Abstract
Background:
The contemporary American diet figures centrally in the pathogenesis of numerous chronic diseases—‘diseases of civilization’. We investigated in humans whether a diet similar to that consumed by our preagricultural hunter-gatherer ancestors (that is, a paleolithic type diet) confers health benefits.
Methods:
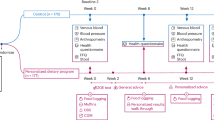
We performed an outpatient, metabolically controlled study, in nine nonobese sedentary healthy volunteers, ensuring no weight loss by daily weight. We compared the findings when the participants consumed their usual diet with those when they consumed a paleolithic type diet. The participants consumed their usual diet for 3 days, three ramp-up diets of increasing potassium and fiber for 7 days, then a paleolithic type diet comprising lean meat, fruits, vegetables and nuts, and excluding nonpaleolithic type foods, such as cereal grains, dairy or legumes, for 10 days. Outcomes included arterial blood pressure (BP); 24-h urine sodium and potassium excretion; plasma glucose and insulin areas under the curve (AUC) during a 2 h oral glucose tolerance test (OGTT); insulin sensitivity; plasma lipid concentrations; and brachial artery reactivity in response to ischemia.
Results:
Compared with the baseline (usual) diet, we observed (a) significant reductions in BP associated with improved arterial distensibility (−3.1±2.9, P=0.01 and +0.19±0.23, P=0.05);(b) significant reduction in plasma insulin vs time AUC, during the OGTT (P=0.006); and (c) large significant reductions in total cholesterol, low-density lipoproteins (LDL) and triglycerides (−0.8±0.6 (P=0.007), −0.7±0.5 (P=0.003) and −0.3±0.3 (P=0.01) mmol/l respectively). In all these measured variables, either eight or all nine participants had identical directional responses when switched to paleolithic type diet, that is, near consistently improved status of circulatory, carbohydrate and lipid metabolism/physiology.
Conclusions:
Even short-term consumption of a paleolithic type diet improves BP and glucose tolerance, decreases insulin secretion, increases insulin sensitivity and improves lipid profiles without weight loss in healthy sedentary humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Appel LJ, Sacks FM, Carey VJ, Obarzanek E, Swain JF, Miller III ER et al. (2005). Effects of protein, monounsaturated fat, and carbohydrate intake on blood pressure and serum lipids: results of the OmniHeart randomized trial. J Am Med Assoc 294, 2455–2464.
Brandt Niel . Evolutionary and Geological Timelines. http://www.talkorigins.org/origins/geo_timeline.html. 2007.
Bucher HC, Cook RJ, Guyatt GH, Lang JD, Cook DJ, Hatala R et al. (1996). Effects of dietary calcium supplementation on blood pressure. A meta-analysis of randomized controlled trials. J Am Med Assoc 275, 1016–1022.
Cohen M (1989). Health and the Rise of Civilization. Yale University Press: New Haven, pp 131–141.
Cordain L (1999). Cereal Grains: Humanity's Double-Edged Sword. In: Simopoulos AP (ed). Evolutionary Aspects of Nutrition and Health: Diet, Exercise, Genetics and Chronic Disease. S Karger: Switzerland, pp 19–73.
Cordain L, Eaton SB, Sebastian A, Mann N, Lindeberg S, Watkins BA et al. (2005). Origins and evolution of the Western diet: health implications for the 21st century. Am J Clin Nutr 81, 341–354.
Davidson MH, Toth PP (2004). Comparative effects of lipid-lowering therapies. Prog Cardiovasc Dis 47, 73–104.
Eaton SB, Cordain L (1997). Evolutionary aspects of diet: old genes, new fuels. Nutritional changes since agriculture. World Rev Nutr Diet 81, 26–37.
Eaton SB, Cordain L, Lindeberg S (2002). Evolutionary health promotion: a consideration of common counterarguments. Prev Med 34, 119–123.
Eaton SB, Eaton III SB (2000). Paleolithic vs modern diets—selected pathophysiological implications. Eur J Nutr 39, 67–70.
Eaton SB, Konner M (1985). Paleolithic nutrition. A consideration of its nature and current implications. N Engl J Med 312, 283–289.
Eaton SB, Konner M, Shostak M (1988). Stone agers in the fast lane; chronic degenerative diseases in evolutionary perspective. Am J Med 84, 739–749.
Jansson B (1990). Dietary, total body, and intracellular potassium-to-sodium ratios and their influence on cancer. Cancer Detect Prev 14, 563–565.
Lindeberg S, Cordain L, Eaton SB (2003a). The biological and clinical potential of a Palaeolithic diet: review. J Nutr Environ Med 13, 149–160.
Lindeberg S, Cordain L, Eaton BS (2003b). Biological and clinical potential of a palaeolithic diet. J Nutr Environ Med 13, 149–160.
Lindeberg S, Jonsson T, Granfeldt Y, Borgstrand E, Soffman J, Sjostrom K et al. (2007). A Palaeolithic diet improves glucose tolerance more than a Mediterranean-like diet in individuals with ischaemic heart disease. Diabetologia 50, 1795–1807.
Mann NJ (2004). Paleolithic nutrition: what can we learn from the past? Asia Pac J Clin Nutr 13, S17.
McDougall I, Brown FH, Fleagle JG (2005). Stratigraphic placement and age of modern humans from Kibish, Ethiopia. Nature 433, 733–736.
Neel JV (1999). When Some Fine Old Genes Meet a ‘New’ Environment. In: AP Simopoulos (ed). Evolutionary Aspects of Nutrition and Health: Diet, Exercise, Genetics and Chronic Disease. S Karger: Switzerland, pp 1–15.
O’Keefe Jr JH, Cordain L (2004). Cardiovascular disease resulting from a diet and lifestyle at odds with our Paleolithic genome: how to become a 21st-century hunter-gatherer. Mayo Clin Proc 79, 101–108.
Osterdahl M, Kocturk T, Koochek A, Wandell PE (2007). Effects of a short-term intervention with a paleolithic diet in healthy volunteers. Eur J Clin Nutr 62, 682–685.
Pryer J, Cappuccio FP, Elliott P (1995). Dietary calcium and blood pressure: a review of the observational studies. J Hum Hypertens 9, 597–604.
Sacks FM, Svetkey LP, Vollmer WM, Appel LJ, Bray GA, Harsha D et al. (2001). Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. DASH-Sodium Collaborative Research Group. N Engl J Med 344, 3–10.
Sebastian A, Frassetto LA, Sellmeyer DE, Merriam RL, Morris Jr RC (2002). Estimation of the net acid load of the diet of ancestral preagricultural Homo sapiens and their hominid ancestors. Am J Clin Nutr 76, 1308–1316.
Sebastian A, Frassetto LA, Sellmeyer DE, Morris Jr RC (2006). The evolution-informed optimal dietary potassium intake of human beings greatly exceeds current and recommended intakes. Semin Nephrol 26, 447–453.
Simopoulos AP (1999). The Center for Genetics NaH. In: AP Simopoulos (ed). Evolutionary Aspects of Nutrition and Health: Diet, Exercise, Genetics and Chronic Disease. Switzerland: S Karger, pp 1–99.
Steinfeld H, Gerber P, Wassenaar T, Castel V, Rosales M, de Haan C (2006). Livestock's Long Shadow: Environmental Issues and Options. Food and Agricultural Organization of the United Nations: Rome. ftp://ftp.fao.org/docrep/fao/010/A0701E/A0701E00.pdf. ISBN 9789251055717, pp 79–111.
Acknowledgements
We thank the staff of the Clinical Research Center for all the work and support that they provided during this study. This study was supported by the GCRC center Grant M01 RR00027
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Frassetto, L., Schloetter, M., Mietus-Synder, M. et al. Metabolic and physiologic improvements from consuming a paleolithic, hunter-gatherer type diet. Eur J Clin Nutr 63, 947–955 (2009). https://doi.org/10.1038/ejcn.2009.4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2009.4
Keywords
This article is cited by
-
Palaeolithic diet score and risk of breast cancer among postmenopausal women overall and by hormone receptor and histologic subtypes
European Journal of Clinical Nutrition (2023)
-
Plant-based diets and sports performance: a clinical review
Sport Sciences for Health (2023)
-
Archaic Food Uses of Large Graminoids in Agro Peligno Wetlands (Abruzzo, Central Italy) Compared With the European Ethnobotanical and Archaeological Literature
Wetlands (2022)
-
The associations of the Palaeolithic diet alone and in combination with lifestyle factors with type 2 diabetes and hypertension risks in women in the E3N prospective cohort
European Journal of Nutrition (2021)


