Abstract
Background/objectives:
Most patients with Type 2 diabetes mellitus(DM) also have metabolic syndrome (MetS), which is associated with an increased risk of coronary heart disease prevalence. Limited information is available on the effect and effective doses of oat intake with a structured dietary intervention in metabolic control and cardiovascular disease (CVD) risk prevention with the population who has Type 2 DM and meets the MetS criteria.
Subjects/methods:
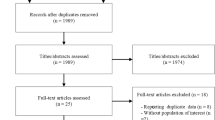
A total of 260 Type 2 DM patients meeting MetS National Cholesterol Education Program Adult Treatment Panel III criteria were selected from 445 patients between 50 and 65 years of age, and they participated in a single-blinded, 30-day centralized management of a dietary program in China. Participants in the program were randomly assigned into one of the four groups: usual care group (control group, only basic health advice), diet group (systematic diet plans and intensive education), 50 g-organic naked oat with whole germ group (ONOG) and 100 g-organic naked oat with whole germ group (daily ONOG replacement boiled into porridge based on diet group). The primary outcomes were the relative changes in glycosylated hemoglobin (HbA1c) and insulin resistance after a 30-day intervention among the four groups.
Results:
HbA1c decreased significantly with the increase in interventions (Ptrend<0.05). Similar results were also obtained in plasma glucose, serum lipid and hypersensitive C-reactive protein (hs-CRP). For the 100 g-ONOG group but not 50 g-ONOG group, HbA1c and hs-CRP reduced significantly by 0.51% and 1.29 mg/l (P<0.05, vs diet group), respectively. The 100 g-ONOG group showed a reduction by 0.22 U*mol/l2 in insulin resistance, compared with the 50 g-ONOG group (P=0.039).
Conclusions:
Compared with diet alone or no diet, 50–100 g/day ONOG supplement to structured dietary intervention, at a dose of 100 g/day especially, contributes to the Type 2 DM patients meeting MetS criteria in their metabolic control and CVD risk prevention, with external factors being controlled.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Yang W, Lu J, Weng J, Jia W, Ji L, Xiao J et al. Prevalence of diabetes among men and women in China. N Engl J Med 2010; 362: 1090–1101.
IDF. IDF Diabetes Atlas 5th edn. International Diabetes Federation: Brussels, Belgium, 2011.
American Diabetes Association. Economic costs of diabetes in the U.S. in 2007. Diabetes Care 2008; 31: 1–20.
Alexander CM, Landsman PB, Teutsch SM, Haffner SM . NCEP-defined metabolic syndrome, diabetes, and prevalence of coronary heart disease among NHANES III participants age 50 years and older. Diabetes 2003; 52: 1210–1214.
Tong PC, Kong AP, So WY, Yang X, Ho CS, Ma RC et al. The usefulness of the International Diabetes Federation and the National Cholesterol Education Program’s Adult Treatment Panel III definitions of the metabolic syndrome in predicting coronary heart disease in subjects with type 2 diabetes. Diabetes Care 2007; 30: 1206–1211.
Franz MJ, Powers MA, Leontos C, Holzmeister LA, Kulkarni K, Monk A et al. The evidence for medical nutrition therapy for type 1 and type 2 diabetes in adults. J Am Diet Assoc 2010; 110: 1852–1889.
Boucher JL, Evert A, Daly A, Kulkarni K, Rizzotto JA, Burton K et al. American Dietetic Association revised standards of practice and standards of professional performance for registered Dietitians (generalist, specialty, and advanced) in diabetes care. J Am Diet Assoc 2011; 111: 156–166.
de Munter JS, Hu FB, Spiegelman D, Franz M, van Dam RM . Whole grain, bran, and germ intake and risk of type 2 diabetes: a prospective cohort study and systematic review. PLoS Med 2007; 4: 1385–1395.
Sahyoun Nadine R, Jacques Paul F, Zhang Xinli L, Juan Wenyen, McKeown Nicola M . Whole-grain intake is inversely associated with the metabolic syndrome and mortality in older adults. Am J Clin Nutr 2006; 83: 124–131.
Esmaillzadeh A, Mirmiran P, Azizi F . Whole-grain consumption and the metabolic syndrome: a favorable association in Tehranian adults. Eur J Clin Nutr 2005; 59: 353–362.
Katcher Heather I, Legro Richard S, Kunselman Allen R, Gillies Peter J, Demers Laurence M, Bagshaw Deborah M et al. The effects of a whole grain-enriched hypocaloric diet on cardiovascular disease risk factors in men and women with metabolic syndrome. Am J Clin Nutr 2008; 87: 79–90.
U.S. Department of Agriculture, U.S. Department of Health and Human Services. Dietary Guidelines for Americans 2010. Available at http://www.cnpp.usda.gov/DGAs2010-PolicyDocument.html (accessed 16 December 2012).
The Elderly Nutrition Branch of Chinese Nutrition Society. Dietary guidelines for elderly Chinese. Shandong, 2010.
Masood SB, Muhammad TN, Muhammad KK, Rabia S, Mehmood SB . Oat: unique among the cereals. Eur J Nutr 2008; 47: 68–79.
Weickert MO, Möhlig M, Schöfl C, Arafat AM, Otto B, Viehoff H et al. Cereal fiber improves whole-body insulin sensitivity in overweight and obese women. Diabetes Care 2006; 29: 775–780.
Lammert A, Kratzsch J, Selhorst J, Humpert PM, Bierhaus A, Birck R et al. Clinical benefit of a short term dietary oatmeal intervention in patients with type 2 diabetes and severe insulin resistance: a pilot study. Exp Clin Endocrinol Diabetes 2008; 116: 132–134.
Cugnet-Anceau C, Nazare JA, Biorklund M, Le Coquil E, Sassolas A, Sothier M et al. A controlled study of consumption of beta-glucan-enriched soups for 2 months by type 2 diabetic free-living subjects. Br J Nutr 2010; 103: 422–428.
Wolever TM, Tosh SM, Gibbs AL, Brand-Miller J, Duncan AM, Hart V et al. Physicochemical properties of oat β-glucan influence its ability to reduce serum LDL cholesterol in humans: a randomized clinical trial. Am J Clin Nutr 2010; 92: 723–732.
Berg A, König D, Deibert P, Grathwohl D, Berg A, Baumstark MW et al. Effect of an oat bran enriched diet on the atherogenic lipid profile in patients with an increased coronary heart disease risk. A controlled randomized lifestyle intervention study. Ann Nutr Metab 2003; 47: 306–311.
Beck EJ, Tapsell LC, Batterham MJ, Tosh SM, Huang XF . Oat beta-glucan supplementation does not enhance the effectiveness of an energy-restricted diet in overweight women. Br J Nutr 2010; 103: 1212–1222.
National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Third Report of the National Cholesterol Education Program(NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 2002; 106: 3143–3421.
Institute of Nutrition and food safety. China CDC 2005 China Food Composition. Peking University Medical Press, 2004.
Chinese Nutrition Society. Dietary Guidelines for Chinese Residents (2011). Tibet People's Publishing House: Lhasa, 2010.
Chinese Diabetes Society. China Medical Nutrition Therapy Guideline for Diabetes 2010. Available at http://cdschina.org/news_show.jsp?id=509.html (accessed 16 December 2012).
Shi H, Lian CL . The relationship between hypersensitive C-reactive protein and disorders of lipid metabolism in patients with Type 2 diabetes mellitus. Chin Med J Metall Indus 2011; 28: 29–30.
Wen HH, Niu LJ . The relationship between serum adiponectin levels and insulin resistance in metabolic syndrome patients without diabetes. J Diseases Monitor Control 2012; 6: 65–67.
Bonora E, Targher G, Alberiche M, Bonadonna RC, Saggiani F, Zenere MB et al. Homeostasis model assessment closely mirrors the glucose clamp technique in the assessment of insulin sensitivity. Diabetes Care 2000; 23: 57–63.
International Diabetes Federation. The IDF consensus worldwide definition of the MS 2006.
Stratton IM, Adler AI, Neil HA, Matthews DR, Manley SE, Cull CA et al. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ 2000; 321: 405–412.
Kones Richard . Primary prevention of coronary heart disease:integration of new data, evolving views, revised goals, and role of rosuvastatin in management. A comprehensive survey. Drug Des Devel Ther 2011; 5: 325–380.
Braaten JT, Scott FW, Wood PJ, Riedel KD, Wolynetz MS, Brulé D et al. High beta-glucan oat bran and oat gum reduce postprandial blood glucose and insulin in subjects with and without type 2 diabetes. Diabet Med 1994; 11: 312–318.
Anderson JW, Hamilton CC, Horn JL, Spencer DB, Dillon DW, Zeigler JA . Metabolic effects of insoluble oat fiber on lean men with type II diabetes. Cereal Chem 1991; 68: 291–294.
Tapola N, Karvonen H, Niskanen L, Mikola M, Sarkkinen E . Glycemic responses of oat bran products in type 2 diabetic patients. Nutr Metab Cardiovasc Dis 2005; 15: 255–261.
Jenkins AL, Jenkins DJ, Zdravkovic U, Würsch P, Vuksan V . Depression of the glycemic index by high levels of beta-glucan fiber in two functional foods tested in type 2 diabetes. Eur J Clin Nutr 2002; 56: 622–628.
Pick ME, Hawrysh ZJ, Gee MI, Toth E, Garg ML, Hardin RT . Oat bran concentrate bread products improve long-term control of diabetes: a pilot study. J Am Diet Assoc 1996; 96: 1254–1261.
Othman RA, Moghadasian MH, Jones PJ . Cholesterol-lowering effects of oat β-glucan. Nutr Rev 2011; 69: 299–309.
Chen J, He J, Wildman RP, Reynolds K, Streiffer RH, Whelton PK . A randomized controlled trial of dietary fiber intake on serum lipids. Eur J Clin Nutr 2006; 60: 62–68.
Biorklund M, Holm J, Onning G . Serum lipids and postprandial glucose and insulin levels in hyperlipidemic subjects after consumption of an oat beta-glucan-containing ready meal. Ann Nutr Metab 2008; 52: 83–90.
Stefanick ML, Mackey S, Sheehan M, Ellsworth N, Haskell WL, Wood PD . Effects of diet and exercise in men and postmenopausal women with low levels of HDL cholesterol and high levels of LDL cholesterol. N Engl J Med 1998; 339: 12–20.
Wolever TM, Tosh SM, Gibbs AL, Brand-Miller J, Duncan AM, Hart V et al. Physicochemical properties of oat β-glucan influence its ability to reduce serum LDL cholesterol in humans:a randomized clinical trial. Am J Clin Nutr 2010; 92: 723–732.
Slavin JL, Jacobs D, Marquart L . Grain processing and nutrition. Crit Rev Food Sci Nutr 2000; 40: 309–326.
Maki KC, Beiseigel JM, Jonnalagadda SS, Gugger CK, Reeves MS, Farmer MV et al. Whole-grain ready-to-eat oat cereal, as part of a dietary program for weight loss, reduces low-density lipoprotein cholesterol in adults with overweight and obesity more than a dietary program including low-fiber control foods. J Am Diet Assoc 2010; 110: 205–214.
Acknowledgements
We thank the Beijing Nutrition Society and the Inner Mongolia Medical College, Third Hospital for their help with the study. We also thank the members of our volunteer team, Zhi Sun, Bin Cai, Fang Li, Guangling Hu, Hao Yu, Jianqin Chen, Junguo He, Meihong Yang, Peng Song, Peng Zhang, Xiping Xu, Yao Meng, Meiyan Liu, Yuxia Jin, Ying Li, Yaohui Jiang, Ya Zhang and Zhibin Sun for their excellent job with the study. We thank Yimin Zhu, Yongliang Zhang, Shuhua Zhao and Chuan Fan, Dewei Liang for editing the manuscript for English. This study was funded by Inner Mongolia Sanzhuliang Natural Oats Industry Corporation. The trial is registered at ClinicalTrials.gov, number NCT01495052. The sponsor of the study had no role in the study design, data collection, data analysis, data interpretation or writing of the report.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Author contributions
The authors’ responsibilities were described as follows: XM participated in the research design, conducted the research, performed the statistical analysis and wrote the manuscript; YL designed the research and had primary responsibility for final content. All authors participated in data acquisition and study supervision. All authors approved the final version of the report.
Supplementary Information accompanies this paper on European Journal of Clinical Nutrition website
Supplementary information
Rights and permissions
About this article
Cite this article
Ma, X., Gu, J., Zhang, Z. et al. Effects of Avena nuda L. on metabolic control and cardiovascular disease risk among Chinese patients with diabetes and meeting metabolic syndrome criteria: secondary analysis of a randomized clinical trial. Eur J Clin Nutr 67, 1291–1297 (2013). https://doi.org/10.1038/ejcn.2013.201
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2013.201
Keywords
This article is cited by
-
A comparison between whole grain and pearled oats: acute postprandial glycaemic responses and in vitro carbohydrate digestion in healthy subjects
European Journal of Nutrition (2020)