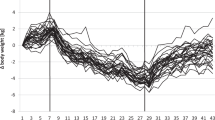
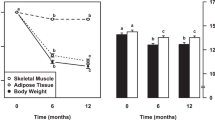
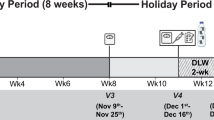
Abstract
Weight changes in response to a change in energy intake are smaller than calculated from the excess or deficit of energy intake. Digestion efficiency is not affected by intake level when consuming the same diet. Over- or underfeeding induces an increase or decrease in energy expenditure. Intake-induced expenditure changes are largely explained by proportional changes in diet-induced energy expenditure, in activity-induced energy expenditure and in maintenance expenditure as a function of changes in body weight and body composition. Additionally, underfeeding causes a metabolic adaptation as reflected in a reduction of maintenance expenditure below predicted values and defined as adaptive thermogenesis. Thus, alternating overfeeding and underfeeding with an iso-energetic amount results in a positive energy balance. The latter might be one of the explanations for the increasing incidence of obesity in our current society with an ample food supply.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Pasquet P, Brigant L, Froment A, Koppert GA, Bard D, De Garine I et al. Massive overfeeding and energy balance in man: the Guru Walla model. Am J Clin Nutr 1992; 56: 483–490.
Westerterp KR . Limits to sustainable metabolic rate. J Exp Biol 2001; 204: 3183–3187.
Van Es AJH, Vogt JE, Niessen C, Veth J, Rodenburg L, Teeuwse V et al. Human energy metabolism below, near and above energy equilibrium. Br J Nutr 1984; 52: 429–444.
Webb P, Annis JF . Adaptation to overeating in lean and overweight men and women. Hum Nutr Clin Nutr 1983; 37C: 117–131.
Kallus SJ, Brandt LJ . The intestinal microbiota and obesity. J Clin Gastroenterol 2012; 46: 16–24.
Krajmalnik-Brown R, Ilhan ZE, Kang DW, DiBaise JK . Effects of gut microbes on nutrient absorption and energy regulation. Nutr Clin Pract 2012; 27: 201–214.
Tappy L . Thermic effect of food and sympathetic nervous system activity in humans. Reprod Nutr Rev 1996; 36: 391–397.
Westerterp KR . Diet induced energy expenditure. Nutr Metab 2004; 1: 5.
Thomas DM, Ciesla A, Levine JA, Stevens JG, Martin CK . A mathematical model of weight change with adaptation. Math Biosci Eng 2009; 6: 873–887.
Hall KD . Modeling metabolic adaptations and energy regulation in humans. Annu Rev Nutr 2012; 32: 35–54.
Flatt JP . The biochemistry of energy expenditure. In: Bray GA, (ed). Recent Advances in Obesity Research 2. Newman: London, UK, 1978 pp 211–228.
Westerterp KR, Donkers JHHLM, Fredrix EWHM, Boekhoudt P . Energy intake, physical activity and body weight: a simulation model. Br J Nutr 1995; 73: 337–347.
De Jonge L, Bray GA . The thermic effect of food and obesity: a critical review. Obes Res 1997; 6: 622–631.
Granata GP, Brandon LJ . The thermic effect of food and obesity: discrepant results and methodological variations. Nutr Rev 2002; 60: 223–233.
Speakman JR Doubly-labelled Water: Theory and Practice. Chapman & Hall: London, UK, 1997.
Plasqui G, Westerterp KR . Physical activity assessment with accelerometers: an evaluation against doubly labelled water. Obesity 2007; 15: 2371–2379.
Westerterp KR . Physical activity, food intake, and body weight regulation: insights from doubly labelled water studies. Nutr Rev 2010; 68: 148–154.
Westerterp KR, Saris WHM, Soeters PB, Ten Hoor F . Determinants of weight loss after vertical banded gastroplasty. Int J Obes 1991; 15: 529–534.
Kempen KPG, Saris WHM, Westerterp KR . Energy balance during an 8-wk energy-restricted diet with and without exercise in obese women. Am J Clin Nutr 1995; 62: 722–729.
Velthuis-te Wierik EJM, Westerterp KR, Van den Berg H . Impact of a moderately energy-restricted diet on energy metabolism and body composition in non-obese men. Int J Obes 1995; 19: 318–324.
Van Gemert WG, Westerterp KR, Van Acker BAC, Wagenmakers AJM, Halliday D, Greve JM et al. Energy, substrate and protein metabolism in morbid obesity before, during and after massive weight loss. Int J Obes 2000; 24: 711–718.
Weinsier RL, Hunter GR, Zuckerman PA, Redden DT, Darnell BE, Larson DE et al. Energy expenditure and free-living physical activity in black and white women: comparison before and after weight loss. Am J Clin Nutr 2000; 71: 1138–1146.
Martin CK, Das SK, Lindblad L, Racette SB, McCrory MA, Weiss EP et al. Effect of calorie restriction on the free-living physical activity levels of non-obese humans: results of three randomized trials. J Appl Physiol 2011; 110: 956–963.
Ekelund U, Åman J, Yngve A, Renman C, Westerterp K, Sjöström M . Physical activity but not energy expenditure is reduced in obese adolescents: a case-control study. Am J Clin Nutr 2002; 76: 935–941.
Wouters-Adriaens MP, Westerterp KR . Low resting energy expenditure in Asians can be attributed to body composition. Obesity 2008; 16: 2212–2216.
Major GC, Doucet E, Trayhurn P, Astrup A, Tremblay A . Clinical significance of adaptive thermogenesis. Int J Obes 2007; 31: 204–212.
Rosenbaum M, Hirsch J, Gallagher DA, Leibel RL . Long-term persistence of adaptive thermogenesis in subjects who have maintained a reduced body weight. Am J Clin Nutr 2008; 88: 906–912.
Schwartz A, Kuk JL, Lamothe G, Doucet E . Greater than predicted decrease in resting energy expenditure and weight loss: results from a systematic review. Obesity 2012; 20: 2307–2310.
Van Gemert WG, Westerterp KR, Greve JM, Soeters PB . Reduction of sleeping metabolic rate after vertical banded gastroplasty. Int J Obes 1998; 22: 343–348.
Johannsen DL, Knuth ND, Huizinga R, Rood JC, Ravussin R, Hall KD . Metabolic slowing with massive weight loss despite preservation of fat-free mass. J Clin Endocrinol Metab 2012; 97: 2489–2496.
Bosy-Westphal A, Kossel E, Goele K, Later W, Hitze B, Settler U et al. Contribution of individual organ mass loss to weight loss-associated decline in resting energy expenditure. Am J Clin Nutr 2009; 90: 993–1001.
Dulloo AG, Jeaquet J, Monani JP . How dieting makes some fatter: from a perspective of human body composition autoregulation. Proc Nutr Soc 2012; 71: 379–389.
Bouten CV, Van Marken Lichtenbelt WD, Westerterp KR . Body mass index and daily physical activity in anorexia nervosa. Med Sci Sports Exerc 1996; 28: 967–973.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no conflict of interest.
Rights and permissions
About this article
Cite this article
Westerterp, K. Metabolic adaptations to over—and underfeeding—still a matter of debate?. Eur J Clin Nutr 67, 443–445 (2013). https://doi.org/10.1038/ejcn.2012.187
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2012.187
Keywords
This article is cited by
-
The Influence of Energy Balance and Availability on Resting Metabolic Rate: Implications for Assessment and Future Research Directions
Sports Medicine (2023)
-
Substrate utilization and metabolic profile in response to overfeeding with a high-fat diet in South Asian and white men: a sedentary lifestyle study
International Journal of Obesity (2020)
-
Effect of Over- and Underfeeding on Body Composition and Related Metabolic Functions in Humans
Current Diabetes Reports (2019)
-
Physical activity, energy expenditure and sedentary parameters in overfeeding studies - a systematic review
BMC Public Health (2018)
-
Potential effects of fat mass and fat-free mass on energy intake in different states of energy balance
European Journal of Clinical Nutrition (2018)