Abstract
BACKGROUND/OBJECTIVES:
Phytoestrogens are estradiol-like natural compounds found in plants that have been associated with protective effects against chronic diseases, including some cancers, cardiovascular diseases and osteoporosis. The purpose of this study was to estimate the dietary intake of phytoestrogens, identify their food sources and their association with lifestyle factors in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort.
SUBJECTS/METHODS:
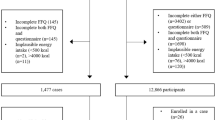
Single 24-hour dietary recalls were collected from 36 037 individuals from 10 European countries, aged 35–74 years using a standardized computerized interview programe (EPIC-Soft). An ad hoc food composition database on phytoestrogens (isoflavones, lignans, coumestans, enterolignans and equol) was compiled using data from available databases, in order to obtain and describe phytoestrogen intakes and their food sources across 27 redefined EPIC centres.
RESULTS:
Mean total phytoestrogen intake was the highest in the UK health-conscious group (24.9 mg/day in men and 21.1 mg/day in women) whereas lowest in Greece (1.3 mg/day) in men and Spain-Granada (1.0 mg/day) in women. Northern European countries had higher intakes than southern countries. The main phytoestrogen contributors were isoflavones in both UK centres and lignans in the other EPIC cohorts. Age, body mass index, educational level, smoking status and physical activity were related to increased intakes of lignans, enterolignans and equol, but not to total phytoestrogen, isoflavone or coumestan intakes. In the UK cohorts, the major food sources of phytoestrogens were soy products. In the other EPIC cohorts the dietary sources were more distributed, among fruits, vegetables, soy products, cereal products, non-alcoholic and alcoholic beverages.
CONCLUSIONS:
There was a high variability in the dietary intake of total and phytoestrogen subclasses and their food sources across European regions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others

References
Franke AA, Halm BM, Kakazu K, Li X, Custer LJ . Phytoestrogenic isoflavonoids in epidemiologic and clinical research. Drug Test Anal 2009; 1: 14–21.
Kuiper GG, Lemmen JG, Carlsson B, Corton JC, Safe SH, van der Saag PT et al. Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor beta. Endocrinology 1998; 139: 4252–4263.
US Departament of Agriculture. USDA Database for the Isoflavone Content of Selected Foods, 2008. (USDA: Beltsville, MD).
Ward HA, Kuhnle GG, Mulligan AA, Lentjes MA, Luben RN, KT Khaw . Breast colorectal, and prostate cancer risk in the European Prospective Investigation into Cancer and Nutrition-Norfolk in relation to phytoestrogen intake derived from an improved database. Am J Clin Nutr 2010; 91: 440–448.
Kuhnle GG, Dell'aquila C, Aspinall SM, Runswick SA, Mulligan AA, Bingham SA . Phytoestrogen content of foods of animal origin: dairy products, eggs, meat, fish, and seafood. J Agric Food Chem 2008; 56: 10099–10104.
Mazur W, Fotsis T, Wahala K, Ojala S, Salakka A, Adlercreutz H . Isotope dilution gas chromatographic-mass spectrometric method for the determination of isoflavonoids, coumestrol, and lignans in food samples. Anal Biochem 1996; 233: 169–180.
Milder IE, Arts IC, van de Putte B, Venema DP, Hollman PC . Lignan contents of Dutch plant foods: a database including lariciresinol, pinoresinol, secoisolariciresinol and matairesinol. Br J Nutr 2005; 93: 393–402.
Adlercreutz H . Lignans and human health. Crit Rev Clin Lab Sci 2007; 44: 483–525.
Heinonen S, Nurmi T, Liukkonen K, Poutanen K, Wahala K, Deyama T et al. In vitro metabolism of plant lignans: new precursors of mammalian lignans enterolactone and enterodiol. J Agric Food Chem 2001; 49: 3178–3186.
Schabath MB, Hernandez LM, Wu X, Pillow PC, Spitz MR . Dietary phytoestrogens and lung cancer risk. JAMA 2005; 294: 1493–1504.
Siow RC, Li FY, Rowlands DJ, de Winter P, Mann GE . Cardiovascular targets for estrogens and phytoestrogens: transcriptional regulation of nitric oxide synthase and antioxidant defense genes. Free Radic Biol Med 2007; 42: 909–925.
Mann GE, Rowlands DJ, Li FY, de Winter P, Siow RC . Activation of endothelial nitric oxide synthase by dietary isoflavones: role of NO in Nrf2-mediated antioxidant gene expression. Cardiovasc Res 2007; 75: 261–274.
Qin LQ, Xu JY, Wang PY, Hoshi K . Soyfood intake in the prevention of breast cancer risk in women: a meta-analysis of observational epidemiological studies. J Nutr Sci Vitaminol 2006; 52: 428–436.
Mulligan AA, Welch AA, McTaggart AA, Bhaniani A, Bingham SA . Intakes and sources of soya foods and isoflavones in a UK population cohort study (EPIC-Norfolk). Eur J Clin Nutr 2007; 61: 248–254.
Yamamoto S, Sobue T, Kobayashi M, Sasaki S, S Tsugane . Soy, isoflavones, and breast cancer risk in Japan. J Natl Cancer Inst 2003; 95: 906–913.
Peterson J, Lagiou P, Samoli E, Lagiou A, Katsouyanni K, La Vecchia C et al. Flavonoid intake and breast cancer risk: a case--control study in Greece. Br J Cancer 2003; 89: 1255–1259.
Akhter M, Iwasaki M, Yamaji T, Sasazuki S, Tsugane S . Dietary isoflavone and the risk of colorectal adenoma: a case-control study in Japan. Br J Cancer 2009; 100: 1812–1816.
Chan SG, Ho SC, Kreiger N, Darlington G, So KF, Chong PY . Dietary sources and determinants of soy isoflavone intake among midlife Chinese Women in Hong Kong. J Nutr 2007; 137: 2451–2455.
Milder IE, Feskens EJ, Arts IC, Bueno de Mesquita HB, Hollman PC, Kromhout D . Intake of the plant lignans secoisolariciresinol, matairesinol, lariciresinol, and pinoresinol in Dutch men and women. J Nutr 2005; 135: 1202–1207.
Cotterchio M, Boucher BA, Kreiger N, Mills CA, Thompson LU . Dietary phytoestrogen intake-lignans and isoflavones-and breast cancer risk (Canada). Cancer Causes Control 2008; 19: 259–272.
Riboli E, Kaaks R . The EPIC Project: rationale and study design. European Prospective Investigation into Cancer and Nutrition. Int J Epidemiol 1997; 26(Suppl 1): S6–14.
Riboli E, Hunt KJ, Slimani N, Ferrari P, Norat T, Fahey M et al. European Prospective Investigation into Cancer and Nutrition (EPIC): study populations and data collection. Public Health Nutr 2002; 5: 1113–1124.
Slimani N, Kaaks R, Ferrari P, Casagrande C, Clavel-Chapelon F, Lotze G et al. European Prospective Investigation into Cancer and Nutrition (EPIC) calibration study: rationale, design and population characteristics. Public Health Nutr 2002; 5: 1125–1145.
Slimani N, Ferrari P, Ocke M, Welch A, Boeing H, Liere M et al. Standardization of the 24-hour diet recall calibration method used in the european prospective investigation into cancer and nutrition (EPIC): general concepts and preliminary results. Eur J Clin Nutr 2000; 54: 900–917.
Slimani N, Deharveng G, Unwin I, Southgate DA, Vignat J, Skeie G et al. The EPIC nutrient database project (ENDB): a first attempt to standardize nutrient databases across the 10 European countries participating in the EPIC study. Eur J Clin Nutr 2007; 61: 1037–1056.
Haftenberger M, Schuit AJ, Tormo MJ, Boeing H, Wareham N, Bueno-de-Mesquita HB et al. Physical activity of subjects aged 50-64 years involved in the European Prospective Investigation into Cancer and Nutrition (EPIC). Public Health Nutr 2002; 5: 1163–1176.
Neveu V, Perez-Jimenez J, Vos F, Crespy V, du Chaffaut L, Mennen L et al. Phenol-Explorer: an online comprehensive database on polyphenol contents in foods. Database. (Oxford) 2010, bap024.
Thompson LU, Boucher BA, Liu Z, Cotterchio M, Kreiger N . Phytoestrogen content of foods consumed in Canada, including isoflavones, lignans, and coumestan. Nutr Cancer 2006; 54: 184–201.
Zamora-Ros R, Knaze V, Luján-Barroso L, Slimani N, Romieu I, Touillaud M et al. Estimation of the intake of anthocyanidins and their food sources in the European Prospective Investigation in to Cancer and Nutrition (EPIC) study. Br J Nutr 2011; 106: 1090–1099.
Zamora-Ros R, Knaze V, Luján-Barroso L, Slimani N, Romieu I, Fedirko V et al. Estimated dietary intakes of flavonols, flavanones and flavones in the European Prospective Investigation into Cancer and Nutrition (EPIC) 24-h dietary recall cohort. Br J Nutr 2011; 106: 1915–1925.
Horn-Ross PL, Barnes S, Lee M, Coward L, Mandel JE, Koo J et al. Assessing phytoestrogen exposure in epidemiologic studies: development of a database (United States). Cancer Causes Control 2000; 11: 289–298.
Kuhnle GG, Dell'aquila C, Aspinall SM, Runswick SA, Mulligan AA, Bingham SA . Phytoestrogen content of cereals and cereal-based foods consumed in the UK. Nutr Cancer 2009; 61: 302–309.
Keinan-Boker L, Peeters PH, Mulligan AA, Navarro C, Slimani N, Mattisson I et al. Soy product consumption in 10 European countries: the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Public Health Nutr 2002; 5: 1217–1226.
Boker LK, van der Schouw YT, De Kleijn MJ, Jacques PF, Grobbee DE, Peeters PH . Intake of dietary phytoestrogens by Dutch women. J Nutr 2002; 132: 1319–1328.
Hedelin M, Klint A, Chang ET, Bellocco R, Johansson JE, Andersson SO et al. Dietary phytoestrogen, serum enterolactone and risk of prostate cancer: the cancer prostate Sweden study (Sweden). Cancer Causes Control 2006; 17: 169–180.
Hedelin M, Lof M, Olsson M, Adlercreutz H, Sandin S, Weiderpass E . Dietary phytoestrogens are not associated with risk of overall breast cancer but diets rich in coumestrol are inversely associated with risk of estrogen receptor and progesterone receptor negative breast tumors in Swedish women. J Nutr 2008; 138: 938–945.
Bandera EV, Williams MG, Sima C, Bayuga S, Pulick K, Wilcox H et al. Phytoestrogen consumption and endometrial cancer risk: a population-based case-control study in New Jersey. Cancer Causes Control 2009; 20: 1117–1127.
Peeters PH, Slimani N, van der Schouw YT, Grace PB, Navarro C, Tjonneland A et al. Variations in plasma phytoestrogen concentrations in European adults. J Nutr 2007; 137: 1294–1300.
Zamora-Ros R, Andres-Lacueva C, Lamuela-Raventos RM, Berenguer T, Jakszyn P, Barricarte A et al. Estimation of dietary sources and flavonoid intake in a Spanish adult population (EPIC-Spain). J Am Diet Assoc 2010; 110: 390–398.
Lagiou P, Samoli E, Lagiou A, Peterson J, Tzonou A, Dwyer J et al. Flavonoids, vitamin C and adenocarcinoma of the stomach. Cancer Causes Control 2004; 15: 67–72.
Rossi M, Garavello W, Talamini R, Negri E, Bosetti C, Dal Maso L et al. Flavonoids and the risk of oral and pharyngeal cancer: a case-control study from Italy. Cancer Epidemiol Biomarkers Prev 2007; 16: 1621–1625.
Dilis V, Trichopoulou A . Antioxidant intakes and food sources in Greek adults. J Nutr 2010; 140: 1274–1279.
Travis RC, Allen NE, Appleby PN, Spencer EA, Roddam AW, Key TJ . A prospective study of vegetarianism and isoflavone intake in relation to breast cancer risk in British women. Int J Cancer 2008; 122: 705–710.
Chun OK, Chung SJ, Song WO . Estimated dietary flavonoid intake and major food sources of US adults. J Nutr 2007; 137: 1244–1252.
Hirose K, Imaeda N, Tokudome Y, Goto C, Wakai K, Matsuo K et al. Soybean products and reduction of breast cancer risk: a case-control study in Japan. Br J Cancer 2005; 93: 15–22.
Iwasaki M, Hamada GS, Nishimoto IN, Netto MM, Motola J, Laginha FM et al. Isoflavone, polymorphisms in estrogen receptor genes and breast cancer risk in case-control studies in Japanese, Japanese Brazilians and non-Japanese Brazilians. Cancer Sci 2009; 100: 927–933.
Lee SA, Wen W, Xiang YB, Barnes S, Liu D, Cai Q et al. Assessment of dietary isoflavone intake among middle-aged Chinese men. J Nutr 2007; 137: 1011–1016.
Frankenfeld CL, Lampe JW, Shannon J, Gao DL, Ray RM, Prunty J et al. Frequency of soy food consumption and serum isoflavone concentrations among Chinese women in Shanghai. Public Health Nutr 2004; 7: 765–772.
Rice MM, LaCroix AZ, Lampe JW, van Belle G, Kestin M, Sumitani M et al. Dietary soy isoflavone intake in older Japanese American women. Public Health Nutr 2001; 4: 943–952.
Pellegrini N, Valtuena S, Ardigo D, Brighenti F, Franzini L, Del Rio D et al. Intake of the plant lignans matairesinol, secoisolariciresinol, pinoresinol, and lariciresinol in relation to vascular inflammation and endothelial dysfunction in middle age-elderly men and post-menopausal women living in Northern Italy. Nutr Metab Cardiovasc Dis 2010; 20: 64–71.
Nurmi T, Mursu J, Penalvo JL, Poulsen HE, Voutilainen S . Dietary intake and urinary excretion of lignans in Finnish men. Br J Nutr 2010; 103: 677–685.
Hernandez-Ramirez RU, Galvan-Portillo MV, Ward MH, Agudo A, Gonzalez CA, Onate-Ocana LF et al. Dietary intake of polyphenols, nitrate and nitrite and gastric cancer risk in Mexico City. Int J Cancer 2009; 125: 1424–1430.
Bhakta D, Higgins CD, Sevak L, Mangtani P, Adlercreutz H, McMichael AJ et al. Phyto-oestrogen intake and plasma concentrations in South Asian and native British women resident in England. Br J Nutr 2006; 95: 1150–1158.
French MR, Thompson LU, Hawker GA . Validation of a phytoestrogen food frequency questionnaire with urinary concentrations of isoflavones and lignan metabolites in premenopausal women. J Am Coll Nutr 2007; 26: 76–82.
Hjartaker A, Lagiou A, Slimani N, Lund E, Chirlaque MD, Vasilopoulou E et al. Consumption of dairy products in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort: data from 35 955 24-hour dietary recalls in 10 European countries. Public Health Nutr 2002; 5: 1259–1271.
Horn-Ross PL, John EM, Canchola AJ, Stewart SL, Lee MM . Phytoestrogen intake and endometrial cancer risk. J Natl Cancer Inst 2003; 95: 1158–1164.
Yang L, Parkin DM, Whelan S, Zhang S, Chen Y, Lu F et al. Statistics on cancer in China: cancer registration in 2002. Eur J Cancer Prev 2005; 14: 329–335.
Manach C, Williamson G, Morand C, Scalbert A, Remesy C . Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am J Clin Nutr 2005; 81: 230S–242S.
Williamson G, Manach C . Bioavailability and bioefficacy of polyphenols in humans. II. Review of 93 intervention studies. Am J Clin Nutr 2005; 81: 243S–255S.
Ward HA, Kuhnle GG . Phytoestrogen consumption and association with breast, prostate and colorectal cancer in EPIC Norfolk. Arch Biochem Biophys 2010; 501: 170–175.
Yan L, Spitznagel EL . Soy consumption and prostate cancer risk in men: a revisit of a meta-analysis. Am J Clin Nutr 2009; 89: 1155–1163.
Slimani N, Fahey M, Welch AA, Wirfalt E, Stripp C, Bergstrom E et al. Diversity of dietary patterns observed in the European Prospective Investigation into Cancer and Nutrition (EPIC) project. Public Health Nutr 2002; 5: 1311–1328.
Slimani N, Bingham S, Runswick S, Ferrari P, Day NE, Welch AA et al. Group level validation of protein intakes estimated by 24-hour diet recall and dietary questionnaires against 24-hour urinary nitrogen in the European Prospective Investigation into Cancer and Nutrition (EPIC) calibration study. Cancer Epidemiol Biomarkers Prev 2003; 12: 784–795.
Skeie G, Braaten T, Hjartaker A, Lentjes M, Amiano P, Jakszyn P et al. Use of dietary supplements in the European Prospective Investigation into Cancer and Nutrition calibration study. Eur J Clin Nutr 2009; 63(Suppl 4): S226–S238.
Acknowledgements
This work was carried out with the financial support of the European Commission: Public Health and Consumer Protection Directorate 1993 to 2004; Research Directorate-General 2005; Ligue contre le Cancer, Institut Gustave Roussy, Mutuelle Générale de l’Education Nationale, Institut National de la Santé et de la Recherche Médicale (INSERM; France); German Federal Ministry of Education and Research; Danish Cancer Society: Health Research Fund (FIS) of the Spanish Ministry of Health (RTICC DR06/0020); the participating regional governments and institutions of Spain; Cancer Research UK; Medical Research Council, UK; the Stroke Association, UK; British Heart Foundation; Department of Health, UK; Food Standards Agency, UK; the Wellcome Trust, UK; the Stavros Niarchos Foundation and the Hellenic Health Foundation; Italian Association for Research on Cancer; Compagnia San Paolo, Italy; Dutch Ministry of Public Health, Welfare and Sports; Dutch Ministry of Health; Dutch Prevention Funds; LK Research Funds; Dutch ZON (Zorg Onderzoek Nederland); World Cancer Research Fund (WCRF); Swedish Cancer Society; Swedish Scientific Council; Regional Government of Skane, Sweden; Nordforsk - Centre of Excellence programe HELGA; Some authors are partners of ECNIS, a network of excellence of the 6FP of the EC. RZR is thankful for a postdoctoral programe Fondo de Investigación Sanitaria (FIS; no. CD09/00 133) from the Spanish Ministry of Science and Innovation. We thank Raul M. García for developing an application to link the FCDB and the 24-HDR. We would also like to thank Marleen Lentjes, Veronica van Scheltinga, Alison McTaggart and Amit Bhaniani for their invaluable contributions to the creation of the EPIC-Norfolk phytoestrogen database.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Contributors: RZ-R and CAG designed the research; RZ-R. and VK conducted the research; RZ-R and LL-B performed the statistical analysis; RZ-R wrote the manuscript; all authors read, critically reviewed and approved the final manuscript.
Rights and permissions
About this article
Cite this article
Zamora-Ros, R., Knaze, V., Luján-Barroso, L. et al. Dietary intakes and food sources of phytoestrogens in the European Prospective Investigation into Cancer and Nutrition (EPIC) 24-hour dietary recall cohort. Eur J Clin Nutr 66, 932–941 (2012). https://doi.org/10.1038/ejcn.2012.36
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2012.36
Keywords
This article is cited by
-
Gender-specific effects of soybean consumption on cardiovascular events in elderly individuals from rural Northeast China – a prospective cohort study
BMC Geriatrics (2023)
-
Estimated dietary intake of polyphenols from cereal foods and associated lifestyle and demographic factors in the Melbourne Collaborative Cohort Study
Scientific Reports (2023)
-
Dietary factors associated with gastric cancer - a review
Translational Medicine Communications (2022)
-
Soy intake and chronic disease risk: findings from prospective cohort studies in Japan
European Journal of Clinical Nutrition (2021)
-
Intake of isoflavones reduces the risk of all-cause mortality in middle-aged Japanese
European Journal of Clinical Nutrition (2021)