Abstract
Background/objective:
Dietary assessment in children is difficult, suggesting a need to develop more objective biomarkers of intake. Resonance Raman spectroscopy (RRS) is a non-invasive, validated method of measuring carotenoid status in skin as a biomarker of fruit/vegetable intake. The purpose of this study was to examine the feasibility of using RRS in preschool children, to describe inter-individual variability in skin carotenoid status and to identify factors associated with the biomarker in this population.
Subjects/methods:
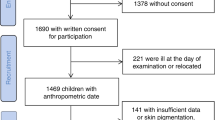
We conducted a cross-sectional study of 381 economically disadvantaged preschoolers in urban centers in Connecticut (USA). In all, 85.5% were black non-Hispanic or Hispanic/Latino, and 14.1% were obese and 16.9% were overweight by age- and sex-specific body mass index (BMI) percentiles. Children had their skin carotenoid status assessed by RRS in the palm of the hand. Fruit/vegetable consumption was assessed by a brief parent/guardian-completed food frequency screener and a liking survey.
Results:
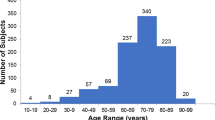
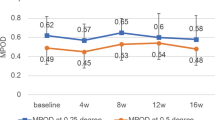
We observed inter-individual variation in RRS values that was nearly normally distributed. In multiple regression analysis, higher carotenoid status, measured by RRS, was positively associated with fruit/vegetable consumption (P=0.02) and fruit/vegetable preference (P<0.01). Lower carotenoid status was observed among younger children, those participating in the US Supplemental Nutrition Assistance Program, and those with greater adiposity (P<0.05 for all).
Conclusions:
We observed wide variability in skin carotenoid status in a population of young children, as assessed by RRS. Parent-reported fruit/vegetable intake and several demographic factors were significantly associated with RRS-measured skin carotenoid status. We recommend further development of this biomarker in children, including evaluating response to controlled interventions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Livingstone MB, Robson PJ, Wallace JM : Issues in dietary intake assessment of children and adolescents. Br J Nutr 2004; 92 (Suppl 2), S213–S222.
Baranowski T, Domel SB : A cognitive model of children's reporting of food intake. Am J Clin Nutr 1994; 59 (1 Suppl), 212S–217S.
Livingstone MB, Robson PJ : Measurement of dietary intake in children. Proc Nutr Soc 2000; 59, 279–293.
Kirks BA, Wolff HK : A comparison of methods for plate waste determinations. J Am Diet Assoc 1985; 85, 328–331.
Martin CK, Newton Jr RL, Anton SD, Allen HR, Alfonso A, Han H et al. Measurement of children's food intake with digital photography and the effects of second servings upon food intake. Eat Behav 2007; 8, 148–156.
Yatsuya H, Ohwaki A, Tamakoshi K, Wakai K, Koide K, Otsuka R et al. Reproducibility and validity of a simple checklist-type questionnaire for food intake and dietary behavior. J Epidemiol 2003; 13, 235–245.
Duffy VB, Hayes JE, Sullivan BS, Faghri P : Surveying food and beverage liking: a tool for epidemiological studies to connect chemosensation with health outcomes. Ann NY Acad Sci 2009; 1170, 558–568.
Michaud DS, Feskanich D, Rimm EB, Colditz GA, Speizer FE, Willett WC et al. Intake of specific carotenoids and risk of lung cancer in 2 prospective US cohorts. Am J Clin Nutr 2000; 72, 990–997.
Zhang M, Holman CD, Binns CW : Intake of specific carotenoids and the risk of epithelial ovarian cancer. Br J Nutr 2007; 98, 187–193.
Giovannucci E, Ascherio A, Rimm EB, Stampfer MJ, Colditz GA, Willett WC : Intake of carotenoids and retinol in relation to risk of prostate cancer. J Natl Cancer Inst 1995; 87, 1767–1776.
Le Marchand L, Hankin JH, Kolonel LN, Beecher GR, Wilkens LR, Zhao LP : Intake of specific carotenoids and lung cancer risk. Cancer Epidemiol Biomarkers Prev 1993; 2, 183–187.
Wang L, Gaziano JM, Norkus EP, Buring JE, Sesso HD : Associations of plasma carotenoids with risk factors and biomarkers related to cardiovascular disease in middle-aged and older women. Am J Clin Nutr 2008; 88, 747–754.
Voutilainen S, Nurmi T, Mursu J, Rissanen TH : Carotenoids and cardiovascular health. Am J Clin Nutr 2006; 83, 1265–1271.
Ito Y, Kurata M, Suzuki K, Hamajima N, Hishida H, Aoki K : Cardiovascular disease mortality and serum carotenoid levels: a Japanese population-based follow-up study. J Epidemiol 2006; 16, 154–160.
SanGiovanni JP, Chew EY, Clemons TE, Ferris III FL, Gensler G, Lindblad AS et al. The relationship of dietary carotenoid and vitamin A, E, and C intake with age-related macular degeneration in a case-control study: AREDS Report No. 22. Arch Ophthalmol 2007; 125, 1225–1232.
Seddon JM, Ajani UA, Sperduto RD, Hiller R, Blair N, Burton TC et al. Dietary carotenoids, vitamins A, C, and E, and advanced age-related macular degeneration. Eye Disease Case-Control Study Group. JAMA 1994; 272, 1413–1420.
Ray AL, Semba RD, Walston J, Ferrucci L, Cappola AR, Ricks MO et al. Low serum selenium and total carotenoids predict mortality among older women living in the community: the women's health and aging studies. J Nutr 2006; 136, 172–176.
Institute of Medicine, National Academy of Sciences; Food and Nutrition Board, Panel on Dietary Antioxidants and Related Compounds. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. National Academy Press: Washington, DC 2000.
Natarajan L, Flatt SW, Sun X, Gamst AC, Major JM, Rock CL et al. Validity and systematic error in measuring carotenoid consumption with dietary self-report instruments. Am J Epidemiol 2006; 163, 770–778.
Parker RS : Analysis of carotenoids in human plasma and tissues. Methods Enzymol 1993; 214, 86–93.
Karppi J, Nurmi T, Olmedilla-Alonso B, Granado-Lorencio F, Nyyssonen K : Simultaneous measurement of retinol, alpha-tocopherol and six carotenoids in human plasma by using an isocratic reversed-phase HPLC method. J Chromatogr B Analyt Technol Biomed Life Sci 2008; 867, 226–232.
Hata TR, Scholz TA, Ermakov IV, McClane RW, Khachik F, Gellermann W et al. Non-invasive Raman spectroscopic detection of carotenoids in human skin. J Invest Dermatol 2000; 115, 441–448.
Ermakov IV, Sharifzadeh M, Ermakova M, Gellermann W : Resonance Raman detection of carotenoid antioxidants in living human tissue. J Biomed Opt 2005; 10, 064028.
Ermakov IV, Ermakova MR, McClane RW, Gellermann W : Resonance Raman detection of carotenoid antioxidants in living human tissues. Opt Lett 2001; 26, 1179–1181.
Mayne ST, Cartmel B, Scarmo S, Lin H, Leffell DJ, Welch E et al. Noninvasive assessment of dermal carotenoids as a biomarker of fruit and vegetable intake. Am J Clin Nutr 2010; 92, 794–800.
Ermakov IV, Gellermann W : Validation model for Raman based skin carotenoid detection. Arch Biochem Biophys 2010; 504, 40–49.
Rerksuppaphol S, Rerksuppaphol L : Effect of fruit and vegetable intake on skin carotenoid detected by non-invasive Raman spectroscopy. J Med Assoc Thai 2006; 89, 1206–1212.
Darvin M, Fluhr J, Caspers P, van der Pool A, Richter H, Patzelt A et al. In vivo distribution of carotenoids in different anatomical locations of human skin: comparative assessment with two different Raman spectroscopy methods. Exp Dermatol 2009; 18, 1060–1063.
Meinke M, Darvin M, Vollert H, Lademann J : Bioavailability of natural carotenoids in human skin compared to blood. Eur J Pharm Biopharm 2010; 76, 269–274.
Blume-Peytavi U, Rolland A, Darvin ME, Constable A, Pineau I, Voit C et al. Cutaneous lycopene and beta-carotene levels measured by resonance Raman spectroscopy: high reliability and sensitivity to oral lactolycopene deprivation and supplementation. Eur J Pharm Biopharm 2009; 73, 187–194.
Kuczmarski RJ, Ogden CL, Grummer-Strawn LM, Flegal KM, Guo SS, Wei R et al. CDC growth charts: United States. In: Advance Data from Vital and Health Statistics. US Department of Health and Human Services, National Center for Health Statistics: Hyattsville, MD, 2000.
Cole TJ, Bellizzi MC, Flegal KM, Dietz WH : Establishing a standard definition for child overweight and obesity worldwide: international survey. BMJ 2000; 320, 1240–1243.
Agricultural Research Service, Food Surveys Research Group. USDA Food and Nutrient Database for Dietary Studies, 1.0. Agricultural Research Service, Food Surveys Research Group: Beltsville, MD 2004.
Schatzkin A, Kipnis V, Carroll RJ, Midthune D, Subar AF, Bingham S et al. A comparison of a food frequency questionnaire with a 24-h recall for use in an epidemiological cohort study: results from the biomarker-based Observing Protein and Energy Nutrition (OPEN) study. Int J Epidemiol 2003; 32, 1054–1062.
Ermakov IV, Ermakova MR, Gellermann W, Lademann J : Noninvasive selective detection of lycopene and beta-carotene in human skin using Raman spectroscopy. J Biomed Opt 2004; 9, 332–338.
Neuhouser ML, Rock CL, Eldridge AL, Kristal AR, Patterson RE, Cooper DA et al. Serum concentrations of retinol, alpha-tocopherol and the carotenoids are influenced by diet, race and obesity in a sample of healthy adolescents. J Nutr 2001; 131, 2184–2191.
Virtanen SM, van't Veer P, Kok F, Kardinaal AF, Aro A : Predictors of adipose tissue carotenoid and retinol levels in nine countries. The EURAMIC Study. Am J Epidemiol 1996; 144, 968–979.
Strauss RS : Comparison of serum concentrations of alpha-tocopherol and beta-carotene in a cross-sectional sample of obese and nonobese children (NHANES III). National Health and Nutrition Examination Survey. J Pediatr 1999; 134, 160–165.
Brady WE, Mares-Perlman JA, Bowen P, Stacewicz-Sapuntzakis M : Human serum carotenoid concentrations are related to physiologic and lifestyle factors. J Nutr 1996; 126, 129–137.
Parker RS : Carotenoids in human blood and tissues. J Nutr 1989; 119, 101–104.
Dibsdall LA, Lambert N, Bobbin RF, Frewer LJ : Low-income consumers' attitudes and behaviour towards access, availability and motivation to eat fruit and vegetables. Public Health Nutr 2003; 6, 159–168.
Rose D, Richards R : Food store access and household fruit and vegetable use among participants in the US Food Stamp Program. Public Health Nutr 2004; 7, 1081–1088.
Acknowledgements
We thank Angela Corcoran for her contributions to data collection, and the preschool staff, children and parents who participated in this study. This work was supported by funding from R01 CA096838 (STM) and American Diabetes Association (VBD); Sub-Award No. A 05202 and M03A00158 (IVE and WG), R01 EY11600 (PSB) and an unrestricted departmental grant from Research to Prevent Blindness (New York, NY) (PSB).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on European Journal of Clinical Nutrition website
Supplementary information
Rights and permissions
About this article
Cite this article
Scarmo, S., Henebery, K., Peracchio, H. et al. Skin carotenoid status measured by resonance Raman spectroscopy as a biomarker of fruit and vegetable intake in preschool children. Eur J Clin Nutr 66, 555–560 (2012). https://doi.org/10.1038/ejcn.2012.31
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2012.31
Keywords
This article is cited by
-
Preschool- and childcare center-based interventions to increase fruit and vegetable intake in preschool children in the United States: a systematic review of effectiveness and behavior change techniques
International Journal of Behavioral Nutrition and Physical Activity (2023)
-
Obesity prevention practices in early care and education settings: an adaptive implementation trial
Implementation Science (2022)
-
Stakeholder selected strategies for obesity prevention in childcare: results from a small-scale cluster randomized hybrid type III trial
Implementation Science (2021)
-
Community supported agriculture plus nutrition education improves skills, self-efficacy, and eating behaviors among low-income caregivers but not their children: a randomized controlled trial
International Journal of Behavioral Nutrition and Physical Activity (2021)
-
Serum carotenoids are strongly associated with dermal carotenoids but not self-reported fruit and vegetable intake among overweight and obese women
International Journal of Behavioral Nutrition and Physical Activity (2019)