (top panel) and an affected individual (bottom panel) with one normal allele and one expanded allele (>100 repeats) were separated by capillary electrophoresis. (b) Southern blotting of long-range PCR products of the CCTG repeat in the CNBP gene. Long-range PCR fragments were subjected to agarose gel electophoresis and capillary transfer to a nylon membrane. Subsequently, the membrane was hybridized with an end-labeled (CAGG)5 probe. Visualized repeats were from a healthy compound heterozygous individual (lane 1) and a patients (lane 2) with one normal and one expanded allele. (c) Southern blotting of genomic DNA probed for the CNBP gene. Genomic DNA samples were fragmented using EcoRI and subjected to electophoresis and capillary blotting to a nylon membrane. Subsequently, the membrane was hybridized with a mixture of the CL3N58 (within the CNBP gene) and a reference probe (both 32P-labeled). Visualized fragments were from a healthy control (lane 1), and patients with one normal and one expanded allele (lanes 2 and 3). Note the decrease in intensity of the normal allele in the patients with an expansion, when compared with the control probe signal. Normal sized CNPB alleles and expansions, as well as the reference probe, are indicated.
Southern blotting of long-range PCR-products
This Southern blot method (see Figure 2b for an example) requires precise PCR conditions to ensure the amplification of larger fragments, monitored by using appropriate positive and negative (healthy) controls. Southern blotting of these fragments is followed by probing with an end-labeled (CCTG)5or6 probe.30 On these Southern blots, expanded CNBP alleles will appear in virtually all cases as smears or multiple fragments, which is (in part) due to somatic heterogeneity. Using this technique in DM2 diagnostics, one should be aware of false positives, probably occurring because of background signals of total genomic DNA after overexposure of the blot signals. Therefore, negative controls (healthy subjects) should be free of smears on the blot.
Southern blotting of genomic DNA
Direct analysis by Southern blotting (for an example, see Figure 2c), after digestion of genomic DNA with EcoRI, TaqI or alternative enzymes, followed by hybridization using the CL3N58 probe, was the first method used for studying the DM2 mutation.11 Owing to the extreme size of the expansion and the level of somatic instability as well as cross hybridizations of the probe, only a very heterogeneous smear can be visualized, limiting the sensitivity of this test to ∼80% of known carriers when applied without additional precautions. Several improvements of the basic protocol can be adopted in order to minimize possible sources of false-negative results, such as the inclusion of a reference probe to allow quantification of the non-expanded alleles.31 However, like in DM1 analysis, this procedure is time-consuming and has been replaced by QP-PCR or by Southern blotting of long-range PCR-products by most diagnostic laboratories.
Interpretation and Reporting
Myotonic dystrophy type 1
There is an association between CTG-repeat size and the age of onset and severity of symptoms (see Table 1). However, there is a large inter-individual variability and one should be very cautious with phenotypic predictions in individual cases.15, 16, 17, 18, 19 Therefore, the boundaries in repeat lengths of the four groups in Table 1 should not be interpreted too rigidly.
In DM, it is sufficient to report the size range of a repeat rather than an exact repeat length, that is, ‘a normal (CTG)n repeat length of (n=5–35)’ or ‘a large expansion of the (CTG)n repeat (n >150)’. Whenever an exact allele size is reported it should be accompanied by a statement of the testing laboratory’s uncertainty of measurement in the sizing.
For the reporting of exact normal allele sizes (n=5–35), which are determined by PCR and subsequent capillary electrophoresis, internal control standards from sequence-verified samples should be included. Alternatively, exact allele sizes can be obtained by DNA sequence analysis. For larger expansions, a range of repeat lengths should be estimated based on comparisons with molecular weight standards and/or characterized control samples.
The result of the appropriate tests will lead to one of the following possibilities and recommended reporting guidelines. (i) No expansion (homozygous or compound heterozygous for a normal allele size): This excludes the diagnosis of DM1. When it concerns a fetus, it is not affected. (ii) A heterozygous expansion in the size range of 36–50 repeats: (a) This excludes the diagnosis of DM1; when it concerns a fetus, it is not affected, (b) repeats of this length may or may not expand in future generations and (c) relatives (including offspring) of the counselee may be at risk of developing DM1. Therefore, they should be offered counseling. An offer of repeat-length analysis to those relatives at risk is warranted. (iii) A heterozygous expansion in the size range of 51–150 repeats: (a) when symptoms are evident, the diagnosis of DM1 is confirmed, (b) When symptoms of DM1 are not evident (asymptomatic family member or fetus), the individual is at risk of developing DM1, although individuals with a repeat expansion of this size may also remain symptomless, (c) counselees of reproductive age, and especially women, are at risk of having children with the congenital form of DM1 and (d) relatives (including offspring) of the counselee may be at risk of developing DM1. Owing to anticipation in DM1, offspring may be more severely affected. Relatives should therefore be offered counseling. An offer of repeat-length analysis to those relatives is warranted. (iv) A heterozygous expansion with a size over 150 repeats: (a) when symptoms are evident, the diagnosis of DM1 is confirmed, (b) when symptoms of DM1 are not evident (asymptomatic family member), the individual is at risk of developing DM1, although individuals with a repeat expansion of this size may also remain symptomless, (c) when it concerns a fetus, it is very likely to be affected and has a high risk to be more severely affected than the affected parent, (d) counselees in the reproductive age, and especially women, are at risk of having children with the congenital form of DM1 and (e) relatives (including offspring) of the counselee may be at risk of developing DM1. Because of anticipation in DM1, the offspring may be more severely affected. Therefore, relatives should be offered counseling. An offer of repeat-length analysis to those relatives is warranted.
A tabular format of the reporting guidelines is available as Supplementary Data.
Myotonic dystrophy type 2
As variability in the sizes of the different subunits of the complex repeat, (TG)n(TCTG)n(CCTG)n, in the CNBP gene exists, the precise length of the pathogenic CCTG unit within this repeat can only be determined by DNA sequencing. However, as repeats to 26 CCTG units are considered to be nonpathogenic, whereas disease-associated alleles contain 75 units and more, exact repeat sizing is not routinely performed. Experiences from several laboratories suggest that most frequently encountered pathogenic repeats reside in the high range (with a mean of 5000 repeats). Alleles between 26 and 75 CCTG units (gray area) are very rare but have been reported.32 Their clinical relevance, however, is not well characterized.
If reporting allele sizes (rather than a range), estimates of the uncertainty of measurement in the sizing in the testing laboratory should be reported. For allele sizes determined by PCR and subsequent capillary electrophoresis, a deviation of two repeat units may be present. For (larger) expansions, a range of repeat lengths should be estimated on basis of markers and/or control samples. However, in the reports of most laboratories the exact length of the repeat is not specified. The result of appropriate testing leads to one of the following possibilities and subsequent recommended reporting guidelines: (i) no expansion (homozygous or compound heterozygous for a normal allele size): this excludes the diagnosis of DM2. (ii) A heterozygous expansion of over 75 CCTG-repeat units: (a) the diagnosis of DM2 is confirmed and (b) relatives (including offspring) of the counselee may be at risk of developing DM2. Therefore, they should be offered counseling. An offer of repeat-length analysis to those relatives is warranted.
A tabular format of the reporting guidelines is available as Supplementary Data.
References
The International Myotonic Dystrophy Consortium (IDMC). New nomenclature and DNA testing guidelines for myotonic dystrophy type 1 (DM1). Neurology 2000; 54: 1218–1221.
Harper PS : Myotonic Dystrophy, 3rd edn, Harcourt Publishers Ltd: London, 2001.
Suominen T, Bachinski LL, Auvinen S et al. Population frequency of myotonic dystrophy: higher than expected frequency of myotonic dystrophy type 2 (DM2) mutation in Finland. Eur J Hum Genet 2011; 19: 776–782.
Groh WJ, Groh MR, Saha C et al. Electrocardiographic abnormalities and sudden death in myotonic dystrophy type 1. N Engl J Med 2008; 358: 2688–2697.
Day JW, Roelofs R, Leroy B, Pech I, Benzow K, Ranum LP : Clinical and genetic characteristics of a five-generation family with a novel form of myotonic dystrophy (DM2). Neuromuscul Disord 1999; 9: 19–27.
Udd B, Krahe R, Wallgren-Pettersson C, Falck B, Kalimo H : Proximal myotonic dystrophy--a family with autosomal dominant muscular dystrophy, cataracts, hearing loss and hypogonadism: heterogeneity of proximal myotonic syndromes? Neuromuscul Disord 1997; 7: 217–228.
Rotondo G, Sansone V, Cardani R et al. Proximal myotonic dystrophy mimicking progressive muscular atrophy. Eur J Neurol 2005; 12: 160–161.
Brook JD, McCurrach ME, Harley HG et al. Molecular basis of myotonic dystrophy: expansion of a trinucleotide (CTG) repeat at the 3' end of a transcript encoding a protein kinase family member. Cell 1992; 68: 799–808.
Fu YH, Pizzuti A, Fenwick RG et al. An unstable triplet repeat in a gene related to myotonic muscular dystrophy. Science 1992; 255: 1256–1258.
Mahadevan M, Tsilfidis C, Sabourin L et al. Myotonic dystrophy mutation: an unstable CTG repeat in the 3' untranslated region of the gene. Science 1992; 255: 1253–1255.
Liquori CL, Ricker K, Moseley ML et al. Myotonic dystrophy type 2 caused by a CCTG expansion in intron 1 of ZNF9. Science 2001; 293: 864–867.
Ricker K, Grimm T, Koch MC et al. Linkage of proximal myotonic myopathy to chromosome 3q. Neurology 1999; 52: 170–171.
Ranum LP, Rasmussen PF, Benzow KA, Koob MD, Day JW : Genetic mapping of a second myotonic dystrophy locus. Nat Genet 1998; 19: 196–198.
Martorell L, Monckton DG, Sanchez A, Lopez De MA, Baiget M : Frequency and stability of the myotonic dystrophy type 1 premutation. Neurology 2001; 56: 328–335.
Lavedan C, Hofmann-Radvanyi H, Shelbourne P et al. Myotonic dystrophy: size- and sex-dependent dynamics of CTG meiotic instability, and somatic mosaicism. Am J Hum Genet 1993; 52: 875–883.
Hamshere MG, Harley H, Harper P, Brook JD, Brookfield JF : Myotonic dystrophy: the correlation of (CTG) repeat length in leucocytes with age at onset is significant only for patients with small expansions. J Med Genet 1999; 36: 59–61.
Ashizawa T, Dubel JR, Dunne PW et al. Anticipation in myotonic dystrophy. II. Complex relationships between clinical findings and structure of the GCT repeat. Neurology 1992; 42: 1877–1883.
Harley HG, Rundle SA, MacMillan JC et al. Size of the unstable CTG repeat sequence in relation to phenotype and parental transmission in myotonic dystrophy. Am J Hum Genet 1993; 52: 1164–1174.
Gennarelli M, Novelli G, Andreasi BF et al. Prediction of myotonic dystrophy clinical severity based on the number of intragenic [CTG]n trinucleotide repeats. Am J Med Genet 1996; 65: 342–347.
Howeler CJ, Busch HF, Geraedts JP, Niermeijer MF, Staal A : Anticipation in myotonic dystrophy: fact or fiction? Brain 1989; 112 (Pt 3): 779–797.
Musova Z, Mazanec R, Krepelova A et al. Highly unstable sequence interruptions of the CTG repeat in the myotonic dystrophy gene. Am J Med Genet A 2009; 149A: 1365–1374.
Braida C, Stefanatos RK, Adam B et al. Variant CCG and GGC repeats within the CTG expansion dramatically modify mutational dynamics and likely contribute toward unusual symptoms in some myotonic dystrophy type 1 patients. Hum Mol Genet 2010; 19: 1399–1412.
Ranum LP, Day JW : Myotonic dystrophy: clinical and molecular parallels between myotonic dystrophy type 1 and type 2. Curr Neurol Neurosci Rep 2002; 2: 465–470.
Warner JP, Barron LH, Goudie D et al. A general method for the detection of large CAG repeat expansions by fluorescent PCR. J Med Genet 1996; 33: 1022–1026.
Radvansky J, Ficek A, Minarik G, Palffy R, Kadasi L : Effect of unexpected sequence interruptions to conventional PCR and repeat primed PCR in myotonic dystrophy type 1 testing. Diagn Mol Pathol 2011; 20: 48–51.
Gennarelli M, Pavoni M, Amicucci P, Novelli G, Dallapiccola B : A single polymerase chain reaction-based protocol for detecting normal and expanded alleles in myotonic dystrophy. Diagn Mol Pathol 1998; 7: 135–137.
Buxton J, Shelbourne P, Davies J et al. Detection of an unstable fragment of DNA specific to individuals with myotonic dystrophy. Nature 1992; 355: 547–548.
Shelbourne P, Davies J, Buxton J et al. Direct diagnosis of myotonic dystrophy with a disease-specific DNA marker. N Engl J Med 1993; 328: 471–475.
Day JW, Ricker K, Jacobsen JF et al. Myotonic dystrophy type 2: molecular, diagnostic and clinical spectrum. Neurology 2003; 60: 657–664.
Schoser BG, Kress W, Walter MC, Halliger-Keller B, Lochmuller H, Ricker K : Homozygosity for CCTG mutation in myotonic dystrophy type 2. Brain 2004; 127: 1868–1877.
Jakubiczka S, Vielhaber S, Kress W et al. Improvement of the diagnostic procedure in proximal myotonic myopathy/myotonic dystrophy type 2. Neurogenetics 2004; 5: 55–59.
Bachinski LL, Czernuszewicz T, Ramagli LS et al. Premutation allele pool in myotonic dystrophy type 2. Neurology 2009; 72: 490–497.
Acknowledgements
These guidelines were established at the EMQN Best Practice Meeting, held at 31st of October 2008 in Nijmegen, The Netherlands. The authors would like to acknowledge EuroGentest for funding and all participants of the ‘EMQN Best Practice Meeting’ for fruitful discussions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on European Journal of Human Genetics website
Supplementary information
Rights and permissions
This work is licensed under the Creative Commons Attribution-NonCommercial-No Derivative Works 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Kamsteeg, EJ., Kress, W., Catalli, C. et al. Best practice guidelines and recommendations on the molecular diagnosis of myotonic dystrophy types 1 and 2. Eur J Hum Genet 20, 1203–1208 (2012). https://doi.org/10.1038/ejhg.2012.108
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejhg.2012.108
This article is cited by
-
Myotone Dystrophien, nichtdystrophe Myotonien und periodische Paralysen
DGNeurologie (2024)
-
Genetic characterization of primary lateral sclerosis
Journal of Neurology (2023)
-
Clinical score for early diagnosis of myotonic dystrophy type 2
Neurological Sciences (2023)
-
Proteinopathies associated to repeat expansion disorders
Journal of Neural Transmission (2022)
-
Parental repeat length instability in myotonic dystrophy type 1 pre- and protomutations
European Journal of Human Genetics (2020)