Abstract
Background/Objectives:
The primary aim was to examine the daily glycemic response to normal-protein (NP) vs higher-protein (HP) breakfasts in overweight adolescents who habitually skip breakfast (H-BS). The secondary aim examined whether the glycemic response to these meals differed in H-BS vs habitual breakfast consumers (H-BC).
Subjects/Methods:
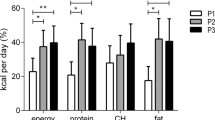
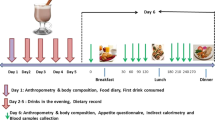
Thirty-five girls (age: 19±1 year; body mass index: 28.4±0.7 kg/m2) participated in the semi-randomized crossover-design study. The participants were grouped according to habitual breakfast frequency. H-BS (n=20) continued to skip breakfast (BS) or consumed a NP (12 g protein) or HP (32 g protein) breakfast for 3 days, whereas the H-BC (n=15) completed the NP and HP breakfast conditions for 3 days. On day 4 of each pattern, an 8 h testing day was completed. The respective breakfast and a standard lunch meal were provided, and plasma was collected to assess morning, afternoon, and total glucose and insulin area under the curves (AUC).
Results:
In H-BS, the addition of a HP breakfast increased total glucose AUC vs BS (P<0.05), whereas NP breakfast increased total insulin AUC vs BS (P<0.05). In H-BC, the HP breakfast reduced morning, afternoon and total glucose AUCs vs NP (all, P<0.05). No differences in insulin were detected. When comparing the HP–NP differential glycemic responses between groups, H-BS experienced greater afternoon and total glucose AUCs following HP vs NP breakfasts (both, P<0.05). No differences in insulin responses were observed between groups.
Conclusions:
Novel differences in the glucose response to HP vs NP breakfasts were observed and were influenced by the frequency of habitual breakfast consumption in overweight adolescents.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Deshmukh-Taskar P, Nicklas TA, Radcliffe JD, O'Neil CE, Liu Y . The relationship of breakfast skipping and type of breakfast consumed with overweight/obesity, abdominal obesity, other cardiometabolic risk factors and the metabolic syndrome in young adults. The National Health and Nutrition Examination Survey (NHANES): 1999–2006. Public Health Nutr 2012; 16: 1–10.
Mekary RA, Giovannucci E, Willett WC, van Dam RM, Hu FB . Eating patterns and type 2 diabetes risk in men: breakfast omission, eating frequency, and snacking. Am J Clin Nutr 2012; 95: 1182–1189.
Jovanovic A, Gerrard J, Taylor R . The second-meal phenomenon in type 2 diabetes. Diabetes Care 2009; 32: 1199–1201.
Jovanovic A, Leverton E, Solanky B, Ravikumar B, Snaar JE, Morris PG et al. The second-meal phenomenon is associated with enhanced muscle glycogen storage in humans. Clin Sci (Lond) 2009; 117: 119–127.
Arai H, Mizuno A, Sakuma M, Fukaya M, Matsuo K, Muto K et al. Effects of a palatinose-based liquid diet (Inslow) on glycemic control and the second-meal effect in healthy men. Metabolism 2007; 56: 115–121.
Jenkins DJ, Wolever TM, Nineham R, Sarson DL, Bloom SR, Ahern J et al. Improved glucose tolerance four hours after taking guar with glucose. Diabetologia 1980; 19: 21–24.
Jenkins DJ, Wolever TM, Taylor RH, Griffiths C, Krzeminska K, Lawrie JA et al. Slow release dietary carbohydrate improves second meal tolerance. Am J Clin Nutr 1982; 35: 1339–1346.
Blachier F, Mourtada A, Sener A, Malaisse WJ . Stimulus-secretion coupling of arginine-induced insulin release. Uptake of metabolized and nonmetabolized cationic amino acids by pancreatic islets. Endocrinology 1989; 124: 134–141.
Floyd JC Jr., Fajans SS, Pek S, Thiffault CA, Knopf RF, Conn JW . Synergistic effect of essential amino acids and glucose upon insulin secretion in man. Diabetes 1970; 19: 109–115.
Veldhorst MA, Nieuwenhuizen AG, Hochstenbach-Waelen A, Westerterp KR, Engelen MP, Brummer RJ et al. Comparison of the effects of a high- and normal-casein breakfast on satiety, 'satiety' hormones, plasma amino acids and subsequent energy intake. Br J Nutr 2009; 101: 295–303.
Belza A, Ritz C, Sorensen MQ, Holst JJ, Rehfeld JF, Astrup A . Contribution of gastroenteropancreatic appetite hormones to protein-induced satiety. Am J Clin Nutr 2013; 97: 980–989.
Blom WA, Lluch A, Stafleu A, Vinoy S, Holst JJ, Schaafsma G et al. Effect of a high-protein breakfast on the postprandial ghrelin response. Am J Clin Nutr 2006; 83: 211–220.
Acheson KJ, Blondel-Lubrano A, Oguey-Araymon S, Beaumont M, Emady-Azar S, Ammon-Zufferey C et al. Protein choices targeting thermogenesis and metabolism. Am J Clin Nutr 2011; 93: 525–534.
Al Awar R, Obeid O, Hwalla N, Azar S . Postprandial acylated ghrelin status following fat and protein manipulation of meals in healthy young women. Clin Sci 2005; 109: 405–411.
Ratliff J, Leite JO, de Ogburn R, Puglisi MJ, VanHeest J, Fernandez ML . Consuming eggs for breakfast influences plasma glucose and ghrelin, while reducing energy intake during the next 24 hours in adult men. Nutr Res 2010; 30: 96–103.
Agus MS, Swain JF, Larson CL, Eckert EA, Ludwig DS . Dietary composition and physiologic adaptations to energy restriction. Am J Clin Nutr 2000; 71: 901–907.
Leidy HJ, Armstrong CLH, Tang M, Mattes RD, Campbell WW . The influence of higher protein intake and greater eating frequency on appetite control in overweight and obese men. Obesity 2010; 18: 1725–1732.
Veldhorst MA, Nieuwenhuizen AG, Hochstenbach-Waelen A, Westerterp KR, Engelen MP, Brummer RJ et al. Effects of high and normal soyprotein breakfasts on satiety and subsequent energy intake, including amino acid and 'satiety' hormone responses. Eur J Nutr 2009; 48: 92–100.
Leidy HJ, Mattes RD, Campbell WW . Effects of acute and chronic protein intake on metabolism, appetite, and ghrelin during weight loss. Obesity 2007; 15: 1215–1225.
Makris AP, Borradaile KE, Oliver TL, Cassim NG, Rosenbaum DL, Boden GH et al. The individual and combined effects of glycemic index and protein on glycemic response, hunger, and energy intake. Obesity (Silver Spring) 2011; 19: 2365–2373.
Clifton PM, Keogh JB, Noakes M . Long-term effects of a high-protein weight-loss diet. Am J Clin Nutr 2008; 87: 23–29.
Noakes M, Keogh JB, Foster PR, Clifton PM . Effect of an energy-restricted, high-protein, low-fat diet relative to a conventional high-carbohydrate, low-fat diet on weight loss, body composition, nutritional status, and markers of cardiovascular health in obese women. Am J Clin Nutr 2005; 81: 1298–1306.
Farshchi HR, Taylor MA, Macdonald IA . Deleterious effects of omitting breakfast on insulin sensitivity and fasting lipid profiles in healthy lean women. Am J Clin Nutr 2005; 81: 388–396.
Rodriguez G, Moreno L, Sarria A, Fleta J, Bueno M . Resting energy expenditure in children and adolescents: agreement between calorimetry and prediction equations. Clin Nutr 2002; 21: 255–260.
Standl E, Schnell O, Ceriello A . Postprandial hyperglycemia and glycemic variability: should we care? Diabetes Care 2011; 34 (Suppl 2), S120–S127.
Marfella R, Verrazzo G, Acampora R, La Marca C, Giunta R, Lucarelli C et al. Glutathione reverses systemic hemodynamic changes induced by acute hyperglycemia in healthy subjects. Am J Physiol 1995; 268: E1167–E1173.
Lorenzo C, Okoloise M, Williams K, Stern MP, Haffner SM . The metabolic syndrome as predictor of type 2 diabetes: the San Antonio heart study. Diabetes Care 2003; 26: 3153–3159.
Smith NL, Barzilay JI, Shaffer D, Savage PJ, Heckbert SR, Kuller LH et al. Fasting and 2-hour postchallenge serum glucose measures and risk of incident cardiovascular events in the elderly: the Cardiovascular Health Study. Arch Intern Med 2002; 162: 209–216.
Meigs JB, Nathan DM, D'Agostino RB Sr., Wilson PW . Fasting and postchallenge glycemia and cardiovascular disease risk: the Framingham Offspring Study. Diabetes Care 2002; 25: 1845–1850.
Qiao Q, Pyorala K, Pyorala M, Nissinen A, Lindstrom J, Tilvis R et al. Two-hour glucose is a better risk predictor for incident coronary heart disease and cardiovascular mortality than fasting glucose. Eur Heart J 2002; 23: 1267–1275.
Brunner EJ, Shipley MJ, Witte DR, Fuller JH, Marmot MG . Relation between blood glucose and coronary mortality over 33 years in the Whitehall Study. Diabetes Care 2006; 29: 26–31.
Pereira MA, Erickson E, McKee P, Schrankler K, Raatz SK, Lytle LA et al. Breakfast frequency and quality may affect glycemia and appetite in adults and children. J Nutr 2011; 141: 163–168.
Reaven G . Age and glucose intolerance: effect of fitness and fatness. Diabetes Care 2003; 26: 539–540.
Reaven GM, Chen N, Hollenbeck C, Chen YD . Effect of age on glucose tolerance and glucose uptake in healthy individuals. J Am Geriatr Soc 1989; 37: 735–740.
Dangin M, Boirie Y, Garcia-Rodenas C, Gachon P, Fauquant J, Callier P et al. The digestion rate of protein is an independent regulating factor of postprandial protein retention. Am J Physiol Endocrinol Metab 2001; 280: E340–E348.
Willett W, Manson J, Liu S . Glycemic index, glycemic load, and risk of type 2 diabetes. Am J Clin Nutr 2002; 76: 274S–280S.
Acknowledgements
We thank Laura Ortinau and Tyler Lasley for assisting with the glucose and insulin data collection. Regarding the author contributions: AYA and HJL designed the research, conducted the research and analyzed the data. AYA developed the first draft of the paper. AYA, JPT and HJL had primary responsibility for manuscript revisions and final content. All authors substantially contributed to the completion of the manuscript and all have read and approved the final manuscript. The Beef Checkoff and the Egg Nutrition Center supplied funds to complete the study but were not involved in the design, implementation, analysis or interpretation of data.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Alwattar, A., Thyfault, J. & Leidy, H. The effect of breakfast type and frequency of consumption on glycemic response in overweight/obese late adolescent girls. Eur J Clin Nutr 69, 885–890 (2015). https://doi.org/10.1038/ejcn.2015.12
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2015.12